
CNCT ORG CHEM 6 2020
6th Edition
ISBN: 9781266807244
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 47P
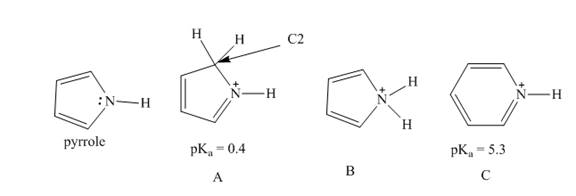
a. Explain why protonation of pyrrole occurs at
b. Explain why A is more acidic than C, the conjugate acid of pyridine.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the following two compounds, indicate and label where the electrophilic and nucleophilic
sites are.
요
N
Please correct answer and don't use Hand rating
None
Chapter 15 Solutions
CNCT ORG CHEM 6 2020
Ch. 15.2 - Prob. 1PCh. 15.2 - Problem 17.2 What orbitals are used to form the...Ch. 15.3 - Prob. 4PCh. 15.3 - Problem-17.5 What is the structure of propofol,...Ch. 15.6 - Prob. 7PCh. 15.8 - Prob. 8PCh. 15.8 - Prob. 11PCh. 15.8 - Prob. 12PCh. 15.8 - Problem 17.16 Rank the following compounds in...Ch. 15.8 - Problem 17.17 Draw the seven resonance structures...
Ch. 15 - 17.23 Name each compound and state how many lines...Ch. 15 - Prob. 21PCh. 15 - Prob. 22PCh. 15 - 17.27 Give the IUPAC name for each compounds.
a....Ch. 15 - 17.28 Draw a structure corresponding to each...Ch. 15 - 17.29 a. Draw the 14 constitutional isomers of...Ch. 15 - Prob. 26PCh. 15 - Prob. 27PCh. 15 - 17.38
How many electrons does C contain?
How...Ch. 15 - Prob. 36PCh. 15 - 17.40 Explain the observed rate of reactivity of...Ch. 15 - 17.41 Draw a stepwise mechanism for the following...Ch. 15 - Prob. 39PCh. 15 - 17.43 Draw additional resonance structures for...Ch. 15 - Prob. 41PCh. 15 - Prob. 42PCh. 15 - 17.46 Which compound in each pair is the stronger...Ch. 15 - 17.47 Treatment of indene with forms its...Ch. 15 - Prob. 45PCh. 15 - 17.49 Draw the conjugate bases of pyrrole and...Ch. 15 - 17.50 a. Explain why protonation of pyrrole occurs...Ch. 15 - Prob. 48PCh. 15 - Prob. 49PCh. 15 - 17.53 How many signals does each compound...Ch. 15 - 17.54 Which of the diethylbenzene isomers (ortho,...Ch. 15 - 17.55 Propose a structure consistent with each...Ch. 15 - 17.56 Propose a structure consistent with each...Ch. 15 - 17.57 Thymol (molecular formula ) is the major...Ch. 15 - 17.58 You have a sample of a compound of molecular...Ch. 15 - 17.59 Explain why tetrahydrofuran has a higher...Ch. 15 - 17.60 Rizatriptan (trade name Maxalt) is a...Ch. 15 - 17.61 Zolpidem (trade name Ambien) promotes the...Ch. 15 - 17.62 Answer the following questions about...Ch. 15 - 17.63 Stanozolol is an anabolic steroid that...Ch. 15 - Prob. 61P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Nonearrow_forward3. Propose a synthesis for the following transformation. Do not draw an arrow-pushing mechanism below, but make sure to draw the product of each proposed step (3 points). CN + En CNarrow_forward3) Propagation of uncertainty. Every measurement has uncertainty. In this problem, we'll evaluate the uncertainty in every step of a titration of potassium hydrogen phthalate (a common acid used in titrations, abbreviated KHP, formula CsH5KO4) with NaOH of an unknown concentration. The calculation that ultimately needs to be carried out is: concentration NaOH 1000 x mass KHP × purity KHP molar mass KHP x volume NaOH Measurements: a) You use a balance to weigh 0.3992 g of KHP. The uncertainty is ±0.15 mg (0.00015 g). b) You use a buret to slowly add NaOH to the KHP until it reaches the endpoint. It takes 18.73 mL of NaOH. The uncertainty of the burst is 0.03 mL.. c) The manufacturer states the purity of KHP is 100%±0.05%. d) Even though we don't think much about them, molar masses have uncertainty as well. The uncertainty comes from the distribution of isotopes, rather than random measurement error. The uncertainty in the elements composing KHP are: a. Carbon: b. Hydrogen: ±0.0008…arrow_forward
- Don't used hand raiting and don't used Ai solutionarrow_forwardHow would you use infrared spectroscopy to distinguish between the following pairs of constitutional isomers? (a) CH3C=CCH3 || and CH3CH2C=CH (b) CH3CCH=CHCH3 and CH3CCH2CH=CH2 Problem 12-41 The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can. (a) 100 Relative abundance (%) 80 60 60 40 200 20 (b) 100 Transmittance (%) 10 20 20 80- 60- 40- 20 40 60 80 100 120 140 m/z 500 4000 3500 3000 2500 2000 1500 Wavenumber (cm-1) 1000arrow_forwardPropagation of uncertainty. You have a stock solution certified by the manufacturer to contain 150.0±0.03 µg SO42-/mL. You would like to dilute it by a factor of 100 to obtain 1.500 µg/mL. Calculate the uncertainty in the two methods of dilution below. Use the following uncertainty values for glassware: Glassware Uncertainty (assume glassware has been calibrated and treat the values below as random error) 1.00 mL volumetric pipet 0.01 mL 10.00 mL volumetric pipet 0.02 mL 100.00 mL volumetric flask 0.08 mL Transfer 10.00 mL with a volumetric pipet and dilute it to 100 mL with a volumetric flask. Then take 10.00 mL of the resulting solution and dilute it a second time with a 100 mL flask. 2. Transfer 1.00 mL with a volumetric pipet and dilute it to 100 mL with a volumetric flask.arrow_forward
- Draw all resonance structures for the following ion: CH₂ Draw all resonance structures on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default. 2D ד CONT HD EXP CON ? 1 [1] Α 12 Marvin JS by Chemaxon A DOO H C N Br I UZ OSPFarrow_forwardWhat is the average mass of the 10 pennies? Report your value with correct significant figures. What is the error (uncertainty) associated with each mass measurement due to the equipment? What is the uncertainty associated with the average value? Note that the uncertainty of the balance will propagate throughout the calculation. What is the standard deviation of the 10 mass measurements? Explain the difference between the propagated uncertainty and the standard deviation. Which number would you use to describe the uncertainty in the measurement? Calculate the total mass of the pennies with associated uncertainty. Calculate the average density of a penny based on these data. Propagate the uncertainty values for both mass and volume in your calculations.arrow_forwardCan you help me and explain the answers please.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY