
Chemistry: Atoms First
18th Edition
ISBN: 9781938168154
Author: Richard Langley, Klaus Theopold, Paul Flowers
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12, Problem 7E
In Figure 12.8 all possible distributions and microstates are shown for four different particles shared between two boxes. Determine the entropy change,
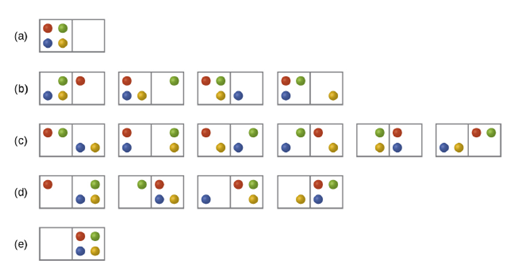
Figure 12.8 The sixteen microstates associated with placing four particles in two boxes are shown. The microstates are collected into five distributions-(a), (b), (c), (d), and (e)-based on the numbers of particles in each box.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using Benzene as starting materid show
how each of the Following molecules Contel
Ve syntheswed
CHI
9.
b
-50311
с
CHY
503H
Ночто
d.
อ
•NOV
e
11-0-650
NO2
The molecule PYRIDINE,
6th electrons and is therefore aromatre
and is Assigned the Following structure
contering
Since aromatk moleculoy undergo electrophilic
anomatic substitution, Pyridine shodd undergo
The Following reaction
+ HNO3
12504
a. write all of the possible Mononitration Products
that could Result From this reaction
18. Bared upon the reaction mechanison determime
which of these producty would be the major
Product of the hegetion
a. Explain Why electron withdrawing groups
tend to be meta-Directors. Your answer Should
lyclude all apropriate. Resonance contributing
Structures
fo. Explain why -ll is an outho -tura
drccton even though chlorine has a very High
Electronegativity
Chapter 12 Solutions
Chemistry: Atoms First
Ch. 12 - What is a spontaneous reaction?Ch. 12 - What is a nonspontaneous reaction?Ch. 12 - Indicate whether the following processes are...Ch. 12 - A helium-filled balloon spontaneously deflates...Ch. 12 - Many plastic materials are organic polymers that...Ch. 12 - In Figure 12.8 all possible distributions and...Ch. 12 - In Figure 12.8 all possible distributions and...Ch. 12 - How does the process described in the previous...Ch. 12 - Consider a system similar to the one in Figure...Ch. 12 - Consider the system shown in Figure 12.9. What is...
Ch. 12 - Consider the system shown in Figure 12.9. What is...Ch. 12 - Arrange the following sets of systems in order of...Ch. 12 - At room temperature, the entropy of the halogens...Ch. 12 - Consider two processes: sublimation of I2(s) and...Ch. 12 - Indicate which substance in the given pairs has...Ch. 12 - Predict the sign of the entropy change for the...Ch. 12 - Predict the sign of the entropy change for the...Ch. 12 - Write the balanced chemical equation for the...Ch. 12 - Write the balanced chemical equation for the...Ch. 12 - What is the difference between S and S for a...Ch. 12 - Calculate S for the following changes. (a)...Ch. 12 - Determine the entropy change for the combustion of...Ch. 12 - Determine the entropy change for the combustion of...Ch. 12 - Thermite reactions have been used for welding...Ch. 12 - Using the relevant S values listed in Appendix G,...Ch. 12 - From the following information, determine S for...Ch. 12 - By calculating Suniv, at each temperature,...Ch. 12 - Use the standard entropy data in Appendix G to...Ch. 12 - Use the standard entropy data in Appendix G to...Ch. 12 - What is the difference between G and G for a...Ch. 12 - A reaction has H=100kJ/mol and S=250J/mol.K . Is...Ch. 12 - Explain what happens as a reaction starts with G0...Ch. 12 - Use the standard free energy of formation data in...Ch. 12 - Use the standard free energy data in Appendix G to...Ch. 12 - Given: P4(s)+5O2(g)P4O10(s) G=2697.0kJ/mol...Ch. 12 - Is the formation of ozone (O3(g)) from oxygen...Ch. 12 - Consider the decomposition of red mercury(II)...Ch. 12 - Among other things, an ideal fuel for the control...Ch. 12 - Calculate G for each of the following reactions...Ch. 12 - Calculate G for each of the following reactions...Ch. 12 - Calculate the equilibrium constant at 25 C for...Ch. 12 - Determine G for the following reactions. (a)...Ch. 12 - Given that the Gf for Pb2+(aq) and Cl-(aq) is...Ch. 12 - Determine the standard free energy change, Gf, for...Ch. 12 - Determine the standard enthalpy change, entropy...Ch. 12 - The evaporation of one mole of water at 298 K has...Ch. 12 - In glycolysis, the reaction of glucose (Glu) to...Ch. 12 - One of the important reactions in the biochemical...Ch. 12 - Without doing a numerical calculation, determine...Ch. 12 - When ammonium chloride is added to water and...Ch. 12 - An important source of copper is from the copper...Ch. 12 - What happens to G (becomes more negative or more...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Veritious +H2504 4.50+ + 1/₂ Felly ◎+ 7 b. Praw he potential energy Diagrams For each OF Mese Ronctions and account for any differences that appeak in the two potential Puergy Diagramsarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 3 attempts remaining 1. excess Br2, NaOH 2. neutralizing workup Qarrow_forward
- Given the electrode Pt | Ag | Ag+ (aq), describe it.arrow_forwardAt 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.arrow_forwardElectrochemistry. State the difference between E and E0.arrow_forward
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?arrow_forwardIn an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forward
- If the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forwardGiven the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY