
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
10th Edition
ISBN: 9781305957510
Author: ZUMDAHL, Steven S.; Zumdahl, Susan A.; DeCoste, Donald J.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 16Q
Consider the following potential energy plots:
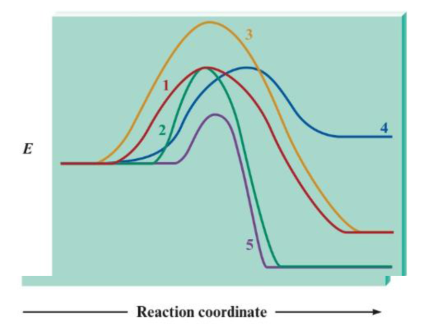
a. Rank the reactions from fastest to slowest and explain your answer. If any reactions have equal rates, explain why.
b. Label the reactions as endothermic or exothermic, and support your answer.
c. Rank the exothermic reactions from greatest to least change in potential energy. and support your answer.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Feedback: Your answer is incorrect.
Predict the major products of the following organic reaction:
CN
Δ
+
A ?
NC
Some important notes:
• Draw the major product, or products, of the reaction in the drawing area below.
• If there aren't any products, because no reaction will take place, check the box below the drawing area instead.
• Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers.
esc
Check
80
MH
F1
F2
F3
F4
F5
50
@
# C
%
95
€
Save For Later
Sub
2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C
A
DII
F6
F7
F8
7
*
8
Λ
&
6
F9
F10
9
0
4
Incorrect
Feedback: Your answer is incorrect.
Predict the major products of the following organic reaction:
ཤིགས་བྱ རྩ་ཅད་ཀྱིས་༢༩
+
Some important notes:
A
^ ?
• Draw the major product, or products, of the reaction in the drawing area below.
• If there aren't any products, because no reaction will take place, check the box below the drawing area instead.
• Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers.
E
Check
0
لا
Save For La
©2025 McGraw Hill LLC. All Rights Reserved. Terms of
All
F9
A
Predict the major products of the following organic reaction:
+
Δ
A ?
Some important notes:
• Draw the major product, or products, of the reaction in the drawing area below.
• If there aren't any products, because no reaction will take place, check the box below the drawing area instead.
• Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers.
Explanation
Check
Click and drag to start drawing a structure.
2025 McGraw Hill LLC. All Rights Reserved. Terms of Use
Priva
Chapter 17 Solutions
Student Solutions Manual for Zumdahl/Zumdahl/DeCoste?s Chemistry, 10th Edition
Ch. 17 - Define the following: a. spontaneous process b....Ch. 17 - What is the second law of thermodynamics? For any...Ch. 17 - What determines Ssurr for a process? To calculate...Ch. 17 - The free energy change, G, for a process at...Ch. 17 - What is the third law of thermodynamics? What are...Ch. 17 - What is the standard free energy change, G, for a...Ch. 17 - If you calculate a value for G for a reaction...Ch. 17 - Consider the equation G = G + RT ln(Q). What is...Ch. 17 - Even if G is negative, the reaction may not occur....Ch. 17 - Discuss the relationship between wmax and the...
Ch. 17 - For the process A(l) A(g), which direction is...Ch. 17 - For a liquid, which would you expect to be larger,...Ch. 17 - Gas A2 reacts with gas B2 to form gas AB at a...Ch. 17 - What types of experiments can be carried out to...Ch. 17 - A friend tells you, Free energy G and pressure P...Ch. 17 - Prob. 6ALQCh. 17 - Predict the sign of S for each of the following...Ch. 17 - Is Ssurr favorable or unfavorable for exothermic...Ch. 17 - At 1 atm, liquid water is heated above 100C. For...Ch. 17 - Prob. 10ALQCh. 17 - The synthesis of glucose directly from CO2 and H2O...Ch. 17 - When the environment is contaminated by a toxic or...Ch. 17 - Entropy has been described as times arrow....Ch. 17 - Human DNA contains almost twice as much...Ch. 17 - A mixture of hydrogen gas and chlorine gas remains...Ch. 17 - Consider the following potential energy plots: a....Ch. 17 - Ssurr is sometimes called the energy disorder...Ch. 17 - Given the following illustration, what can be said...Ch. 17 - The melting point for carbon diselenide (CSe2) is...Ch. 17 - The third law of thermodynamics states that the...Ch. 17 - The deciding factor on why HF is a weak acid and...Ch. 17 - List three different ways to calculate the...Ch. 17 - What information can be determined from G for a...Ch. 17 - Monochloroethane (C2H5Cl) can be produced by the...Ch. 17 - Consider the following relationships: G = 1, H =...Ch. 17 - Consider the reaction N2O2(g) 2NO2(g) where...Ch. 17 - At 1500 K, the process I2(g)2I(g)10atm10atm is not...Ch. 17 - Which of the following processes are spontaneous?...Ch. 17 - Which of the following processes are spontaneous?...Ch. 17 - Table 16-1 shows the possible arrangements of four...Ch. 17 - Consider the following illustration of six...Ch. 17 - Consider the following energy levels, each capable...Ch. 17 - Redo Exercise 29 with two particles A and B, which...Ch. 17 - Choose the substance with the larger positional...Ch. 17 - Which of the following involve an increase in the...Ch. 17 - Predict the sign of Ssurr for the following...Ch. 17 - Calculate Ssurr for the following reactions at 25C...Ch. 17 - Given the values of H and S, which of the...Ch. 17 - At what temperatures will the following processes...Ch. 17 - Ethanethiol (C2H5SH; also called ethyl mercaptan)...Ch. 17 - For mercury, the enthalpy of vaporization is 58.51...Ch. 17 - For ammonia (NH3), the enthalpy of fusion is 5.65...Ch. 17 - The enthalpy of vaporization of ethanol is 38.7...Ch. 17 - Predict the sign of S for each of the following...Ch. 17 - Predict the sign of S for each of the following...Ch. 17 - For each of the following pairs of substances,...Ch. 17 - For each of the following pairs, which substance...Ch. 17 - Predict the sign of S and then calculate S for...Ch. 17 - Predict the sign of S and then calculate S for...Ch. 17 - For the reaction C2H2(g)+4F2(g)2CF4(g)+H2(g) S is...Ch. 17 - For the reaction CS2(g)+3O2(g)CO2(g)+2SO2(g) S is...Ch. 17 - It is quite common for a solid to change from one...Ch. 17 - Two crystalline forms of white phosphorus are...Ch. 17 - Consider the reaction 2O(g)O2(g) a. Predict the...Ch. 17 - Hydrogen cyanide is produced industrially by the...Ch. 17 - From data in Appendix 4, calculate H, S, and G for...Ch. 17 - The major industrial use of hydrogen is in the...Ch. 17 - For the reaction at 298 K, 2NO2(g)N2O4(g) the...Ch. 17 - At 100C and 1.00 atm, H = 40.6 kJ/mol for the...Ch. 17 - For the sublimation of iodine at 25C I2(s) I2(g)...Ch. 17 - Given the following data:...Ch. 17 - Given the following data:...Ch. 17 - For the reaction SF4(g)+F2(g)SF6(g) the value of G...Ch. 17 - The value of G for the reaction...Ch. 17 - Consider the reaction...Ch. 17 - Consider the reaction 2POCl3(g)2PCl3(g)+O2(g) a....Ch. 17 - Using data from Appendix 4, calculate H, S and G...Ch. 17 - Consider two reactions for the production of...Ch. 17 - Using data from Appendix 4, calculate G for the...Ch. 17 - Using data from Appendix 4, calculate G for the...Ch. 17 - Consider the reaction 2NO2(g)N2O4(g) For each of...Ch. 17 - Consider the following reaction:...Ch. 17 - One of the reactions that destroys ozone in the...Ch. 17 - Hydrogen sulfide can be removed from natural gas...Ch. 17 - Consider the following reaction at 25.0C:...Ch. 17 - The standard free energies of formation and the...Ch. 17 - Calculate G forH2O(g)+12O2(g)H2O2(g) at 600. K,...Ch. 17 - The Ostwald process for the commercial production...Ch. 17 - Cells use the hydrolysis of adenosine...Ch. 17 - One reaction that occurs in human metabolism is...Ch. 17 - Consider the following reaction at 800. K:...Ch. 17 - Consider the following reaction at 298 K:...Ch. 17 - Consider the relationship In(K)=HRT+SR The...Ch. 17 - The equilibrium constant K for the reaction...Ch. 17 - A reaction has K = 1.9 1014 at 25C and K = 9.1 ...Ch. 17 - Using Appendix 4 and the following data, determine...Ch. 17 - Some water is placed in a coffee-cup calorimeter....Ch. 17 - A green plant synthesizes glucose by...Ch. 17 - When most biological enzymes are heated, they lose...Ch. 17 - Acrylonitrile is the starting material used in...Ch. 17 - Calculate the entropy change for the vaporization...Ch. 17 - As O2(l) is cooled at 1 atm, it freezes at 54.5 K...Ch. 17 - Consider the following reaction:...Ch. 17 - Using the following data, calculate the value of...Ch. 17 - Many biochemical reactions that occur in cells...Ch. 17 - Carbon monoxide is toxic because it bonds much...Ch. 17 - In the text, the equation G=G+RTIn(Q) was derived...Ch. 17 - Prob. 101AECh. 17 - Use the equation in Exercise 79 to determine H and...Ch. 17 - Prob. 103AECh. 17 - Consider the following diagram of free energy (G)...Ch. 17 - Prob. 105CWPCh. 17 - For rubidium Hvapo=69.0KJ/mol at 686C, its boiling...Ch. 17 - Given the thermodynamic data below, calculate S...Ch. 17 - Consider the reaction: H2S(g)+SO2(g)3S(g)+2H2O(l)...Ch. 17 - The following reaction occurs in pure water:...Ch. 17 - Prob. 110CWPCh. 17 - Consider the reaction: PCl3(g)+Cl2(g)PCl5(g) At...Ch. 17 - The equilibrium constant for a certain reaction...Ch. 17 - Consider two perfectly insulated vessels. Vessel 1...Ch. 17 - Liquid water at 25C is introduced into an...Ch. 17 - Using data from Appendix 4, calculate H, G, and K...Ch. 17 - Entropy can be calculated by a relationship...Ch. 17 - a. Using the free energy profile for a simple...Ch. 17 - Consider the reaction H2(g)+Br2(g)2HBr(g) where H...Ch. 17 - Consider the system A(g)B(g) at25C. a. Assuming...Ch. 17 - The equilibrium constant for a certain reaction...Ch. 17 - If wet silver carbonate is dried in a stream of...Ch. 17 - Carbon tetrachloride (CCl4) and benzene (C6H6)...Ch. 17 - Sodium chloride is added to water (at 25C) until...Ch. 17 - You have a 1.00-L sample of hot water (90.0C)...Ch. 17 - Consider a weak acid, HX. If a 0.10-M solution of...Ch. 17 - The vaporization of ethanol C2H5OH(l) C2H5OH(g)...Ch. 17 - Some nonelectrolyte solute (molar mass = 142...Ch. 17 - For the equilibrium A(g)+2B(g)C(g) the initial...Ch. 17 - What is the pH of a 0. 125-M solution of the weak...Ch. 17 - Impure nickel, refined by smelting sulfide ores in...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Give the IUPAC name for each compound.
Organic Chemistry
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
2. Which of the following is the best example of the use of a referent? _
a. A red bicycle
b. Big as a dump tru...
Physical Science
Some people compare DNA to a blueprint stored in the office of a construction company. Explain how this analogy...
Biology: Concepts and Investigations
56. Global Positioning System. Learn more about the global positioning system and its uses. Write a short repo...
The Cosmic Perspective (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- esc 2 Incorrect Feedback: Your answer is incorrect. Can the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ? A O • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. . If your answer is no, check the box under the drawing area instead. Check F1 ! @ X C Save For Later Submit Assignment 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility 80 et A ད 1 4 F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 F12 # $ 45 % A 6 87 & * 8 9 ) 0 + ||arrow_forwardCan the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? ?A Δ O • If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. • If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilit ku F11arrow_forward१ eq ine teaching and × + rn/takeAssignment/takeCovalentActivity.do?locator-assignment-take [Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is In progress mit Answer Retry Entire Group 5 more group attempts remaining Cengage Learning | Cengage Technical Support Save and Exitarrow_forward
- Draw the molecules.arrow_forwardDraw the mechanism for the acid-catalyzed dehydration of 2-methyl-hexan-2-ol with arrows please.arrow_forward. Draw the products for addition reactions (label as major or minor) of the reaction between 2-methyl-2-butene and with following reactants : Steps to follow : A. These are addition reactions you need to break a double bond and make two products if possible. B. As of Markovnikov rule the hydrogen should go to that double bond carbon which has more hydrogen to make stable products or major product. Here is the link for additional help : https://study.com/academy/answer/predict-the-major-and-minor-products-of-2-methyl- 2-butene-with-hbr-as-an-electrophilic-addition-reaction-include-the-intermediate- reactions.html H₂C CH3 H H3C CH3 2-methyl-2-butene CH3 Same structure CH3 IENCESarrow_forward
- Draw everything on a piece of paper including every single step and each name provided using carbons less than 3 please.arrow_forwardTopics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. H The IUPAC name isarrow_forward[Review Topics] [References] Write an acceptable IUPAC name for the compound below. (Only systematic names, not common names are accepted by this question.) Keep the information page open for feedback reference. The IUPAC name is Submit Answer Retry Entire Group 9 more group attempts remainingarrow_forward
- Please draw.arrow_forwardA chromatogram with ideal Gaussian bands has tR = 9.0 minutes and w1/2 = 2.0 minutes. Find the number of theoretical plates that are present, and calculate the height of each theoretical plate if the column is 10 centimeters long.arrow_forwardAn open tubular column has an inner diameter of 207 micrometers, and the thickness of the stationary phase on the inner wall is 0.50 micrometers. Unretained solute passes through in 63 seconds and a particular solute emerges at 433 seconds. Find the distribution constant for this solute and find the fraction of time spent in the stationary phase.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY