
Chemistry (OER)
2nd Edition
ISBN: 9781947172616
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 104E
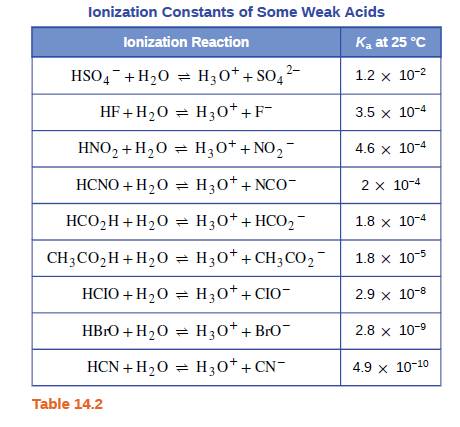
Which acid in Table 14.2 is most appropriate for preparation of a buffer solution with a pH of 3.7? Explain your choice.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Please help me answer the following questions. My answers weren't good enough. Need to know whyy the following chemicals were not used in this experiment related to the melting points and kf values. For lab notebook not a graded assignments.
Draw the arrow pushing reaction mechanism. DO NOT ANSWER IF YOU WONT DRAW IT. Do not use chat gpt.
Complete the following esterification reaction by drawing the structural
formula of the product formed.
HOH
HO
i
catalyst
catalyst
OH
HO
(product has rum flavor)
(product has orange flavor)
Chapter 14 Solutions
Chemistry (OER)
Ch. 14 - Write equations that show NH3 as both a conjugate...Ch. 14 - Write equations that show H2PO4- acting both as an...Ch. 14 - Show by suitable net ionic equations that each of...Ch. 14 - Show by suitable net ionic equations that each of...Ch. 14 - Show by suitable net ionic equations that each of...Ch. 14 - Show by suitable net ionic equations that each of...Ch. 14 - What is the conjugate acid of each of the...Ch. 14 - What is the conjugate acid of each of the...Ch. 14 - Identify and label the Bronsted-Lowry acid, its...Ch. 14 - Identify and label the Bronsted-Lowry acid, its...
Ch. 14 - What are amphiprotic species? Illustrate with...Ch. 14 - State which of the following species are...Ch. 14 - State which of the following species are...Ch. 14 - Is the self-ionization of water endothermic or...Ch. 14 - Explain why a sample of pure water at 40 C is...Ch. 14 - The ionization constant for water (Kw) is 2.91014...Ch. 14 - The ionization constant for water (Kw) is...Ch. 14 - Calculate the pH and the pOH of each of the...Ch. 14 - Calculate the pH and the pOH of each of the...Ch. 14 - What are the pH and pOH of a solution of 2.0 M...Ch. 14 - What are the hydronium and hydroxide ion...Ch. 14 - Calculate the hydrogen ion concentration and the...Ch. 14 - Calculate the hydronium ion concentration and the...Ch. 14 - The hydronium ion concentration in a sample of...Ch. 14 - The hydroxide ion concentration in household...Ch. 14 - Explain why the neutralization reaction of a...Ch. 14 - Explain why the neutralization reaction of a weak...Ch. 14 - Use this list of important industrial compounds...Ch. 14 - The odor of vinegar is due to the presence of...Ch. 14 - Household ammonia is a solution of the weak base...Ch. 14 - Explain why the ionization constant, Ka, for H2SO4...Ch. 14 - Explain why the ionization constant, Ka, for HI is...Ch. 14 - Gastric juice, the digestive ?uid produced in the...Ch. 14 - Nitric acid reacts with insoluble copper (II)...Ch. 14 - What is the ionization constant at 25 C for the...Ch. 14 - What is the ionization constant at 25 C for the...Ch. 14 - Which base, CH3NH2 or (CH3)2NH, is the stronger...Ch. 14 - Which is the stronger acid, NH4+ or HBrO?Ch. 14 - Which is the stronger base, (CH3)3N or H2BO3-?Ch. 14 - Predict which acid in each of the following pairs...Ch. 14 - Predict which compound in each of the following...Ch. 14 - Rank the compounds in each of the following groups...Ch. 14 - Rank the compounds in each of the following groups...Ch. 14 - Both HF and HCN ionize in water to a limited...Ch. 14 - The active ingredient formed by aspirin in the...Ch. 14 - What do we represent when we write:...Ch. 14 - Explain why equilibrium calculations are not...Ch. 14 - Are the concentrations of hydronium ion and...Ch. 14 - What two common assumptions can simplify...Ch. 14 - What two common assumptions can simplify...Ch. 14 - Which of the following will increase the percent...Ch. 14 - Which of the following will increase the percent...Ch. 14 - What is the effect on the concentrations of NO2-,...Ch. 14 - What is the effect on the concentration of...Ch. 14 - Why is the hydronium ion concentration in a...Ch. 14 - From the equilibrium concentrations given,...Ch. 14 - From the equilibrium concentrations given,...Ch. 14 - Determine Kb for the nitrite ion, NO2-. In a...Ch. 14 - Determine Ka for hydrogen sulfate ion, HSO4-. In a...Ch. 14 - Calculate the ionization constant for each of the...Ch. 14 - Calculate the ionization constant for each of the...Ch. 14 - For which of the following solutions must we...Ch. 14 - Even though both NH3 and C6H5NH2 are weak bases,...Ch. 14 - Calculate the equilibrium concentration of the...Ch. 14 - Calculate the equilibrium concentration of the...Ch. 14 - Calculate the equilibrium concentration of the...Ch. 14 - Calculate the equilibrium concentration of the...Ch. 14 - Using the Ka value of , place Al(H2O)63+ in the...Ch. 14 - Calculate the concentration of all solute species...Ch. 14 - Propionic acid, C2H5CO2H (Ka=1.34105), is used in...Ch. 14 - White vinegar is a 5.0% by mass solution of acetic...Ch. 14 - The ionization constant of lactic acid,...Ch. 14 - Nicotine, C10H14N2, is a base that will accept two...Ch. 14 - The pH of a 0.20-M solution of HP is 1.92....Ch. 14 - The pH of a 0.15-M solution of HSO4- is 1.43....Ch. 14 - The pH of a 0.10-M solution of caffeine is 11.16....Ch. 14 - Tile pH of a solution of household ammonia, a...Ch. 14 - Determine whether aqueous solutions of the...Ch. 14 - Determine whether aqueous solutions of the...Ch. 14 - Novocaine, C13H21O2N2Cl, is the salt of the base...Ch. 14 - Which of the following concentrations would be...Ch. 14 - Calculate the concentration of each species...Ch. 14 - Calculate the concentration of each species...Ch. 14 - Salicylic acid, HOC6H4CO2H, and its derivatives...Ch. 14 - The ion HTe- is an amphiprotic species; it can act...Ch. 14 - Explain why a buffer can be prepared from a...Ch. 14 - Explain why the pH does not change significantly...Ch. 14 - Explain why the pH does not change significantly...Ch. 14 - What is [H3O+] in a solution of 0.25 M CH3CO2H and...Ch. 14 - What is [H3O+] in a solution of 0.075 M HNO2 and...Ch. 14 - What is [OH-] in a solution of 0.125 M CH3NH2 and...Ch. 14 - What is [OH-] in a solution of 1.25 M NH3 and 0.78...Ch. 14 - What concentration of NH4NO3 is required to make...Ch. 14 - What concentration of NaF is required to make...Ch. 14 - What is the effect on the concentration of acetic...Ch. 14 - What is the effect on the concentration of...Ch. 14 - What will be the pH of a buffer solution prepared...Ch. 14 - Calculate the pH of a buffer solution prepared...Ch. 14 - How much solid NaCH3CO23H2O must be added to 0300...Ch. 14 - What mass of NH4Cl must be added to 0.750 L of a...Ch. 14 - A buffer solution is prepared from equal volumes...Ch. 14 - A 5.36-g sample of NH4Cl was added to 25.0 mL of...Ch. 14 - Which acid in Table 14.2 is most appropriate for...Ch. 14 - Which acid in Table 14.2 is most appropriate for...Ch. 14 - Which base in Table 14.3 is must appropriate for...Ch. 14 - Which base in Table 14.3 is most appropriate for...Ch. 14 - Saccharin, C7H4NSO3H, is a weak acid (Ka=2.1102)....Ch. 14 - What is the pH of 1.000 L of a solution of 100.0 g...Ch. 14 - Explain how to choose the appropriate acid-base...Ch. 14 - Explain why an acid-base indicator changes color...Ch. 14 - Why can we ignore the contribution of water to the...Ch. 14 - Why can we ignore the contribution of water to the...Ch. 14 - Draw a curve for a series of solutions of HF. Plot...Ch. 14 - Draw a curve similar to that shown in Figure 14.23...Ch. 14 - Calculate the pH at the following points in a...Ch. 14 - The indicator dinitrophenol is an acid with a Ka...
Additional Science Textbook Solutions
Find more solutions based on key concepts
3. CAUTION Why is genetic drift aptly named?
a. It causes allele frequencies to drift up or down randomly.
b. I...
Biological Science (6th Edition)
A Slice of pizza has 500 kcal. If we could burn the pizza and use all the heat to warm a 50-L container of cold...
Campbell Biology in Focus (2nd Edition)
Use a globe or map to determine, as accurately as possible, the latitude and longitude of Athens, Greece.
Applications and Investigations in Earth Science (9th Edition)
2. Whether an allele is dominant or recessive depends on
a. how common the allele is, relative to other alleles...
Campbell Biology: Concepts & Connections (9th Edition)
[14.110] The following mechanism has been proposed for the gas-phase reaction of chloroform (CHCI3) and chlorin...
Chemistry: The Central Science (14th Edition)
In a certain plant, fruit is either red or yellow, and fruit shape is either oval or long. Red and oval are the...
Concepts of Genetics (12th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG stands for the standard Gibbs free energy of reaction and K stands for the equilibrium constant. In each table, there may be one statement that is faise because it contradicts the other three statements. If you find a false statement, check the box next to t Otherwise, check the "no false statements" box under the table. statement false? AG"1 no false statements: statement false? AG-0 0 InK-0 0 K-1 0 AH-TAS no false statements 2arrow_forwardComplete the following esterification reactions by drawing the line formulas of the carboxylic acid and alcohol required to form the ester shown. catalyst catalyst catalyst apricot fragrancearrow_forwardShow the saponification products of the following ester: You don't need to draw in the Na+ cation. catalyst, A catalyst, A catalyst, Aarrow_forward
- What would happen if the carboxylic acid and alcohol groups were on the same molecule? In essence, the molecule reacts with itself. Draw the structure of the products formed in this manner using the reactants below. If two functional groups interact with one another on the same molecule, this is called an “intramolecular" (within one) rather than "intermolecular" (between two or more) attack. OH OH catalyst OH HO catalyst catalyst HO OHarrow_forwardQ3: Write in the starting alkyl bromide used to form the following products. Include any reactants, reagents, and solvents over the reaction arrow. If more than one step is required, denote separate steps by using 1), 2), 3), etc. H OH racemic OH OH 5 racemicarrow_forwardDraw the Lewis structure of the SO3-O(CH3)2 complex shown in the bottom right of slide 2in lecture 3-3 (“Me” means a CH3 group) – include all valence electron pairs and formal charges.From this structure, should the complex be a stable molecule? Explain.arrow_forward
- please add appropriate arrows, and tell me clearly where to add arrows, or draw itarrow_forwardWhat I Have Learned Directions: Given the following reaction and the stress applied in each reaction, answer the question below. A. H2(g) + Cl2(g) 2 HCl(g) Stress applied: Decreasing the pressure 1. What is the Keq expression? 2. What will be the effect in the number of moles of HCl(g)? 3. What will be the Equilibrium Shift or the reaction? B. Fe3O4(s) + 4 H2(g) + heat 53 Fe(s) + 4 H₂O(g) Stress applied: Increasing the temperature 1. What is the Keq expression?. 2. What will be the effect in the volume of water vapor collected? 3. What will be the Equilibrium Shift or the reaction? C. 4 NH3(g) + 5 O2(g) 4 NO(g) + 6 H2O(g) + heat Stress applied: Increasing the volume of the container 1. What is the Keq expression?. 2. What will be the effect in the amount of H₂O? 3. What will be the Equilibrium Shift or the reaction?arrow_forwardConsider the solubility products (Ksp values) for the following compounds:SrSO4 (Ksp = 7.6 x 10−7), BaSO4 (Ksp = 1.5 x 10−9), SrCO3 (Ksp = 7.0 x 10−10), BaCO3 (Ksp = 1.6 x 10−9)Which anion is the harder base, CO32− or SO42−? Justify your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY