
Concept explainers
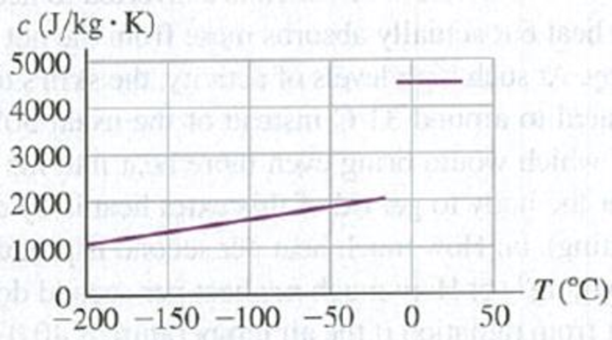
Careful measurements show that the specific heat of the solid phase depends on temperature (Fig. P17.117). How will the actual time needed for this cryoprotectant to come to equilibrium with the cold plate compare with the time predicted by using the values in the table? Assume that all values other than the specific heat (solid) are correct. The actual time (a) will be shorter; (b) will be longer; (c) will be the same; (d) depends on the density of the cryoprotectant.
Figure P17.117
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
University Physics with Modern Physics, Volume 2 (Chs. 21-37); Mastering Physics with Pearson eText -- ValuePack Access Card (14th Edition)
Additional Science Textbook Solutions
College Physics
Tutorials in Introductory Physics
Cosmic Perspective Fundamentals
Conceptual Physical Science (6th Edition)
Essential University Physics: Volume 1 (3rd Edition)
The Cosmic Perspective Fundamentals (2nd Edition)
- (a) What is the rate of heat conduction through the 3.00-cm-thick fur of a large animal having a I .40-m surface area? Assume that the animal's skin temperature is 32.0 , that the air temperature is 5.00 , and that has the same thermal conductivity as air. (b) What food intake will the animal need in one day to replace this heat transfer?arrow_forwardAn aluminum rod 0.500 m in length and with a cross sectional area of 2.50 cm2 is inserted into a thermally insulated vessel containing liquid helium at 4.20 K. The rod is initially at 3(H) K. (a) If one-halt of the rod is inserted into the helium, how many liters of helium boil off by the time the inserted half cools to 4.20 K? Assume the upper half does not yet cool, (b) If the circular surface of the upper end of the rod is maintained at 300 K. what is the approximate boil-off rate of liquid helium in liters per second after the lower half has reached 4.20 K? (Aluminum has thermal conductivity of 3 100 YV/m K at 4.20 K; ignore its temperature variation. The density of liquid helium is 125 kg/m3.)arrow_forward5 kg of water are contained in a closed,rigid tank at an initial pressure of 1000 kpa and a qualit of 40%. Heat transfer occurs until the tank contains only saturated vapor. Sketch the process on P-V diagram and T-V diagram and Determine the volume of the tank, in m^3, and the final pressure.arrow_forward
- In the lab, you have nails with mass Mn and temperature Tn.You submerge those nails in the water with mass Mw and temperature Tw. Specific heat capacities of nails Cn and water Cw areCn = 0.12 cal/gCCw = 1.00 cal/gC Presume no energy loss (all energy transfer is only between water and nails).Find the resulting temperature of nails in the water after temperature is equalized given the following values: Mn = 139 gTn = 119 CMw = 119 gTw = 27 C Enter 2 digits after decimal point.arrow_forwardA certain car has 14 L of coolant circulating at a temperature of 95 degrees Celsius through the engine’s cooling system. Assume that, in this normal condition, the coolant completely fills the 3.5 L volume of the aluminum radiator and the 10.5 L internal cavities within the aluminum engine. When a car overheats, the radiator, engine, and coolant expand and a small reservoir connected to the radiator catches any resultant coolant overflow. Estimate how much coolant overflows to the reservoir if the system goes from 95 degrees Celsius to 106 degrees Celsius. Model the radiator and engine as hollow shells of aluminum. The coefficient of volume expansion for coolant is 410x10^-6 degrees Celsiusarrow_forwardHelp.... Its wintertime and you are in need of portable electrical heater for your room and you are comparing two different products. The first heater has metal grills made of aluminum and has dimensions of 0.75 m x 0.05 m x 0.01 m while the second heater has metal grills made of iron and has dimensions of 1.00 m x 0.03 m x 0.01 m. You wish to heat your room from 10 degrees Celsius to a nice toasty 28 degrees Celsius and your wall outlet provides 120 V. 1) What is the resistance of the aluminum and iron heater at 28 degrees Celsius? 2) What is the current for the aluminum and iron heater operating at 28 degrees Celsius? 3) If you wish to operate your heater for 4 hours and the cost of electricity is $0.12 per kilowatt-hour, which heater has cheaper operational costs?arrow_forward
- At very low temperatures the molar heat capacity of rock salt varies with temperature according to Debye's T3 law: C = k where k = 1940 J/(mol K) and 0 = 281 K. (a) How much heat is required to raise the temperature of 1.50 mol of rock salt from 13.0 K to 36.0 K? (Hint: Use the change in thermal energy in the form dQ (b) What is the average molar heat capacity in this range? (c) What is the true molar heat capacity at 36.0 K? nCdT and integrate.)arrow_forwardAn experiment measures the temperature of a 200 g substance while steadily supplying heat to it. as shown the results of the experiment. What are (a) the specific heat of the liquid phase and (b) the heat of vaporization?arrow_forwardA certain car has 14 L of liquid coolant circulating at a temperature of 95 degrees Celsius through the engine’s cooling system. Assume that, in this normal condition, the coolant completely fills the 3.5 L volume of the aluminum radiator and the 10.5 L internal cavities within the aluminum engine. When a car overheats, the radiator, engine, and coolant expand and a small reservoir connected to the radiator catches any resultant coolant overflow. Estimate how much coolant overflows to the reservoir if the system goes from 95 degrees Celsius to 106 degrees Celsius. Model the radiator and engine as hollow shells of aluminum. The coefficient of volume expansion for coolant is 410x10^-6 degrees Celsiusarrow_forward
- The apparatus shown in Figure P11.10 was used by Joule to measure the mechanical equivalent of heat. Work is done on the water by a rotating paddle wheel, which is driven by two blocks falling at a constant speed. The temperature of the stirred water increases due to the friction between the water and the paddles. If the energy lost in the bearings and through the walls is neglected, then the loss in potential energy associated with the blocks equals the work done by the paddle wheel on the water. If each block has a mass of 1.50 kg and the insulated tank is filled with 200 g of water, what is the increase in temperature of the water after the blocks fall through a distance of 3.00 m?arrow_forwardOil enters the outer tube of an insulated concentric tube heat exchanger at a temperature of 500K and exits at a temperature of 400K. The mass flow rate of the oil is 5kg/s, and the oil has a specific heat of 1.91 kJ/kgK. Saturated liquid water at 100°C enters the inner tube and exits at 100°C with a quality of 0.2. Determine the mass flow rate of the water.arrow_forwardThe apparatus shown in Figure P11.12 was used by Joule to measure the mechanical equivalent of heat. Work is done on the water by a rotating paddle wheel, which is driven by two blocks falling at a constant speed. The temperature of the stirred water increases due to the friction between the water and the paddles. If the energy lost in the bearings and through the walls is neglected, then the loss in potential energy associated with the blocks equals the work done by the paddle wheel on the water. If each block has a mass of 1.50 kg and the insulated tank is filled with 0.200 kg of water, what is the increase in temperature of the water after the blocks fall through a distance of 3.00 m?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning



