
(a)
Interpretation:
The Ptotal at
Concept Introduction:
Integrated rate laws for zero, first and second order reactions are,
Zeroth order:
First order:
Second order:
(a)
Answer to Problem 108AE
Explanation of Solution
The order of the reaction should be determined first.
Initial pressure of C2H5OH =
If x is the pressure of C2H5OH reacts, then at any time, t
| t (s) | PTotal (torr) | x (torr) | Pethanol (torr) |
| 0 | 250 | 0 | 250 |
| 10 | 265 | 15 | 235 |
| 20 | 280 | 30 | 220 |
| 30 | 295 | 45 | 205 |
| 40 | 310 | 60 | 190 |
| 50 | 325 | 75 | 175 |
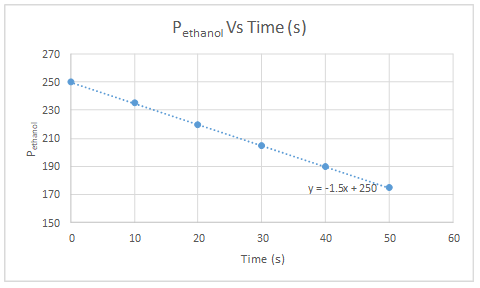
Pethanol versus time plot is linear. So, decomposition of ethanol follows zeroth order kinetics with respect to ethanol.
According to the equation on chart, Ptotal can be calculated as follows.
(b)
Interpretation:
The value of the rate constant and the units should be determined.
Concept Introduction:
Integrated rate laws for zero, first and second order reactions are,
Zeroth order:
First order:
Second order:
(b)
Answer to Problem 108AE
Explanation of Solution
Pethanol versus time plot is linear. So, decomposition of ethanol follows zeroth order kinetics with respect to ethanol.
Integrated rate law for the reaction;
So, the slope of the graph gives the value of the rate constant.
(c)
Interpretation:
The order of the reaction should be determined.
Concept Introduction:
Integrated rate laws for zero, first and second order reactions are,
Zeroth order:
First order:
Second order:
(c)
Answer to Problem 108AE
Zeroth order
Explanation of Solution
Pethanol versus time plot is linear. So, decomposition of ethanol follows zeroth order kinetics with respect to ethanol.
Integrated rate law for the reaction;
(d)
Interpretation:
Ptotal at
Concept Introduction:
Integrated rate laws for zero, first and second order reactions are,
Zeroth order:
First order:
Second order:
(d)
Answer to Problem 108AE
Explanation of Solution
According to the equation on chart, Ptotal can be calculated as follows.
But this is an impossible answer. Because the maximum pressure that can be after the completion of reaction is 500 torr.
When all the ethanol is decomposed
Therefore, at
Want to see more full solutions like this?
Chapter 15 Solutions
EBK CHEMICAL PRINCIPLES
- For a certain gas-phase reaction at constant pressure, the equilibrium constant Kp is observed to double when the temperature increases from 300 K to 400 K. Calculate the enthalpy change of the reaction, Ah, using this information.arrow_forwardHydrogen bonding in water plays a key role in its physical properties. Assume that the energy required to break a hydrogen bond is approximately 8 kJ/mol. Consider a simplified two-state model where a "formed" hydrogen bond is in the ground state and a "broken" bond is in the excited state. Using this model: • Calculate the fraction of broken hydrogen bonds at T = 300 K, and also at T = 273 K and T = 373 K. • At what temperature would approximately 50% of the hydrogen bonds be broken? • What does your result imply about the accuracy or limitations of the two-state model in describing hydrogen bonding in water? Finally, applying your understanding: • Would you expect it to be easier or harder to vaporize water at higher temperatures? Why? If you were to hang wet laundry outside, would it dry more quickly on a warm summer day or on a cold winter day, assuming humidity is constant?arrow_forward(3 pts) Use the Kapustinskii equation to calculate the lattice enthalpy for MgBr2 anddiscuss any differences between this result and that from #4.arrow_forward
- (3 pts) Silver metal adopts a fcc unit cell structure and has an atomic radius of 144 pm. Fromthis information, calculate the density of silver. Show all work.arrow_forward4. (3 pts) From the information below, determine the lattice enthalpy for MgBr2. Show all work. AH/(kJ mol-¹) Sublimation of Mg(s) +148 lonization of Mg(g) +2187 to Mg2+(g) Vaporization of Br₂(1) +31 Dissociation of Br,(g) +193 Electron gain by Br(g) -331 Formation of MgBr₂(s) -524arrow_forward1. (4 pts-2 pts each part) Consider the crystal structures of NaCl, ZnS, and CsCl (not necessarily shown in this order). a. For one of the three compounds, justify that the unit cell is consistent with stoichiometry of the compound. b. In each of the crystal structures, the cations reside in certain holes in the anions' packing structures. For each compound, what type of holes are occupied by the cations and explain why those particular types of holes are preferred.arrow_forward
- (2 pts) What do you expect to happen in a Na2O crystal if a Cl− ion replaces one of the O2−ions in the lattice?arrow_forward(2 pts) WSe2 is an ionic compound semiconductor that can be made to be p-type or n-type.What must happen to the chemical composition for it to be p-type? What must happen tothe chemical composition for it to be n-type?arrow_forward8. (2 pts) Silicon semiconductors have a bandgap of 1.11 eV. What is the longest photon wavelength that can promote an electron from the valence band to the conduction band in a silicon-based photovoltaic solar cell? Show all work. E = hv = hc/λ h = 6.626 x 10-34 Js c = 3.00 x 108 m/s 1 eV 1.602 x 10-19 Jarrow_forward
- A solution containing 100.0 mL of 0.155 M EDTA buffered to pH 10.00 was titrated with 100.0 mL of 0.0152 M Hg(ClO4)2 in a cell: calomel electrode (saturated)//titration solution/Hg(l) Given the formation constant of Hg(EDTA)2-, logKf= 21.5, and alphaY4-=0.30, find out the cell voltage E. Hg2+(aq) + 2e- = Hg(l) E0= 0.852 V E' (calomel electrode, saturated KCl) = 0.241 Varrow_forwardFrom the following reduction potentials I2 (s) + 2e- = 2I- (aq) E0= 0.535 V I2 (aq) + 2e- = 2I- (aq) E0= 0.620 V I3- (aq) + 2e- = 3I- (aq) E0= 0.535 V a) Calculate the equilibrium constant for I2 (aq) + I- (aq) = I3- (aq). b) Calculate the equilibrium constant for I2 (s) + I- (aq) = I3- (aq). c) Calculate the solubility of I2 (s) in water.arrow_forward2. (3 pts) Consider the unit cell for the spinel compound, CrFe204. How many total particles are in the unit cell? Also, show how the number of particles and their positions are consistent with the CrFe204 stoichiometry - this may or may not be reflected by the particle colors in the diagram. (HINT: In the diagram, the blue particle is in an interior position while each red particle is either in a corner or face position.)arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





