
Pearson eText Chemistry: The Central Science -- Access Card
14th Edition
ISBN: 9780136848981
Author: Brown, Theodore, Lemay, H., Bursten, Bruce, Murphy, Catherine, Woodward, Patrick, Stoltzfus, Matthew
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 4E
Suppose that for the reaction K+L → M, you monitor the production of M over time, and then plot the following graph from your data:
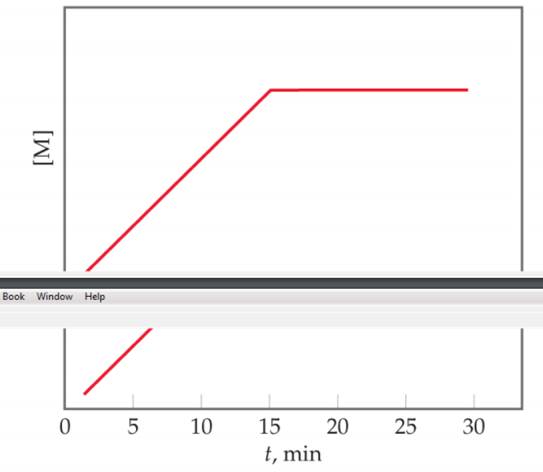
- Is the reaction occurring at a constant rate from t=0 to t=15 min?
- Is the reaction completed at t= 15 min? (c) Suppose the reaction as plotted here were started with 0.20 mol K and 0.40 mol L. After 30 min, an additional 0.20 mol K are added to the reaction mixture. Which of the following correctly describes how the plot would look from t= 30 min to t=60 min? (i) [M] would remain at the same constant value it has at t=30 min, (II) [M] would increase with the same slope as t=0 to 15 min, until t=45 min at which point the plot becomes horizontal again, or (iii) [M] decreases and reaches 0 at t= 45 min. [Section 14.2]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Apply the NANSTE law to the MnO4- + 8H+ + 5e- ⇄ Mn2+ + 4H2O
In the Nernst Law, how much is RT / F?
Hi can you please help me solve these problems? thank you
Chapter 14 Solutions
Pearson eText Chemistry: The Central Science -- Access Card
Ch. 14.2 - If the experiment in Figure 14.2 is run for 60 s,...Ch. 14.2 - Prob. 14.1.2PECh. 14.2 - Which of the following could be the instantaneous...Ch. 14.2 - Using Figure 14.3, determine the instantaneous...Ch. 14.2 - At a certain time in a reaction, substance A is...Ch. 14.2 - Prob. 14.3.2PECh. 14.3 - Suppose the rate law for the reaction in this...Ch. 14.3 - Assuming that rate = k[A][B], rank the mixtures...Ch. 14.3 - Prob. 14.5.1PECh. 14.3 - Prob. 14.5.2PE
Ch. 14.3 - Consider the reaction examined above in the Sample...Ch. 14.3 - The following data were measured for the reaction...Ch. 14.4 - At 25 ° C, the decomposition of dinitrogen...Ch. 14.4 - Practice Exercise 2 The decomposition of dimethyl...Ch. 14.4 - Practice Exercise 1 For a certain reaction A ...Ch. 14.4 - Prob. 14.8.2PECh. 14.4 - Practice Exercise 1 We noted in an earlier...Ch. 14.4 - Practice Exercise 2 Using Equation 14.17,...Ch. 14.5 - Practice Exercise 1 This of the following change...Ch. 14.5 - Practice Exercise 2 Rank the rate constants of the...Ch. 14.5 - Practice Exercise 1 Using the data in Sample...Ch. 14.5 - Practice Exercise 2 To one significant figure,...Ch. 14.6 - Practice Exercise 1 Consider the following...Ch. 14.6 - For the reaction Mo(CO)6 +P(CH3)3 Mo(CO)5P(CH3)3...Ch. 14.6 - Practice Exercise 1 Consider the following...Ch. 14.6 - Practice Exercise 2 Consider the following...Ch. 14.6 - Practice Exercise 1 An Alternative two-step...Ch. 14.6 - Prob. 14.14.2PECh. 14.6 - Practice Exercise 1
Consider the...Ch. 14.6 - Prob. 14.15.2PECh. 14 - Prob. 1DECh. 14 - An automotive fuel injector dispenses a fine spray...Ch. 14 - Consider the following graph of the concentration...Ch. 14 - You study the rate of a reaction, measuring both...Ch. 14 - Suppose that for the reaction K+L M, you monitor...Ch. 14 - Prob. 5ECh. 14 - A friend studies a first-order reaction and...Ch. 14 - Prob. 7ECh. 14 - Which of the following linear plots do you expect...Ch. 14 - Prob. 9ECh. 14 - Prob. 10ECh. 14 - The following graph shows two different reaction...Ch. 14 - Prob. 12ECh. 14 - Prob. 13ECh. 14 - Draw a possible transition state for the...Ch. 14 - The following diagram represents an imaginary...Ch. 14 - 14.16 Draw a graph showing the reaction pathway...Ch. 14 - Prob. 17ECh. 14 - 14.18 (a) what are the units usually used to...Ch. 14 - Prob. 19ECh. 14 - A flask is charged with 0.100 mol of A and allowed...Ch. 14 - The isomerization of methyl isontrile (CH3NC) to...Ch. 14 - Prob. 22ECh. 14 - Prob. 23ECh. 14 - For each of the following gas-phase reactions,...Ch. 14 - (a) Consider the combustion of hydrogen, 2H2 (g) +...Ch. 14 - Prob. 26ECh. 14 - A reaction A+B C obeys the following rate law:...Ch. 14 - Prob. 28ECh. 14 - 14.29 The decomposition reaction of N2O5 in carbon...Ch. 14 - Prob. 30ECh. 14 - Prob. 31ECh. 14 - The reaction between ethyl bromide (C2H5Br) and...Ch. 14 - Prob. 33ECh. 14 - The reaction 2ClO2 (aq) + 2OH- (aq) ClO3- (aq) +...Ch. 14 - The following data were measured for the reaction...Ch. 14 - The following data were collected for the rate of...Ch. 14 - Consider the gas-phase reaction between nitric...Ch. 14 - Prob. 38ECh. 14 - Prob. 39ECh. 14 - Prob. 40ECh. 14 - Prob. 41ECh. 14 - Molecular iodine, I2 (g), dissociates into iodine...Ch. 14 - Prob. 43ECh. 14 - Prob. 44ECh. 14 - The reaction SO2Cl2 (g) O2 (g) + Cl2 (g) is first...Ch. 14 - Prob. 46ECh. 14 - Prob. 47ECh. 14 - Prob. 48ECh. 14 - Prob. 49ECh. 14 - Prob. 50ECh. 14 - (a) what factors determine whether a collision...Ch. 14 - (a) in which of the following reactions you expect...Ch. 14 - Calculate the fraction of atoms in a sample of...Ch. 14 - (a) the activation energy for the isomerization of...Ch. 14 - The gas-phase reaction CL (g) + HBr (g) + HCl (g)...Ch. 14 - Prob. 56ECh. 14 - Indicate whether each statement is true or false....Ch. 14 - Indicate whether each statement is true or false....Ch. 14 - Based on their activation energies and energy...Ch. 14 - Prob. 60ECh. 14 - Prob. 61ECh. 14 - Prob. 62ECh. 14 - The rate of the reaction CH3COOC2H5 (aq) + OH- ...Ch. 14 - Prob. 64ECh. 14 - Prob. 65ECh. 14 - Prob. 66ECh. 14 - What is the molecularity of each of the following...Ch. 14 - Prob. 68ECh. 14 - (a) based on the following reaction profile, how...Ch. 14 - Prob. 70ECh. 14 - Prob. 71ECh. 14 - Prob. 72ECh. 14 - The reaction 2NO (g) + CL2 (g) 2NOCl (g) was...Ch. 14 - You have studied the gas-phase oxidation of HBr by...Ch. 14 - Prob. 75ECh. 14 - Prob. 76ECh. 14 - Prob. 77ECh. 14 - Prob. 78ECh. 14 - Prob. 79ECh. 14 - The addition of No accelerates the decomposition...Ch. 14 - 14.81b Many metallic catalysts, particularly the...Ch. 14 - Prob. 82ECh. 14 - When D2 reacts with ethylene (C2H4) in the...Ch. 14 - Prob. 84ECh. 14 - Prob. 85ECh. 14 - The enzyme urease catalyzez the reaction of urea,(...Ch. 14 - Prob. 87ECh. 14 - Prob. 88ECh. 14 - Prob. 89AECh. 14 - Prob. 90AECh. 14 - Prob. 91AECh. 14 - Prob. 92AECh. 14 - Prob. 93AECh. 14 - Prob. 94AECh. 14 - Prob. 95AECh. 14 - Prob. 96AECh. 14 - [14.97]A first order reaction A B has the rate...Ch. 14 - Prob. 98AECh. 14 - Prob. 99AECh. 14 - Prob. 100AECh. 14 - Prob. 101AECh. 14 - Prob. 102AECh. 14 - Cyclopentadiene (C5H6) reacts with itself to form...Ch. 14 - Prob. 104AECh. 14 - At 280C, raw milk sours in 4.0 h but takes 48 h to...Ch. 14 - Prob. 106AECh. 14 - Prob. 107AECh. 14 - Prob. 108AECh. 14 - Prob. 109AECh. 14 - The following mechanism has been proposed for the...Ch. 14 - Prob. 111AECh. 14 - Prob. 112AECh. 14 - Platinum nanoparticles of diameter ~2 nm are...Ch. 14 - 14.114 One of the many remarkable enzymes in the...Ch. 14 - 14.115N Suppose that, in the absence of catalyst,...Ch. 14 - Prob. 116AECh. 14 - Dinitrogen pentoxide (N2O5) decomposes in...Ch. 14 - The reaction between ethyl iodide and hydroxide...Ch. 14 - Prob. 119IECh. 14 - Prob. 120IECh. 14 - Prob. 121IECh. 14 - The rates of many atmospheric reactions are...Ch. 14 - Prob. 123IECh. 14 - Prob. 124IECh. 14 - Prob. 125IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Hi can you please help me solve this problem? thank youarrow_forwardAn electrode process takes place at a metal-solution interface. Indicate the current condition that must be met for Faradaic rectification to occur.arrow_forwardAt a metal-solution interface, an electron is exchanged, and the symmetry factor beta < 0.5 is found in the Butler-Volmer equation. What does this indicate?arrow_forward
- Please do these questions within the SCH4U course please with full steps since I am still unsure how to format my answers! Thank you so much.arrow_forwardWhen two solutions, one of 0.1 M KCl (I) and the other of 0.1 M MCl (II), are brought into contact by a membrane. The cation M cannot cross the membrane. At equilibrium, x moles of K+ will have passed from solution (I) to (II). To maintain the neutrality of the two solutions, x moles of Cl- will also have to pass from I to II. Explain this equality: (0.1 - x)/x = (0.1 + x)/(0.1 - x)arrow_forwardCalculate the variation in the potential of the Pt/MnO4-, Mn2+ pair with pH, indicating the value of the standard potential. Data: E0 = 1.12.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning  Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY