
EBK BASIC CHEMISTRY
5th Edition
ISBN: 8220101472335
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 1.4, Problem 1.23QAP
Interpretation Introduction
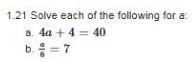
To determine: The value of a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the missing intermediate 1 and the final product 2. Please include a detailed explanation explaining the steps of malonic ester synthesis. Please include drawings of the intermediate and how it occurs and how the final product is former.
What would be the reagents and conditions above and below the arrow that will complete the proposed acetoacetic ester synthesis? If it cannot be done efficiently, then I will choose that answer. There could be 2 or 4 reagents involved. Please provide a detailed explanation and drawings showing how it would proceed with the correct reagents.
For benzene, the ∆H° of vaporization is 30.72 kJ/mol and the ∆S° of vaporization is 86.97 J/mol・K. At 1.00 atm and 228.0 K, what is the ∆G° of vaporization for benzene, in kJ/mol?
Chapter 1 Solutions
EBK BASIC CHEMISTRY
Ch. 1.1 - Write a one-sentence definition for each of the...Ch. 1.1 - Write a one-sentence definition for each of the...Ch. 1.1 - Obtain a bottle of multivitamins and read the list...Ch. 1.1 - Prob. 1.4QAPCh. 1.1 - Read the labels on some items found in your...Ch. 1.1 - Read the labels on products used to wash your...Ch. 1.2 - Define each of the following terms of the...Ch. 1.2 - Identify each of the following activities in the...Ch. 1.2 - Prob. 1.9QAPCh. 1.2 - Identify each activity, a to f, as an observation...
Ch. 1.2 - Prob. 1.11QAPCh. 1.2 - Identify each of the following as an observation...Ch. 1.3 - Prob. 1.13QAPCh. 1.3 - Prob. 1.14QAPCh. 1.3 - Prob. 1.15QAPCh. 1.3 - Prob. 1.16QAPCh. 1.4 - What is the place value for the bold digit? 7.3288...Ch. 1.4 - Prob. 1.18QAPCh. 1.4 - Prob. 1.19QAPCh. 1.4 - Prob. 1.20QAPCh. 1.4 - Prob. 1.21QAPCh. 1.4 - Prob. 1.22QAPCh. 1.4 - Prob. 1.23QAPCh. 1.4 - Prob. 1.24QAPCh. 1.4 - Prob. 1.25QAPCh. 1.4 - What is measured on the horizontal axis? What is...Ch. 1.5 - Prob. 1.27QAPCh. 1.5 - Prob. 1.28QAPCh. 1.5 - Write each of the following as a standard number:...Ch. 1.5 - Prob. 1.30QAPCh. 1.5 - Prob. 1.31QAPCh. 1.5 - Prob. 1.32QAPCh. 1 - Prob. 1.33FUCh. 1 - Prob. 1.34FUCh. 1 - Prob. 1.35UTCCh. 1 - Prob. 1.36UTCCh. 1 - Prob. 1.37UTCCh. 1 - Prob. 1.38UTCCh. 1 - Prob. 1.39UTCCh. 1 - Prob. 1.40UTCCh. 1 - Prob. 1.41UTCCh. 1 - Prob. 1.42UTCCh. 1 - Prob. 1.43AQAPCh. 1 - Prob. 1.44AQAPCh. 1 - Prob. 1.45AQAPCh. 1 - Prob. 1.46AQAPCh. 1 - Prob. 1.47AQAPCh. 1 - Prob. 1.48AQAPCh. 1 - Evaluate each of the following: (1.4) 4x(-8)=...Ch. 1 - Prob. 1.50AQAPCh. 1 - Prob. 1.51AQAPCh. 1 - Prob. 1.52AQAPCh. 1 - Prob. 1.53AQAPCh. 1 - Prob. 1.54AQAPCh. 1 - Prob. 1.55AQAPCh. 1 - Prob. 1.56AQAPCh. 1 - Prob. 1.57CQCh. 1 - Prob. 1.58CQCh. 1 - Solve each of the following for X: (1.4) 2x + 5 =...Ch. 1 - Solve each of the following for z: (1.4) 3z ( 6)...Ch. 1 - What does the title indicate about the graph?...Ch. 1 - What is measured on the horizontal axis? (1.4)...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reaction. it is spontaneous only at High T, it is spontaneous at low T it is nonspontaneous at all T it is spontanrous at all T. it is non spontaneous only at low T.arrow_forwardThe reaction Q(g) + R(g) → Z(l) is shown to be exothermic. Which of the following is true concerning the reactionarrow_forwardWhich of the following has the largest standard molar entropy, S° (298.15 K) He H2 NaCl KBr Hgarrow_forward
- Which of the following is true for a particular reaction if ∆G° is -40.0 kJ/mol at 290 K and –20.0 kJ/mol at 390 K?arrow_forwardWhat is the major product of the following reaction? O O OH OH 1. BH 2. H₂O₂, NaOH OH OHarrow_forwardDraw the products formed when each ester is hydrolyzed with water and sulfuric acid.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY