
Chemistry: A Molecular Approach
3rd Edition
ISBN: 9780321809247
Author: Nivaldo J. Tro
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 75E
Consider the titration curves (labeled a and b) for two weak acids, both titrated with 0.100 M NaOH.
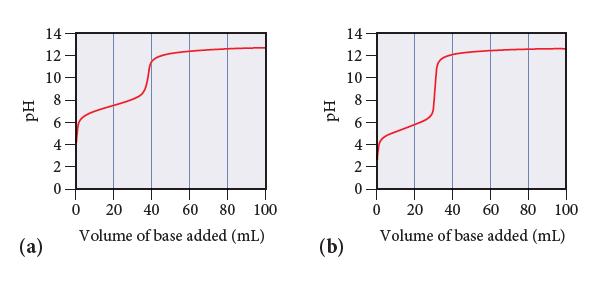
(i) Which acid solution is more concentrated?
(ii) Which acid has the larger Ka?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Electrochemistry. Briefly describe the Donnan potential.
Indicate what the Luther equation is used for?
Indicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.
Chapter 16 Solutions
Chemistry: A Molecular Approach
Ch. 16 - Prob. 1SAQCh. 16 - Q2. What is the pH of a buffer that is 0.120 M in...Ch. 16 - Q3. A buffer with a pH of 9.85 contains CH3NH2 and...Ch. 16 - Q4. A 500.0-mL buffer solution is 0.10 M in...Ch. 16 - Q5. Consider a buffer composed of the weak acid HA...Ch. 16 - Q6. Which combination is the best choice to...Ch. 16 - Q7. A 25.0-mL sample of an unknown HBr solution is...Ch. 16 - Q8. A 10.0-mL sample of 0.200 M hydrocyanic acid...Ch. 16 - Q9. A 20.0-mL sample of 0.150 M ethylamine is...Ch. 16 - Q10. Three 15.0-mL acid samples—0.10 M HA, 0.10 M...
Ch. 16 - Q11. A weak unknown monoprotic acid is titrated...Ch. 16 - Q12. Calculate the molar solubility of lead(II)...Ch. 16 - Q13. Calculate the molar solubility of magnesium...Ch. 16 - Q14. A solution is 0.025 M in Pb2 +. What minimum...Ch. 16 - Q15. Which compound is more soluble in an acidic...Ch. 16 - 1. What is the pH range of human blood? How is...Ch. 16 - 2. What is a buffer? How does a buffer work? How...Ch. 16 - 3. What is the common ion effect?
Ch. 16 - 4. What is the Henderson–Hasselbalch equation, and...Ch. 16 - 5. What is the pH of a buffer solution when the...Ch. 16 - 6. Suppose that a buffer contains equal amounts of...Ch. 16 - 7. How do you use the Henderson–Hasselbalch...Ch. 16 - 8. What factors influence the effectiveness of a...Ch. 16 - 9. What is the effective pH range of a buffer...Ch. 16 - 10. Describe acid–base titration. What is the...Ch. 16 - 11. The pH at the equivalence point of the...Ch. 16 - 12. The volume required to reach the equivalence...Ch. 16 - 13. In the titration of a strong acid with a...Ch. 16 - 14. In the titration of a weak acid with a strong...Ch. 16 - 15. The titration of a polyprotic acid with...Ch. 16 - 16. In the titration of a polyprotic acid, the...Ch. 16 - 17. What is the difference between the endpoint...Ch. 16 - 18. What is an indicator? How can an indicator...Ch. 16 - 19. What is the solubility product constant? Write...Ch. 16 - 20. What is molar solubility? How can you obtain...Ch. 16 - 21. How does a common ion affect the solubility of...Ch. 16 - 22. How is the solubility of an ionic compound...Ch. 16 - 23. For a given solution containing an ionic...Ch. 16 - 24. What is selective precipitation? Under which...Ch. 16 - 25. What is qualitative analysis? How does...Ch. 16 - 26. What are the main groups in the general...Ch. 16 - 27. In which of these solutions will HNO2 ionize...Ch. 16 - 28. A formic acid solution has a pH of 3.25. Which...Ch. 16 - 29. Solve an equilibrium problem (using an ICE...Ch. 16 - 30. Solve an equilibrium problem (using an ICE...Ch. 16 - 31. Calculate the percent ionization of a 0.15 M...Ch. 16 - 32. Calculate the percent ionization of a 0.13 M...Ch. 16 - 33. Solve an equilibrium problem (using an ICE...Ch. 16 - 34. Solve an equilibrium problem (using an ICE...Ch. 16 - 35. A buffer contains significant amounts of...Ch. 16 - 36. A buffer contains significant amounts of...Ch. 16 - Prob. 37ECh. 16 - Prob. 38ECh. 16 - 39. Use the Henderson–Hasselbalch equation to...Ch. 16 - 40. Use the Henderson–Hasselbalch equation to...Ch. 16 - 41. Calculate the pH of the solution that results...Ch. 16 - 42. Calculate the pH of the solution that results...Ch. 16 - 43. Calculate the ratio of NaF to HF required to...Ch. 16 - 44. Calculate the ratio of CH3NH2 to CH3NH3Cl...Ch. 16 - Prob. 45ECh. 16 - 46. What mass of ammonium chloride should you add...Ch. 16 - 47. A 250.0-mL buffer solution is 0.250 M in...Ch. 16 - 48. A 100.0-mL buffer solution is 0.175 M in HClO...Ch. 16 - Prob. 49ECh. 16 - 50. For each solution, calculate the initial and...Ch. 16 - Prob. 51ECh. 16 - 52. A 100.0-mL buffer solution is 0.100 M in NH3...Ch. 16 - 53. Determine whether or not the mixing of each...Ch. 16 - 54. Determine whether or not the mixing of each...Ch. 16 - 55. Blood is buffered by carbonic acid and the...Ch. 16 - 56. The fluids within cells are buffered by H2PO4–...Ch. 16 - 57. Which buffer system is the best choice to...Ch. 16 - Prob. 58ECh. 16 - 59. A 500.0-mL buffer solution is 0.100 M in HNO2...Ch. 16 - Prob. 60ECh. 16 - Prob. 61ECh. 16 - 62. Two 25.0-mL samples, one 0.100 M HCl and the...Ch. 16 - 63. Two 20.0-mL samples, one 0.200 M KOH and the...Ch. 16 - 64. The graphs labeled (a) and (b) show the...Ch. 16 - 65. Consider the curve shown here for the...Ch. 16 - 66. Consider the curve shown here for the...Ch. 16 - 67. Consider the titration of a 35.0-mL sample of...Ch. 16 - Prob. 68ECh. 16 - 69. Consider the titration of a 25.0-mL sample of...Ch. 16 - Prob. 70ECh. 16 - 71. Consider the titration of a 20.0-mL sample of...Ch. 16 - Prob. 72ECh. 16 - Prob. 73ECh. 16 - Prob. 74ECh. 16 - Consider the titration curves (labeled a and b)...Ch. 16 - Prob. 76ECh. 16 - Prob. 77ECh. 16 - 78. A 0.446-g sample of an unknown monoprotic acid...Ch. 16 - Prob. 79ECh. 16 - Prob. 80ECh. 16 - Prob. 81ECh. 16 - Prob. 82ECh. 16 - Prob. 83ECh. 16 - 84. Referring to Table 17.1, pick an indicator for...Ch. 16 - Prob. 85ECh. 16 - Prob. 86ECh. 16 - 87. Refer to the Ksp values in Table 17.2 to...Ch. 16 - 88. Refer to the Ksp values in Table 17.2 to...Ch. 16 - 89. Use the given molar solubilities in pure water...Ch. 16 - Prob. 90ECh. 16 - Prob. 91ECh. 16 - Prob. 92ECh. 16 - 93. Refer to the Ksp value from Table 17.2 to...Ch. 16 - Prob. 94ECh. 16 - 95. Calculate the molar solubility of barium...Ch. 16 - Prob. 96ECh. 16 - Prob. 97ECh. 16 - Prob. 98ECh. 16 - Prob. 99ECh. 16 - Prob. 100ECh. 16 - Prob. 101ECh. 16 - Prob. 102ECh. 16 - Prob. 103ECh. 16 - Prob. 104ECh. 16 - Prob. 105ECh. 16 - Prob. 106ECh. 16 - Prob. 107ECh. 16 - Prob. 108ECh. 16 - Prob. 109ECh. 16 - Prob. 110ECh. 16 - Prob. 111ECh. 16 - Prob. 112ECh. 16 - 113. A 150.0-mL solution contains 2.05 g of sodium...Ch. 16 - Prob. 114ECh. 16 - Prob. 115ECh. 16 - Prob. 116ECh. 16 - Prob. 117ECh. 16 - 118. A 250.0-mL buffer solution initially contains...Ch. 16 - 119. In analytical chemistry, bases used for...Ch. 16 - Prob. 120ECh. 16 - Prob. 121ECh. 16 - Prob. 122ECh. 16 - Prob. 123ECh. 16 - Prob. 124ECh. 16 - Prob. 125ECh. 16 - Prob. 126ECh. 16 - Prob. 127ECh. 16 - Prob. 128ECh. 16 - Prob. 129ECh. 16 - Prob. 130ECh. 16 - 131. The Kb of hydroxylamine, NH2OH, is 1.10 ×...Ch. 16 - 132. A 0.867-g sample of an unknown acid requires...Ch. 16 - Prob. 133ECh. 16 - Prob. 134ECh. 16 - 135. What relative masses of dimethyl amine and...Ch. 16 - Prob. 136ECh. 16 - Prob. 137ECh. 16 - Prob. 138ECh. 16 - 139. Since soap and detergent action is hindered...Ch. 16 - 140. A 0.558-g sample of a diprotic acid with a...Ch. 16 - 141. When excess solid Mg(OH)2 is shaken with 1.00...Ch. 16 - Prob. 142ECh. 16 - Prob. 143ECh. 16 - Prob. 144ECh. 16 - Prob. 145ECh. 16 - Prob. 146ECh. 16 - Prob. 147ECh. 16 - 148. What amount of HCl gas must be added to 1.00...Ch. 16 - 149. Without doing any calculations, determine if...Ch. 16 - 150. A buffer contains 0.10 mol of a weak acid and...Ch. 16 - Prob. 151ECh. 16 - Prob. 152ECh. 16 - Prob. 153E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Hint: In this case you must choose the best answer to demonstrate the stereochemistry of H2 addition. 1.03 2. (CH3)2S BIZ CH₂OH 2. DMS KMnO4, NaOH ΖΗ Pd or Pt (catalyst) HBr 20 1 HBr ROOR (peroxide) HO H-SO HC 12 11 10 BH, THE 2. H2O2, NaOH Brz cold HI 19 18 17 16 MCPBA 15 14 13 A Br H₂O BH3⚫THF Brz EtOH Pd or Ni (catalyst) D₂ (deuterium) 1. Os04 2. H2O2 CH3CO3H (peroxyacid) 1. MCPBA 2. H₂O* H B + H H H "H C H H Darrow_forwardExplain how Beer’s Law can be used to determine the concentration in a selected food sample. Provide examples.arrow_forward
- Explain the importance of having a sampling plan with respect to food analysis. Explain the importance of having a sampling plan with respect to food analysis. Provide examples.arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forwardBriefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY