
Concept explainers
(a)
Interpretation:
The substitutive name and common name of
Concept introduction:
The systematic naming of organic compound is given by
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
Answer to Problem 14.3P
The common name of the compound
Explanation of Solution
The compound
The compound
The number on the carbon atoms in the compound is shown below.
The compound is
The common name of the compound
(b)
Interpretation:
The substitutive name and common name of
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
Answer to Problem 14.3P
The common name of the compound
Explanation of Solution
The compound
The compound
The number on the carbon atoms in the compound is shown below.
The compound is alkyne. Therefore, -yne will be present as the suffix. The triple bond is present on the fifth carbon atom. The compound has ten-carbon atom long chain. Therefore, the substitutive name of the compound is
The common name of the compound
(c)
Interpretation:
The substitutive name of compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
Answer to Problem 14.3P
The substitutive name of the given compound is
Explanation of Solution
The structure of the given compound is shown below.
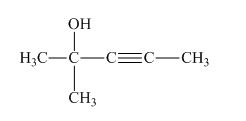
Figure 1
The compound has five carbon atom in the longest chain. Therefore, pent- will be used in the root name. The number of the carbon atoms will be started from the carbon atom which gives the lowest number to hydroxyl group in the chain.
The number on the carbon atoms in the compound is shown below.
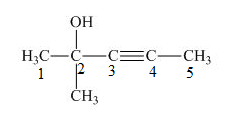
Figure 2
The compound has methyl group and hydroxyl group. Therefore, -yne will be present as primary suffix and –ol will be present on as secondary suffix. The methyl group is resent at the second carbon. Therefore, methyl will be used as prefix. The triple bond is present on the third carbon atom and hydroxyl group is present on the second carbon atom. Therefore, the substitutive name of the compound is
The substitutive name of the given compound is
(d)
Interpretation:
The substitutive name of compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
Answer to Problem 14.3P
The substitutive name of the compound is
Explanation of Solution
The structure of the given compound is shown below.
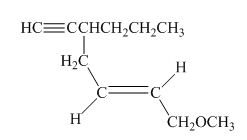
Figure 3
The compound has seven carbon atom in the longest chain. Therefore, hept- will be used in root name. The number of the carbon atoms will be started from the carbon atom which gives lowest number to propyl group in the chain.
The number on the carbon atoms in the compound is shown below.
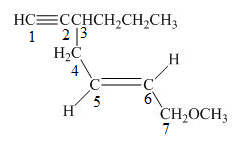
Figure 4
The compound has propyl group and methoxy group. Therefore, methoxy and propyl will be present as suffix. The propyl group is present at the third carbon. The triple bond is present on the first carbon atom and double bond is present at the fifth carbon atom. Therefore, the substitutive name of the compound is
The substitutive name of the given compound is
(e)
Interpretation:
The substitutive name of compound is to be stated.
Concept introduction:
The systematic naming of organic compound is given by IUPAC nomenclature. The naming of organic compound is done such that the structure of the organic compound is correctly interpreted from the name.
Rules for writing IUPAC name from the structural formula are given below.
- First, identify the longest carbon chain.
- The next step is to identify the groups attached to the longest chain.
- Identify the position, location, and a number of the substituents bonded to the carbon chain.
- Use prefix di, tri, tetra if the same type of substituents is present.
- Name the substituents in alphabetical order.
Answer to Problem 14.3P
The substitutive name of the given compound is
Explanation of Solution
The structure of the given compound is shown below.
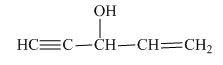
Figure 5
The compound has five carbon atom in the longest chain. Therefore, pent- will be used in root name. The number of the carbon atoms will be started from the carbon atom which gives lowest number to double bond in the chain.
The number on the carbon atoms in the compound is shown below.
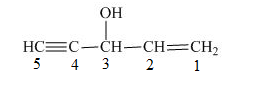
Figure 6
The compound has hydroxyl group. Therefore, hydroxyl will be present as prefix. The hydroxyl group is present at the third carbon. The triple bond is present on the fifth carbon atom and double bond is present at the first carbon atom. Therefore, the substitutive name of the compound is
The substitutive name of the given compound is
Want to see more full solutions like this?
Chapter 14 Solutions
EBK ORGANIC CHEMISTRY
- Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer formed in this reaction. Assume there are hydrogen atoms there are hydrogen atoms on the two ends of the trimer. Ignore inorganic byproducts please.arrow_forwardi need help with the folarrow_forwardPLEASE HELP NOW! URGENT!arrow_forward
- a. Determine whether each of the Followery Molecules is in the R- On the y- Configuration 1-01"/ 1-6-4 Br 4 I el Br b. Draw The Fisher projection For all the Meso compounds that can exist FOR The Following molenlearrow_forward1- Refer to the monosaccharides below to answer each of the following question(s): CH₂OH CHO CH₂OH CH₂OH 0 H- OH 0 0 HO- H H- -OH HO H HO H H OH HO- H CH₂OH H. OH HO H HO- H CH₂OH CH₂OH CH3 a. Sorbose b. Rhamnose c. Erythrulose d. Xylulose Classify each sugar by type; for example, glucose is an aldohexose. a. Xylulose is .. b. Erythrulose is . c. Sorbose is .. d. Rhamnose is .. 2- Consider the reaction below to answer the following question(s). CHO H OH CH₂OH CH₂OH HO- H HO HO + H. -OH HO OH HO. H OH OH H -OH H OH CH₂OH Q Z a. Refer to Exhibit 25-11. Place a triangle around the anomeric carbon in compound Q. Compound Z is: b. 1. the D-anomer. 2. the a-anomer. 3. the ẞ-anomer. 4. the L-anomer. c. Which anomer is the LEAST stable? d. Q and Z are cyclic examples of: a. acetals b. hemiacetals c. alditols d. hemialditolsarrow_forwardi need help identifying the four carbon oxygen bonds in the following:arrow_forward
- Imagine each of the molecules shown below was found in an aqueous solution. Can you tell whether the solution is acidic, basic, or neutral? molecule HO H3N + The solution is... X O acidic OH O basic H3N-CH-C-O O neutral ○ (unknown) O acidic ○ basic CH2 CH 3-S-CH2 O neutral ○ (unknown) H3N O OH O acidic O basic Oneutral O (unknown) 0 H3N-CH-C-O CH3 CH CH3 O acidic O basic O neutral ○ (unknown) ? olo Ar BHarrow_forwardno Ai walkthroughs need other product (product in picture is wrong dont submit the same thing)arrow_forwardHow to solve this!arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





