
Concept explainers
a) Interpretation:
The total estimated mass Zr and Te disposed as nuclear waste by the nuclear technician
Concept Introduction:
(uranium) undergoes a fission reaction which is neutron induced. On doing so, it will split into two smaller nuclei
(zirconium) and (tellurium). In the process, neutrons and gamma rays are released. The
Answer to Problem 14.59PAE
Solution:
3.231 metric ton
Explanation of Solution
The total amount of electrical energy generated by all the nuclear reactors is
From the problem 14.55, we know the amount of energy released during the fission of 1 kg of is
Hence,
Total mass of used =
For every fission of 236.133 grams of , 233.83627 grams of nuclear waste is generated.
Hence, the total estimated mass of Zr and Te (which are the products of fission of ) disposed as nuclear waste is
(b) Interpretation:
Whether any of the products Zr and Te obtained from the fission of
are available in measurable quantities in the nuclear waste
Concept Introduction: (uranium) undergoes a fission reaction which is neutron induced. On doing so, it will split into two smaller nuclei (zirconium) and (tellurium). In the process, neutrons and gamma rays are released. The nuclear equation for this reaction is as follows:
Answer to Problem 14.59PAE
Solution:
Only a measurable quantity of Zr will be left in the waste
Explanation of Solution
The half life of is 16.749 hours. The half life of is 2.49 seconds. As would undergo Beta-minus decay, it would get degraded within 2.49 seconds whereas Zr having larger half life will be present in measurable quantities.
(c) Interpretation:
Decay series of each isotope and
Concept Introduction: (uranium) undergoes a fission reaction which is neutron induced. On doing so, it will split into two smaller nuclei (zirconium) and (tellurium). In the process, neutrons and gamma rays are released. The nuclear equation for this reaction is as follows:
Answer to Problem 14.59PAE
Solution:
The decay series for Te
Explanation of Solution
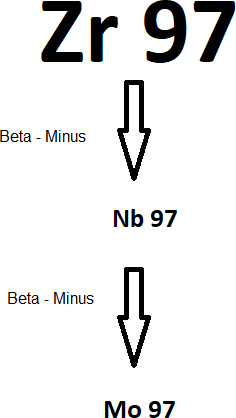
The decay series of is
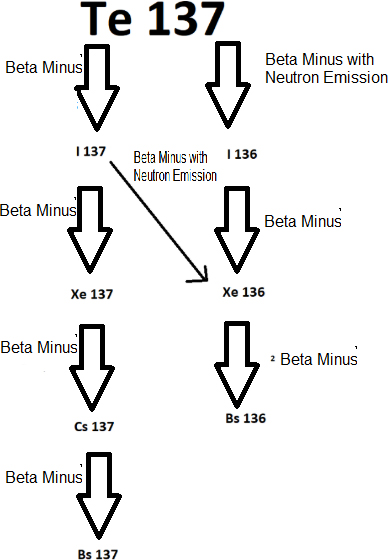
The decay series of is
The decay series of has Cs 137 which will remain in waste for many years.
(d) Interpretation:
The difference between the total mass of the waste using the stable isotope products and the technician’s estimated total mass.
Concept Introduction: (uranium) undergoes a fission reaction which is neutron induced. On doing so, it will split into two smaller nuclei (zirconium) and (tellurium). In the process, neutrons and gamma rays are released. The nuclear equation for this reaction is as follows:
Answer to Problem 14.59PAE
Solution:
The technician’s estimates of total waste were off by 0.079751 kg.
Explanation of Solution
Mass of the stable isotope products =
Mass of the waste so obtained = kg or 3.2309273 metric ton
The difference between the technician’s estimated mass and the above calculated mass is
Want to see more full solutions like this?
Chapter 14 Solutions
Bundle: Chemistry for Engineering Students, 3rd, Loose-Leaf + OWLv2 with QuickPrep 24-Months Printed Access Card
- 111 Carbonyl Chem Choosing reagants for a Wittig reaction What would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 3 1 2 2. n-BuLi • Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure. × ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Usearrow_forwardA student proposes the transformation below in one step of an organic synthesis. There may be one or more reactants missing from the left-hand side, but there are no products missing from the right-hand side. There may also be catalysts, small inorganic reagents, and other important reaction conditions missing from the arrow. • Is the student's transformation possible? If not, check the box under the drawing area. . If the student's transformation is possible, then complete the reaction by adding any missing reactants to the left-hand side, and adding required catalysts, inorganic reagents, or other important reaction conditions above and below the arrow. • You do not need to balance the reaction, but be sure every important organic reactant or product is shown. + T X O O лет-ле HO OH HO OH This transformation can't be done in one step.arrow_forwardDetermine the structures of the missing organic molecules in the following reaction: X+H₂O H* H+ Y OH OH Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. X Sarrow_forward
- Predict the major products of this organic reaction. If there aren't any products, because nothing will happen, check the box under the drawing area instead. No reaction. HO. O :☐ + G Na O.H Click and drag to start drawing a structure. XS xs H₂Oarrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? H H C H- a -H b H Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal groups may have slightly different sizes. a = b = 0 °arrow_forwardWhat are the angles a and b in the actual molecule of which this is a Lewis structure? :0: HCOH a Note for advanced students: give the ideal angles, and don't worry about small differences from the ideal that might be caused by the fact that different electron groups may have slightly different sizes. a = 0 b=0° Sarrow_forward
- Determine the structures of the missing organic molecules in the following reaction: + H₂O +H OH O OH +H OH X Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structure of the missing organic molecule X. Click and drag to start drawing a structure.arrow_forwardIdentify the missing organic reactant in the following reaction: x + x O OH H* + ☑- X H+ O O Х Note: This chemical equation only focuses on the important organic molecules in the reaction. Additional inorganic or small-molecule reactants or products (like H₂O) are not shown. In the drawing area below, draw the skeletal ("line") structure of the missing organic reactant X. Click and drag to start drawing a structure. Carrow_forwardCH3O OH OH O hemiacetal O acetal O neither O 0 O hemiacetal acetal neither OH hemiacetal O acetal O neither CH2 O-CH2-CH3 CH3-C-OH O hemiacetal O acetal CH3-CH2-CH2-0-c-O-CH2-CH2-CH3 O neither HO-CH2 ? 000 Ar Barrow_forward
- What would be the best choices for the missing reagents 1 and 3 in this synthesis? 1. PPh3 2 2. n-BuLi 3 Draw the missing reagents in the drawing area below. You can draw them in any arrangement you like. • Do not draw the missing reagent 2. If you draw 1 correctly, we'll know what it is. • Note: if one of your reagents needs to contain a halogen, use bromine. Explanation Check Click and drag to start drawing a structure.arrow_forwardPredict the products of this organic reaction: NaBH3CN + NH2 ? H+ Click and drag to start drawing a structure. ×arrow_forwardPredict the organic products that form in the reaction below: + OH +H H+ ➤ ☑ X - Y Note: You may assume you have an excess of either reactant if the reaction requires more than one of those molecules to form the products. In the drawing area below, draw the skeletal ("line") structures of the missing organic products X and Y. You may draw the structures in any arrangement that you like, so long as they aren't touching. Click and drag to start drawing a structure. Garrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





