
Concept explainers
To explain: The following,
'a'If isomaltose is a mono-, di-, or polysaccharide?
Answer to Problem 13.55UTC
Solution:
'a'Isomaltose is a disaccharide.
Given: Structure of isomaltose in Haworth projection
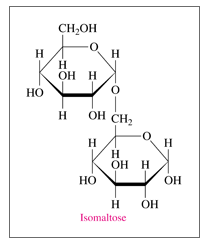
a.
Explanation of Solution
Isomaltose is a disaccharide as it contains two monosaccharides i.e.sugar units which are obtained after hydrolysis.
Therefore isomaltose is a disaccharide
b. The monosaccharides in isomaltose are two glucose units.
c. There is C1-C6 (a-a) O-glycosidic link in isomaltose.
Glycosidic link is the bond which connects two monosaccharides through anomeric carbon (a carbon which is coming from carbonyl group and attached with two oxygen atoms). In case of isomaltose, the anomeric carbon i.e. hemiacetal is linked with another sugar moiety through O atom. Thus this is a C1-C6 (a-a) O-glycosidic link.
Therefore, the monosaccharides in isomaltose are two glucose units and there is C1-C6 O-glycosidic link.
d. The given structure is an a-isomer of isomaltose.
As the OH group in the anomeric carbon is downward i.e. below the plane, so it is a-anomer (a-isomer of isomaltose).
Therefore the given structure is an a-isomer of isomaltose
e. Isomaltose is a reducing sugar
Isomaltose is a reducing sugar as it is capable of reducing Tollen’s reagent and Fehling solution. All the monosaccharides (aldoses) are reducing sugars along with some disaccharides. A disaccharide (as for example isomaltose) which has one anomeric carbon involved in glycosidic linkage but other anomeric carbon is free then it is able to reduce Tollen’s and Fehling solution.
Thus, isomaltose though a disaccharide is a reducing sugar.
- Isomaltose is a disaccharide.
- The monosaccharides in isomaltose are glucose units.
- The glycosidic link in isomaltose is C1-C6 (a-a) O-glycosidic link.
- The given structure is an a-isomer of isomaltose
- Isomaltose is a reducing sugar.
Want to see more full solutions like this?
Chapter 13 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- Draw the major product of this reaction. Nitropropane reacts + pent-3-en-2-one reacts with NaOCH2CH3, CH3CHOHarrow_forwardIndicate whether the product formed in the reaction exhibits tautomerism. If so, draw the structure of the tautomers. OC2H5 + CoHs-NH-NH,arrow_forwardExplain how substitutions at the 5-position of barbituric acid increase the compound's lipophilicity.arrow_forward
- Explain how substitutions at the 5-position of phenobarbital increase the compound's lipophilicity.arrow_forwardName an interesting derivative of barbituric acid, describing its structure.arrow_forwardBriefly describe the synthesis mechanism of barbituric acid from the condensation of urea with a β-diketone.arrow_forward
- Given the hydrazones indicated, draw the structures of the enamines that can be formed. Indicate the most stable enamine (explain). C6H5 C6H5 H C6H5 Harrow_forward4. Propose a Synthesis for the molecule below. You may use any starting materials containing 6 carbons or less (reagents that aren't incorporated into the final molecule such as PhзP do not count towards this total, and the starting material can have whatever non-carbon functional groups you want), and any of the reactions you have learned so far in organic chemistry I, II, and III. Your final answer should show each step separately, with intermediates and conditions clearly drawn.arrow_forwardIndicate the importance of the indole ring. Find a representative example and list 5 structures.arrow_forward
- ΌΗ 1) V2 CO 3 or Nalt In منهarrow_forward6. The equilibrium constant for the reaction 2 HBr (g) → H2(g) + Br2(g) Can be expressed by the empirical formula 11790 K In K-6.375 + 0.6415 In(T K-¹) - T Use this formula to determine A,H as a function of temperature. Calculate A,-H at 25 °C and at 100 °C.arrow_forward3. Nitrosyl chloride, NOCI, decomposes according to 2 NOCI (g) → 2 NO(g) + Cl2(g) Assuming that we start with no moles of NOCl (g) and no NO(g) or Cl2(g), derive an expression for Kp in terms of the equilibrium value of the extent of reaction, Seq, and the pressure, P. Given that K₂ = 2.00 × 10-4, calculate Seq/ of 29/no when P = 0.080 bar. What is the new value по ƒª/ at equilibrium when P = 0.160 bar? Is this result in accord with Le Châtelier's Principle?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





