
College Physics: A Strategic Approach (4th Edition)
4th Edition
ISBN: 9780134704180
Author: Knight
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 75P
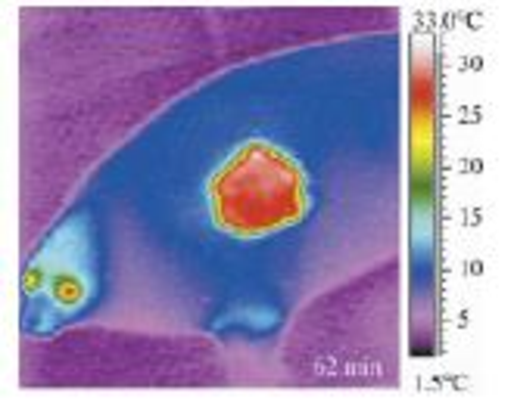
Seals may cool themselves by using thermal windows, patches on their bodies with much higher than average surface temperature. Suppose a seal has a 0.030 m2 thermal window at a temperature of 30°C. If the seal's surroundings are a frosty –10°C, what is the net rate of energy loss by
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part a-D pl
The figure (Figure 1) shows representations of six
thermodynamic states of the same ideal gas sample.
Figure
1 of 1
Part A
■Review | Constants
Rank the states on the basis of the pressure of the gas sample at each state.
Rank pressure from highest to lowest. To rank items as equivalent, overlap them.
▸ View Available Hint(s)
highest
0
☐ ☐ ☐ ☐ ☐ ☐
Reset
Help
B
F
A
D
E
The correct ranking cannot be determined.
Submit
Previous Answers
× Incorrect; Try Again; 4 attempts remaining
Provide Feedback
lowest
Next >
Part A
m
2πkT
) 3/2
Calculate the integral (v) = f vƒ (v)dv. The function f(v) describing the actual distribution of molecular speeds is called the Maxwell-Boltzmann distribution,
=
ƒ(v) = 4π (· v²e-mv²/2kT
. (Hint: Make the change of variable v² =x and use the tabulated integral foxne
integer and a is a positive constant.)
Express your answer in terms of the variables T, m, and appropriate constants.
-ax dx
n!
-
an+1
where n is a positive
(v)
=
ΕΠΙ ΑΣΦ
Submit Previous Answers Request Answer
?
× Incorrect; Try Again; 4 attempts remaining
Al Study Tools
Looking for some guidance? Let's work through a few related
practice questions before you go back to the real thing.
This won't impact your score, so stop at anytime and ask for
clarification whenever you need it.
Ready to give it a try?
Start
Chapter 12 Solutions
College Physics: A Strategic Approach (4th Edition)
Ch. 12 - Which has more mass, a mole of Ne gas or a mole of...Ch. 12 - If you launch a projectile upward with a high...Ch. 12 - Prob. 3CQCh. 12 - If you double the typical speed of the molecules...Ch. 12 - Two gases have the same number of molecules per...Ch. 12 - If the temperature T of an ideal gas doubles, by...Ch. 12 - A bottle of helium gas and a bottle of argon gas...Ch. 12 - A gas cylinder contains 1.0 mol of helium at a...Ch. 12 - Prob. 9CQCh. 12 - Prob. 10CQ
Ch. 12 - You need to precisely measure the dimensions of a...Ch. 12 - A common trick for opening a stubborn lid on a jar...Ch. 12 - Prob. 13CQCh. 12 - Materials A and B have equal densities, but A has...Ch. 12 - Prob. 15CQCh. 12 - You need to raise the temperature of a gas by 10C....Ch. 12 - Prob. 18CQCh. 12 - Prob. 19CQCh. 12 - A sample of ideal gas is in a cylinder with a...Ch. 12 - A student is heating chocolate in a pan on the...Ch. 12 - If you bake a cake at high elevation, where...Ch. 12 - Prob. 23CQCh. 12 - Prob. 24CQCh. 12 - Prob. 25CQCh. 12 - Prob. 26CQCh. 12 - Prob. 27CQCh. 12 - Prob. 29CQCh. 12 - Prob. 30MCQCh. 12 - Prob. 31MCQCh. 12 - A gas is compressed by an isothermal process that...Ch. 12 - Prob. 33MCQCh. 12 - Prob. 34MCQCh. 12 - Prob. 35MCQCh. 12 - Prob. 36MCQCh. 12 - Prob. 37MCQCh. 12 - Prob. 38MCQCh. 12 - Prob. 1PCh. 12 - How many grams of water (H2O) have the same number...Ch. 12 - Prob. 3PCh. 12 - How many cubic millimeters (mm3) are in 1 L?Ch. 12 - Prob. 5PCh. 12 - Prob. 6PCh. 12 - Prob. 7PCh. 12 - Prob. 8PCh. 12 - An ideal gas is at 20C. The gas is cooled,...Ch. 12 - An ideal gas at 0C consists of 1.0 1023 atoms. 10...Ch. 12 - An ideal gas at 20C consists of 2.2 1022 atoms....Ch. 12 - Prob. 12PCh. 12 - Prob. 13PCh. 12 - When you stifle a sneeze, you can damage delicate...Ch. 12 - Prob. 15PCh. 12 - Mars has an atmosphere composed almost entirely of...Ch. 12 - Prob. 18PCh. 12 - The lowest pressure ever obtained in a laboratory...Ch. 12 - Prob. 20PCh. 12 - Helium has the lowest condensation point of any...Ch. 12 - Prob. 22PCh. 12 - Prob. 23PCh. 12 - Prob. 24PCh. 12 - Prob. 25PCh. 12 - Prob. 26PCh. 12 - A cylinder contains 3.0 L of oxygen at 300 K and...Ch. 12 - Prob. 28PCh. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - Prob. 30PCh. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - Prob. 33PCh. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - A 1.0 cm3 air bubble is released from the sandy...Ch. 12 - A weather balloon rises through the atmosphere,...Ch. 12 - Prob. 39PCh. 12 - Prob. 40PCh. 12 - Prob. 41PCh. 12 - Prob. 42PCh. 12 - The length of a steel beam increases by 0.73 mm...Ch. 12 - Prob. 44PCh. 12 - The length of a steel beam increases by 0.73 mm...Ch. 12 - At 20C, the hole in an aluminum ring is 2.500 cm...Ch. 12 - The temperature of an aluminum disk is increased...Ch. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - a. 100 J of heat energy are transferred to 20 g of...Ch. 12 - Prob. 52PCh. 12 - Prob. 53PCh. 12 - Prob. 54PCh. 12 - Alligators and other reptiles dont use enough...Ch. 12 - Prob. 56PCh. 12 - When air is inhaled, it quickly becomes saturated...Ch. 12 - Prob. 58PCh. 12 - Prob. 59PCh. 12 - What minimum heat is needed to bring 100 g of...Ch. 12 - Brewed coffee is often too hot to drink right...Ch. 12 - Prob. 62PCh. 12 - 30 g of copper pellets are removed from a 300C...Ch. 12 - Prob. 64PCh. 12 - A copper block is removed from a 300C oven and...Ch. 12 - Prob. 66PCh. 12 - If a person has a dangerously high fever,...Ch. 12 - A container holds 1.0 g of argon at a pressure of...Ch. 12 - A container holds 1.0 g of oxygen at a pressure of...Ch. 12 - What is the temperature change of 1.0 mol of a...Ch. 12 - Heating 2.5 mol of neon in a rigid container...Ch. 12 - Prob. 72PCh. 12 - A 1.8-cm-thick wood floor covers a 4.0 m 5.5 m...Ch. 12 - A stainless-steel-bottomed kettle, its bottom 24...Ch. 12 - Seals may cool themselves by using thermal...Ch. 12 - Electronics and inhabitants of the International...Ch. 12 - The glowing filament in a lamp is radiating energy...Ch. 12 - Prob. 78PCh. 12 - If you lie on the ground at night with no cover,...Ch. 12 - Prob. 80PCh. 12 - Prob. 81PCh. 12 - Prob. 82PCh. 12 - Prob. 83GPCh. 12 - Prob. 84GPCh. 12 - Prob. 85GPCh. 12 - Prob. 86GPCh. 12 - Prob. 87GPCh. 12 - A 5.0-m-diameter garden pond holds 5.9 103 kg of...Ch. 12 - James Joule (after whom the unit of energy is...Ch. 12 - Prob. 90GPCh. 12 - Prob. 91GPCh. 12 - Prob. 92GPCh. 12 - A 68 kg woman cycles at a constant 15 km/h. All of...Ch. 12 - Prob. 94GPCh. 12 - Prob. 95GPCh. 12 - Prob. 97GPCh. 12 - Your 300 mL cup of coffee is too hot to drink when...Ch. 12 - A gas is compressed from 600 cm3 to 200 cm3 at a...Ch. 12 - An expandable cube, initially 20 cm on each side,...Ch. 12 - Prob. 101GPCh. 12 - Prob. 102GPCh. 12 - Prob. 103GPCh. 12 - Homes are often insulated with fiberglass...Ch. 12 - The surface area of an adult human is about 1.8...Ch. 12 - Prob. 106MSPPCh. 12 - Prob. 107MSPPCh. 12 - Prob. 108MSPPCh. 12 - Prob. 109MSPPCh. 12 - Prob. 110MSPPCh. 12 - Prob. 111MSPPCh. 12 - Prob. 112MSPP
Additional Science Textbook Solutions
Find more solutions based on key concepts
In pea plants, plant height, seed shape, and seed color are governed by three independently assorting genes. Th...
Genetic Analysis: An Integrated Approach (3rd Edition)
Choose the best answer to each of the following. Explain your reasoning. Which one of the following does the tr...
Cosmic Perspective Fundamentals
An obese 55-year-old woman consults her physician about minor chest pains during exercise. Explain the physicia...
Biology: Life on Earth with Physiology (11th Edition)
Chlorine has two isotopes, 35Cl and 37Cl; 75.77% of chlorine is 35Cl, and 24.23% is 37Cl. The atomic mass of 35...
Organic Chemistry (8th Edition)
89. Write isotopic symbols in the form for each isotope.
a. the oxygen isotope with 8 neutrons
b. the fluorine ...
Introductory Chemistry (6th Edition)
25. FIGURE EX4.25 shows the angular-velocity-versus-time graph for a particle moving in a circle, starting from...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Starter the rule of significantarrow_forwardPlease solve this problem and give step by step explanations on each step while breaking it down please. Thank you!!arrow_forwardPlease solve this problem and give step by step explanations on each step while breaking it down please. Thank you!!arrow_forward
- No chatgpt plsarrow_forwardNo chatgpt plsarrow_forwardCar A starts from rest at t = 0 and travels along a straight road with a constant acceleration of 6 ft/s^2 until it reaches a speed of 60ft/s. Afterwards it maintains the speed. Also, when t = 0, car B located 6000 ft down the road is traveling towards A at a constant speed of 80 ft/s. Determine the distance traveled by Car A when they pass each other.Write the solution using pen and draw the graph if needed.arrow_forward
- In the given circuit the charge on the plates of 1 μF capacitor, when 100 V battery is connected to the terminals A and B, will be 2 μF A 1 µF B 3 µFarrow_forwardThe velocity of a particle moves along the x-axis and is given by the equation ds/dt = 40 - 3t^2 m/s. Calculate the acceleration at time t=2 s and t=4 s. Calculate also the total displacement at the given interval. Assume at t=0 s=5m.Write the solution using pen and draw the graph if needed.arrow_forwardThe velocity of a particle moves along the x-axis and is given by the equation ds/dt = 40 - 3t^2 m/s. Calculate the acceleration at time t=2 s and t=4 s. Calculate also the total displacement at the given interval. Assume at t=0 s=5m.Write the solution using pen and draw the graph if needed.arrow_forward
- The velocity of a particle moves along the x-axis and is given by the equation ds/dt = 40 - 3t^2 m/s. Calculate the acceleration at time t=2 s and t=4 s. Calculate also the total displacement at the given interval. Assume at t=0 s=5m.Write the solution using pen and draw the graph if needed. NOT AI PLSarrow_forwardThe velocity of a particle moves along the x-axis and is given by the equation ds/dt = 40 - 3t^2 m/s. Calculate the acceleration at time t=2 s and t=4 s. Calculate also the total displacement at the given interval. Assume at t=0 s=5m.Write the solution using pen and draw the graph if needed.arrow_forwardThe velocity of a particle moves along the x-axis and is given by the equation ds/dt = 40 - 3t^2 m/s. Calculate the acceleration at time t=2 s and t=4 s. Calculate also the total displacement at the given interval. Assume at t=0 s=5m.Write the solution using pen and draw the graph if needed.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning


College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY