
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
12th Edition
ISBN: 9781119664635
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 12PP
PRACTICE PROBLEM 15.12
Predict the major product (or products) that would be obtained when each of the following compounds is nitrated:
(a)
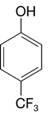
(b)
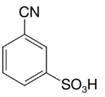
(c)
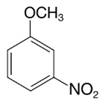
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the product formed when the compound shown below undergoes a reaction with MCPBA in CH2Cl2. MCPBA is meta-chloroperoxybenzoic acid.
k
https://app.aktiv.com
STARTING AMOUNT
6 58°F
Clear
+
F1
X
Dimensional Analysis - Aktiv Chemistry
Your Aktiv Learning trial expires on 02/25/25 at 02:14 PM
Question 19 of 22
Polyethylene terephthalate (PET) is used in plastic water bottles. A water bottle has a
mass of 14.0 grams. Given a density of 1.38 g/cm³, what is the volume of the
plastic used to make the water bottle in cm³ ?
ADD FACTOR
ANSWER
RESET
ว
100
14.0
0.01
10.1
1000
0.099
1.38
0.001
Q Search
F5
-O+
F6
F7
+
F3
F2
W
E
S4
ST
#3
F4
%
5
Y
R
S
&
7
cm³
g/cm³
g
ם
F8
* 00
8
F9
P
ل
DOD
S
F10
F11
F12
Insert
D
F
G
H
J
K
+ 11
A doctor gives a patient 10 Ci of beta radiation. How many betaparticles would the patient receive in 1 minute? (1 Ci = 3.7 x 1010d/s)
Chapter 15 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Ch. 15 - PRACTICE PROBLEM 15.1
Show how loss of a proton...Ch. 15 - Prob. 2PPCh. 15 - PRACTICE PROBLEM 15.3
Outline all steps in a...Ch. 15 - PRACTICE PROBLEM 15.4 Provide a mechanism that...Ch. 15 - Prob. 5PPCh. 15 - Prob. 6PPCh. 15 - Prob. 7PPCh. 15 - PRACTICE PROBLEM 15.8 Write resonance structures...Ch. 15 - PRACTICE PROBLEM 15.9
Provide a mechanism for the...Ch. 15 - PRACTICE PROBLEM 15.10 The trifluoromethyl group...
Ch. 15 - PRACTICE PROBLEM 15.11
Predict the major products...Ch. 15 - PRACTICE PROBLEM 15.12 Predict the major product...Ch. 15 - PRACTICE PROBLEM 15.13
Write mechanisms for the...Ch. 15 - Prob. 14PPCh. 15 - PRACTICE PROBLEM 15.15
Suppose you needed to...Ch. 15 - PRACTICE PROBLEM 15.16 1-Fluoro-2,4-dinitrobenzene...Ch. 15 - Prob. 17PPCh. 15 - PRACTICE PROBLEM 15.18
When...Ch. 15 - PRACTICE PROBLEM 15.19 Birch reduction of toluene...Ch. 15 - Prob. 20PCh. 15 - Prob. 21PCh. 15 - What monobromination product (or products) would...Ch. 15 - 15.23 Predict the major products of the following...Ch. 15 - Prob. 24PCh. 15 - 15.25 Starting with styrene, outline a synthesis...Ch. 15 - Prob. 26PCh. 15 - 15.27 Starting with aniline, outline a synthesis...Ch. 15 - Prob. 28PCh. 15 - Propose structures for compounds GI:Ch. 15 - 2,6-Dichlorophenol has been isolated from the...Ch. 15 - Prob. 31PCh. 15 - 15.32 Give structures (including stereochemistry...Ch. 15 - Provide a detailed mechanism for each of the...Ch. 15 - 15.34 Provide a detailed mechanism for the...Ch. 15 - Prob. 35PCh. 15 - Many polycyclic aromatic compounds have been...Ch. 15 - Prob. 37PCh. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Prob. 40PCh. 15 - Predict the product of the following reaction.Ch. 15 - 15.42 When m-chlorotoluene is treated with sodium...Ch. 15 - Prob. 43PCh. 15 - Prob. 44PCh. 15 - Prob. 45PCh. 15 - Prob. 46PCh. 15 - 15.47 Provide structures for compounds A and B:
Ch. 15 - Prob. 48PCh. 15 - 15.49 Treating cyclohexene with acetyl chloride...Ch. 15 - 15.50 The tert-butyl group can be used as a...Ch. 15 - 15.51 When toluene is sulfonated (concentrated )...Ch. 15 - Prob. 52PCh. 15 - 2-Methylnaphthalene can be synthesized from...Ch. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Prob. 57PCh. 15 - Prob. 58PCh. 15 - Furan undergoes electrophilic aromatic...Ch. 15 - A C-D bond is harder to break than a C-H bond,...Ch. 15 - 15.61 Acetanilide was subjected to the following...Ch. 15 - Prob. 62PCh. 15 - Prob. 63PCh. 15 - Prob. 64PCh. 15 - When compound C, which is often used to model a...Ch. 15 - Open the molecular model file for benzyne and...Ch. 15 - The structure of thyroxine, a thyroid hormone that...Ch. 15 - Prob. 2LGPCh. 15 - 3. Deduce the structures of compounds E–L in the...
Additional Science Textbook Solutions
Find more solutions based on key concepts
How can the freezing of water crack boulders?
Campbell Biology in Focus (2nd Edition)
What term is applied to the numerous almost circular depressions designated with the letter C?
Applications and Investigations in Earth Science (9th Edition)
12. A 2.0 cm × 2.0 cm parallel-plate capacitor has a 2.0 mm spacing. The electric field strength inside the cap...
College Physics: A Strategic Approach (3rd Edition)
Modified True/False 9. A giant bacterium that is large enough to be seen without a microscope is Selenomonas.
Microbiology with Diseases by Body System (5th Edition)
What is the composition of the extracellular matrix of bone tissue?
Principles of Anatomy and Physiology
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part C IN H N. Br₂ (2 equiv.) AlBr3 Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and + e (×) H± 12D T EXP. L CONT. דarrow_forward9. OA. Rank the expected boiling points of the compounds shown below from highest to lowest. Place your answer appropriately in the box. Only the answer in the box will be graded. (3) points) OH OH بر بد بدید 2 3arrow_forwardThere is an instrument in Johnson 334 that measures total-reflectance x-ray fluorescence (TXRF) to do elemental analysis (i.e., determine what elements are present in a sample). A researcher is preparing a to measure calcium content in a series of well water samples by TXRF with an internal standard of vanadium (atomic symbol: V). She has prepared a series of standard solutions to ensure a linear instrument response over the expected Ca concentration range of 40-80 ppm. The concentrations of Ca and V (ppm) and the instrument response (peak area, arbitrary units) are shown below. Also included is a sample spectrum. Equation 1 describes the response factor, K, relating the analyte signal (SA) and the standard signal (SIS) to their respective concentrations (CA and CIS). Ca, ppm V, ppm SCa, arb. units SV, arb. units 20.0 10.0 14375.11 14261.02 40.0 10.0 36182.15 17997.10 60.0 10.0 39275.74 12988.01 80.0 10.0 57530.75 14268.54 100.0…arrow_forward
- A mixture of 0.568 M H₂O, 0.438 M Cl₂O, and 0.710 M HClO are enclosed in a vessel at 25 °C. H₂O(g) + C₁₂O(g) = 2 HOCl(g) K = 0.0900 at 25°C с Calculate the equilibrium concentrations of each gas at 25 °C. [H₂O]= [C₁₂O]= [HOCI]= M Σ Marrow_forwardWhat units (if any) does the response factor (K) have? Does the response factor (K) depend upon how the concentration is expressed (e.g. molarity, ppm, ppb, etc.)?arrow_forwardProvide the structure, circle or draw, of the monomeric unit found in the biological polymeric materials given below. HO OH amylose OH OH 행 3 HO cellulose OH OH OH Ho HOarrow_forward
- OA. For the structure shown, rank the bond lengths (labeled a, b and c) from shortest to longest. Place your answer in the box. Only the answer in the box will be graded. (2 points) H -CH3 THe b Нarrow_forwardDon't used hand raitingarrow_forwardQuizzes - Gen Organic & Biological Che... ☆ myd21.lcc.edu + O G screenshot on mac - Google Search savings hulu youtube google disney+ HBO zlib Homework Hel...s | bartleby cell bio book Yuzu Reader: Chemistry G periodic table - Google Search b Home | bartleby 0:33:26 remaining CHEM 120 Chapter 5_Quiz 3 Page 1: 1 > 2 > 3 > 6 ¦ 5 > 4 > 7 ¦ 1 1 10 8 ¦ 9 a ¦ -- Quiz Information silicon-27 A doctor gives a patient 0.01 mC i of beta radiation. How many beta particles would the patient receive in I minute? (1 Ci = 3.7 x 10 10 d/s) Question 5 (1 point) Saved Listen 2.22 x 107 222 x 108 3.7 x 108 2.22 x 108 none of the above Question 6 (1 point) Listen The recommended dosage of 1-131 for a test is 4.2 μCi per kg of body mass. How many millicuries should be given to a 55 kg patient? (1 mCi = 1000 μСi)? 230 mCiarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY