
(a)
Interpretation: The reagents are to be identified for Synthesis of Racemic mixture of
Concept Introduction:
Epoxides are obtained by the conversion of
Epoxides from Peroxy acids:
The commonly used Peroxy acids are meta-Chloroperoxybenzoic acid (MCPBA) and Peroxyacetic acid. This process is stereospecific.
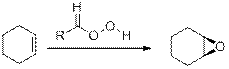
Specifically, the substituent are cis to each other in starting Alkene remains cis to each other in Epoxide, and the substituent’s are trans to each other in starting Alkene remains trans to each other in Epoxide.
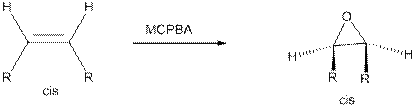
Epoxides from Halohydrins:
Epoxides can be obtained by the conversion of Alkene to Halohydrins. Halohydrins are formed when an Alkene is treated with halogen in presence of Water. Halohydrins formed are later treated with strong base to from Epoxides.
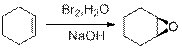
(b)
Interpretation: The reagents are to be identified for Synthesis of Racemic mixture of Epoxides.
Concept Introduction:
Epoxides are obtained by the conversion of Alkene using Peroxy acids and Halohydrins
Epoxides from Peroxy acids:
The commonly used Peroxy acids are meta-Chloroperoxybenzoic acid (MCPBA) and Peroxyacetic acid. This process is stereospecific.
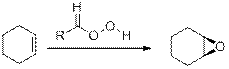
Specifically, the substituent are cis to each other in starting Alkene remains cis to each other in Epoxide, and the substituent’s are trans to each other in starting Alkene remains trans to each other in Epoxide.
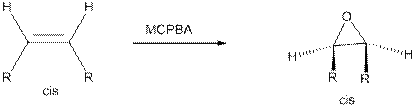
Epoxides from Halohydrins:
Epoxides can be obtained by the conversion of Alkene to Halohydrins. Halohydrins are formed when an Alkene is treated with halogen in presence of Water. Halohydrins formed are later treated with strong base to from Epoxides.
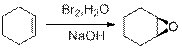
(c)
Interpretation: The reagents are to be identified for Synthesis of Racemic mixture of Epoxides.
Concept Introduction:
Epoxides are obtained by the conversion of Alkene using Peroxy acids and Halohydrins
Epoxides from Peroxy acids:
The commonly used Peroxy acids are meta-Chloroperoxybenzoic acid (MCPBA) and Peroxyacetic acid. This process is stereospecific.
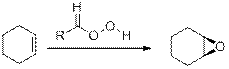
Specifically, the substituent are cis to each other in starting Alkene remains cis to each other in Epoxide, and the substituent’s are trans to each other in starting Alkene remains trans to each other in Epoxide.
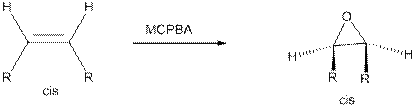
Epoxides from Halohydrins:
Epoxides can be obtained by the conversion of Alkene to Halohydrins. Halohydrins are formed when an Alkene is treated with halogen in presence of Water. Halohydrins formed are later treated with strong base to from Epoxides.
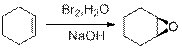
(d)
Interpretation: The reagents are to be identified for Synthesis of Racemic mixture of Epoxides.
Concept Introduction:
Epoxides are obtained by the conversion of Alkene using Peroxy acids and Halohydrins
Epoxides from Peroxy acids:
The commonly used Peroxy acids are meta-Chloroperoxybenzoic acid (MCPBA) and Peroxyacetic acid. This process is stereospecific.
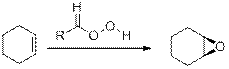
Specifically, the substituent are cis to each other in starting Alkene remains cis to each other in Epoxide, and the substituent’s are trans to each other in starting Alkene remains trans to each other in Epoxide.
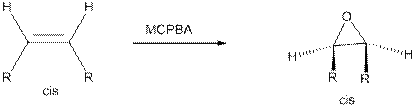
Epoxides from Halohydrins:
Epoxides can be obtained by the conversion of Alkene to Halohydrins. Halohydrins are formed when an Alkene is treated with halogen in presence of Water. Halohydrins formed are later treated with strong base to from Epoxides.
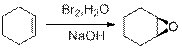
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry, 3e WileyPLUS Registration Card + Loose-leaf Print Companion
- Nonearrow_forward4. Draw and label all possible isomers for [M(py)3(DMSO)2(CI)] (py = pyridine, DMSO dimethylsulfoxide).arrow_forwardThe emission data in cps displayed in Table 1 is reported to two decimal places by the chemist. However, the instrument output is shown in Table 2. Table 2. Iron emission from ICP-AES Sample Blank Standard Emission, cps 579.503252562 9308340.13122 Unknown Sample 343.232365741 Did the chemist make the correct choice in how they choose to display the data up in Table 1? Choose the best explanation from the choices below. No. Since the instrument calculates 12 digits for all values, they should all be kept and not truncated. Doing so would eliminate significant information. No. Since the instrument calculates 5 decimal places for the standard, all of the values should be limited to the same number. The other decimal places are not significant for the blank and unknown sample. Yes. The way Saman made the standards was limited by the 250-mL volumetric flask. This glassware can report values to 2 decimal places, and this establishes our number of significant figures. Yes. Instrumental data…arrow_forward
- 7. Draw a curved arrow mechanism for the following reaction. HO cat. HCI OH in dioxane with 4A molecular sievesarrow_forwardTry: Convert the given 3D perspective structure to Newman projection about C2 - C3 bond (C2 carbon in the front). Also, show Newman projection of other possible staggered conformers and circle the most stable conformation. Use the template shown. F H3C Br Harrow_forwardNonearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





