
Connect 2-Year Online Access for General, Organic, and Biochemistry
9th Edition
ISBN: 9781259677946
Author: Denniston
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 17, Problem 17.65QP
Interpretation Introduction
Interpretation:
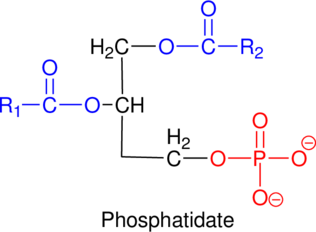
The glycerol-3-phosphate that contains the two moles of fatty acids and one possible phosphatidate structure has to be drawn.
Concept Introduction:
Phosphatidate: Fatty acids are esterified to hydroxyl on
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1) Calculate the longest and shortest wavelengths in the Lyman and Paschen series.
2) Calculate the ionization energy of He* and L2+ ions in their ground states.
3) Calculate the kinetic energy of the electron emitted upon irradiation of a H-atom in ground state by a 50-nm radiation.
Calculate the ionization energy of He+ and Li²+ ions in their ground states.
Thannnxxxxx sirrr
Ahehehehehejh27278283-4;*; shebehebbw $+$;$-;$-28283773838 hahhehdva
Plleeaasseee solllveeee question 3 andd thankss sirr, don't solve it by AI plleeaasseee don't use AI
Chapter 17 Solutions
Connect 2-Year Online Access for General, Organic, and Biochemistry
Ch. 17.3 - Write a complete equation for the acid hydrolysis...Ch. 17.3 - Prob. 17.6PPCh. 17.3 - Prob. 17.3QCh. 17.3 - Prob. 17.4QCh. 17.3 - Prob. 17.5QCh. 17.3 - Prob. 17.6QCh. 17.3 - Prob. 17.7QCh. 17.3 - Prob. 17.8QCh. 17.3 - Prob. 17.9QCh. 17.3 - Prob. 17.10Q
Ch. 17.3 - Prob. 17.11QCh. 17.3 - Prob. 17.12QCh. 17.4 - Prob. 17.13QCh. 17.4 - What is meant by the term fused ring?
Ch. 17.5 - Prob. 17.15QCh. 17.5 - Prob. 17.16QCh. 17 - Prob. 17.17QPCh. 17 - List the biological functions of lipids.
Ch. 17 - In terms of solubility, explain why a diet that...Ch. 17 - Why are lipids (triglycerides) such an efficient...Ch. 17 - What is the difference between a saturated and an...Ch. 17 - Prob. 17.22QPCh. 17 - As the length of the hydrocarbon chain of...Ch. 17 - As the number of carbon-carbon double bonds in...Ch. 17 - Explain the relationship between fatty acid chain...Ch. 17 - Explain the relationship you described in the...Ch. 17 - Prob. 17.27QPCh. 17 - Prob. 17.28QPCh. 17 - Prob. 17.29QPCh. 17 - Prob. 17.30QPCh. 17 - Write an equation for the esterification of...Ch. 17 - Prob. 17.32QPCh. 17 - Prob. 17.33QPCh. 17 - Write an equation for the acid hydrolysis of a...Ch. 17 - Prob. 17.35QPCh. 17 - Prob. 17.36QPCh. 17 - Using line formulas, write an equation for the...Ch. 17 - Using line formulas, write an equation for the...Ch. 17 - Write an equation for the base-catalyzed...Ch. 17 - Prob. 17.40QPCh. 17 - Write an equation for the esterification of...Ch. 17 - Prob. 17.42QPCh. 17 - Prob. 17.43QPCh. 17 - Prob. 17.44QPCh. 17 - Prob. 17.45QPCh. 17 - Prob. 17.46QPCh. 17 - Prob. 17.47QPCh. 17 - Prob. 17.48QPCh. 17 - What do the terms omega-3 and omega-6 indicate...Ch. 17 - Prob. 17.50QPCh. 17 - Prob. 17.51QPCh. 17 - Prob. 17.52QPCh. 17 - Prob. 17.53QPCh. 17 - Prob. 17.54QPCh. 17 - Prob. 17.55QPCh. 17 - Prob. 17.56QPCh. 17 - Prob. 17.57QPCh. 17 - Define the term phosphatidate.
Ch. 17 - Prob. 17.59QPCh. 17 - Prob. 17.60QPCh. 17 - Prob. 17.61QPCh. 17 - Prob. 17.62QPCh. 17 - Prob. 17.63QPCh. 17 - Prob. 17.64QPCh. 17 - Prob. 17.65QPCh. 17 - Prob. 17.66QPCh. 17 - Prob. 17.67QPCh. 17 - Prob. 17.68QPCh. 17 - Prob. 17.69QPCh. 17 - Prob. 17.70QPCh. 17 - Prob. 17.71QPCh. 17 - Prob. 17.72QPCh. 17 - Prob. 17.75QPCh. 17 - Prob. 17.78QPCh. 17 - Prob. 17.79QPCh. 17 - Prob. 17.80QPCh. 17 - Prob. 17.85QPCh. 17 - Prob. 17.86QPCh. 17 - Prob. 17.87QPCh. 17 - Prob. 17.88QPCh. 17 - Prob. 17.89QPCh. 17 - Prob. 17.90QPCh. 17 - Prob. 17.91QPCh. 17 - Prob. 17.92QPCh. 17 - Prob. 17.93QPCh. 17 - Prob. 17.94QPCh. 17 - Prob. 17.95QPCh. 17 - Prob. 17.96QPCh. 17 - Prob. 17.97QPCh. 17 - Prob. 17.98QPCh. 17 - Prob. 17.99QPCh. 17 - Prob. 17.100QPCh. 17 - Prob. 17.101QPCh. 17 - Prob. 17.103QPCh. 17 - What is the function of unsaturation in the...Ch. 17 - Prob. 1CPCh. 17 - Prob. 2CPCh. 17 - “Cholesterol is bad and should be eliminated from...Ch. 17 - Prob. 4CPCh. 17 - When a plant becomes cold-adapted, the composition...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Calculate the chemical shifts in 13C and 1H NMR for 4-chloropropiophenone ? Write structure and label hydrogens and carbonsarrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuu, don't solve it by AI plleeaasseeearrow_forward
- Please sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forward4. Read paragraph 4.15 from your textbook, use your calculated lattice energy values for CuO, CuCO3 and Cu(OH)2 an explain thermal decomposition reaction of malachite: Cu2CO3(OH)2 →2CuO + H2O + CO2 (3 points)arrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forward
- III O Organic Chemistry Using wedges and dashes in skeletal structures Draw a skeletal ("line") structure for each of the molecules below. Be sure your structures show the important difference between the molecules. key O O O O O CHON Cl jiii iiiiiiii You can drag the slider to rotate the molecules. Explanation Check Click and drag to start drawing a structure. Q Search X G ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Use F 3 W C 3/5arrow_forward3. Use Kapustinskii's equation and data from Table 4.10 in your textbook to calculate lattice energies of Cu(OH)2 and CuCO3 (4 points)arrow_forward2. Copper (II) oxide crystalizes in monoclinic unit cell (included below; blue spheres 2+ represent Cu²+, red - O²-). Use Kapustinski's equation (4.5) to calculate lattice energy for CuO. You will need some data from Resource section of your textbook (p.901). (4 points) CuOarrow_forward
- What is the IUPAC name of the following compound? OH (2S, 4R)-4-chloropentan-2-ol O (2R, 4R)-4-chloropentan-2-ol O (2R, 4S)-4-chloropentan-2-ol O(2S, 4S)-4-chloropentan-2-olarrow_forwardIn the answer box, type the number of maximum stereoisomers possible for the following compound. A H H COH OH = H C Br H.C OH CHarrow_forwardSelect the major product of the following reaction. Br Br₂, light D Br Br Br Brarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY