
Concept explainers
(a)
Interpretation:
The mechanism for the given conversion has to be identified and the stereochemical outcome has to be justified.
Concept introduction:
Ring-opening of
Acid-catalyzed ring-opening of epoxide: The epoxide ring is protonated and the nucleophile attack depends on the electronic or steric effect (nature of epoxide).
Regiochemistry: when the epoxide is unsymmetrical, the nucleophile attack at the more substituted position of the protonated epoxide ring.
Base catalyzed ring opening of epoxide:
The nucleophile will attack at the less substituted position under basic conditions
(b)
Interpretation:
The product of the given process has to be predicted.
Concept introduction:
Sharpless epoxidation: An
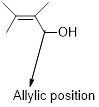
Sharpless catalyst consists of Titanium tetraiospropoxide and one enantiomer of Diethyl tartrate (
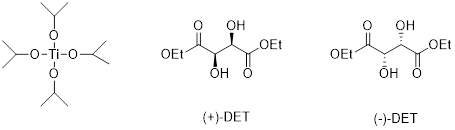
A chiral complex is formed with Titanium tetraisopropoxide and with either
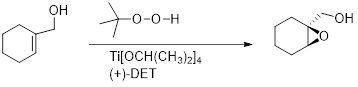
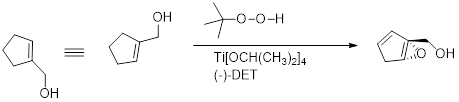
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Organic Chemistry, Third Edition Binder Ready Version
- Hi can you please help me solve this problem? thank youarrow_forwardAn electrode process takes place at a metal-solution interface. Indicate the current condition that must be met for Faradaic rectification to occur.arrow_forwardAt a metal-solution interface, an electron is exchanged, and the symmetry factor beta < 0.5 is found in the Butler-Volmer equation. What does this indicate?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





