
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
2nd Edition
ISBN: 9780137390922
Author: Tro
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 70E
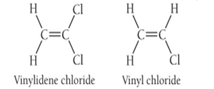
Saran, the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In natural rubber or polyisoprene, the trans isomer leads to a higher degree of crystallinity and density than the cis isomer of the same polymer, because(a) it is more symmetrical and regular.(b) it is less symmetrical.(c) it is irregular.
Most ceramic materials have low thermal conductivities because:(a) Electron mobility is strongly restricted due to their strong ionic-covalent bonding.(b) False, in general they are excellent thermal conductors (they are used in ovens).(c) Electron mobility is dependent on T and therefore they are poor conductors at high temperatures.(d) Electron mobility is very restricted by secondary bonds.
Resistivity and electrical conductivity.(a) In metals, resistivity decreases.(b) In metals, resistivity decreases and conductivity in semiconductors also decreases with increasing temperature.(c) With increasing temperature, resistivity in metals and conductivity in semiconductors also increases.(d) None of the above.
Chapter 12 Solutions
CHEM: STRUC & PROP (LL) VOL 2 >C PKG<
Ch. 12 - What is graphene? Why is graphene unique?Ch. 12 - Prob. 2ECh. 12 - What is a crystalline lattice? How is the lattice...Ch. 12 - Prob. 4ECh. 12 - Prob. 5ECh. 12 - What is the difference between hexagonal closest...Ch. 12 - What are the three basic types of solids and the...Ch. 12 - Prob. 8ECh. 12 - What kinds of forces hold each of the three basic...Ch. 12 - Prob. 10E
Ch. 12 - In an ionic compound, how are the relative sizes...Ch. 12 - Prob. 12ECh. 12 - Show how the fluorite structure accommodates a...Ch. 12 - Prob. 14ECh. 12 - Prob. 15ECh. 12 - Prob. 16ECh. 12 - Prob. 17ECh. 12 - Prob. 18ECh. 12 - Prob. 19ECh. 12 - Describe the difference between vitreous silica...Ch. 12 - Prob. 21ECh. 12 - Prob. 22ECh. 12 - Prob. 23ECh. 12 - Prob. 24ECh. 12 - What is a polymer? What is the difference between...Ch. 12 - Prob. 26ECh. 12 - Prob. 27ECh. 12 - An X-ray beam of unknown wavelength is diffracted...Ch. 12 - Prob. 29ECh. 12 - Determine the coordination number for each...Ch. 12 - Calculate the packing efficiency of the...Ch. 12 - Prob. 32ECh. 12 - Prob. 33ECh. 12 - Molybdenum crystallizes with the body-centred unit...Ch. 12 - Rhodium has a density of 12.41 g / cm3 and...Ch. 12 - Barium has a density of 3.59 g/cm3 and...Ch. 12 - Prob. 37ECh. 12 - Palladium crystallizes with a face-centered cubic...Ch. 12 - Prob. 39ECh. 12 - Identify each solid as molecular, ionic, or...Ch. 12 - Which solid has the highest melting point? Why?...Ch. 12 - Which solid has the highest melting point? Why?...Ch. 12 - Which solid in each pair has the higher melting...Ch. 12 - Which solid in each pair has the higher melting...Ch. 12 - Prob. 45ECh. 12 - Prob. 46ECh. 12 - The unit cells for cesium chloride and barium(ll)...Ch. 12 - Prob. 48ECh. 12 - Prob. 49ECh. 12 - Prob. 50ECh. 12 - Prob. 51ECh. 12 - Consider the zinc blende structure in Figure 12.14...Ch. 12 - Prob. 53ECh. 12 - Prob. 54ECh. 12 - Prob. 55ECh. 12 - What are the name and formula of the compound...Ch. 12 - Prob. 57ECh. 12 - Prob. 58ECh. 12 - Prob. 59ECh. 12 - Prob. 60ECh. 12 - Prob. 61ECh. 12 - How many molecular orbitals are present in the...Ch. 12 - Prob. 63ECh. 12 - Prob. 64ECh. 12 - Prob. 65ECh. 12 - Prob. 66ECh. 12 - Prob. 67ECh. 12 - Prob. 68ECh. 12 - Prob. 69ECh. 12 - Saran, the polymer used to make saran wrap, is an...Ch. 12 - One kind of polyester is a condensation copolymer...Ch. 12 - Nomex, a condensation copolymer used by...Ch. 12 - Prob. 73ECh. 12 - Polyacrylonitrile (PAN) is an addition polymer...Ch. 12 - Prob. 75ECh. 12 - Prob. 76ECh. 12 - Prob. 77ECh. 12 - Prob. 78ECh. 12 - Prob. 79ECh. 12 - Prob. 80ECh. 12 - Prob. 81ECh. 12 - The density of an unknown metal is 12.3 g/cm3 and...Ch. 12 - An unknown metal is found to have a density of...Ch. 12 - Prob. 84ECh. 12 - Prob. 85ECh. 12 - Prob. 86ECh. 12 - Prob. 87ECh. 12 - Prob. 88ECh. 12 - Prob. 89ECh. 12 - Prob. 90ECh. 12 - Prob. 91ECh. 12 - Perovskite is a compound with a cubic unit cell...Ch. 12 - Prob. 93ECh. 12 - Despite Dalton's laws, it is now known that many...Ch. 12 - During the glass manufacturing process, the liquid...Ch. 12 - Why are X-rays used for crystallography? Why not...Ch. 12 - Compare the crystal structure of diamond (C) and...Ch. 12 - Prob. 98ECh. 12 - Prob. 99ECh. 12 - Have each group member select one of the cubic...Ch. 12 - Prob. 101ECh. 12 - Prob. 102ECh. 12 - Prob. 103ECh. 12 - Prob. 104ECh. 12 - Prob. 105ECh. 12 - Prob. 1SAQCh. 12 - A crystalline solid has a body-centered cubic...Ch. 12 - Prob. 3SAQCh. 12 - Prob. 4SAQCh. 12 - Prob. 5SAQCh. 12 - Prob. 6SAQCh. 12 - Prob. 7SAQCh. 12 - Prob. 8SAQCh. 12 - Prob. 9SAQCh. 12 - Prob. 10SAQCh. 12 - Prob. 11SAQCh. 12 - Prob. 12SAQCh. 12 - Prob. 13SAQCh. 12 - Prob. 14SAQCh. 12 - Prob. 15SAQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- State the difference between concrete and Portland cement.(a) There are no differences, in concrete the chemical composition is silicates and in cement aluminates.(b) The chemical composition of concrete is based on silicates and in cement aluminates.(c) Concrete is composed of aggregates bound by cement and cement "only" contains different minerals.(d) Cement is aggregates bound by concrete.arrow_forwardAmorphous polymers are usually transparent and semi-crystalline polymers are usually opaque. Correct?(a) No. They are all made up of polymer chains. True if they were monomers.(b) Yes. The arrangement of the chains determines the passage of light.(c) No. It is the other way around.(d) Crystallinity or amorphousness does not affect the transparency or opacity of the material.arrow_forwardThe name ferrites refers to a family of(a) ceramic materials that exhibit ferrimagnetic behavior due to their ionic composition.(b) polymeric materials that exhibit ferrimagnetic behavior due to their ionic composition.(c) concrete-based materials that exhibit ferrimagnetic behavior due to their ionic composition.(d) superconducting materials that exhibit ferrimagnetic behavior due to their ionic composition.arrow_forward
- State the two main factors affecting ion packing in the solid state.(a) Number of covalent bonds and their unsaturation.(b) Mechanical properties and degradation temperature.(c) Number of crystalline phases present and grain size.(d) Electroneutrality and ion size.arrow_forwardThe ceramic materials alumina (Al2O3) and chromium oxide (Cr2O3) form an isomorphic phase diagram. The solubility will be(a) unlimited of one ceramic in the other.(b) very limited depending on the weight % of Al2O3(c) very limited depending on the weight % of Cr2O3(d) partial of one ceramic in the other.arrow_forwardAmong the main characteristics of optical fibers, indicate which of the following is not included:(a) Opacity and Rigidity(b) Flexibility(c) Transparency(d) Low thicknessarrow_forward
- Most ceramic materials have low thermal conductivities because(a) Electron mobility is strongly restricted due to their strong ionic-covalent bonding.(b) False, in general they are excellent thermal conductors (they are used in ovens).(c) Electron mobility is dependent on T and therefore they are poor conductors at high temperatures.(d) Electron mobility is highly restricted by secondary bonds.arrow_forwardSi increases its conductivity when doped with Ga and P.(a) True, because the conduction mechanism is due to electrons and holes generated by Ga and P as the case may be.(b) True, because a completely different compound is generated.(c) False, because when impurities are introduced, the opposite occurs.(d) False, because the conductivity of Si is only determined by the increase in temperature, which must be controlled.arrow_forwardIndicate whether a configuration and a microstate are the same:a) Yesb) No, a microstate encompasses several configurationsc) No, a configuration is the same as a macrostated) No, a configuration encompasses several microstatesarrow_forward
- The representation of a one-dimensional velocity distribution function for a gas, with increasing temperature the maximum occurs for vi = 0 m/s. Correct?arrow_forwardThe representation of a one-dimensional velocity distribution function for a gas, as the temperature increases:a) it becomes more flattenedb) the maximum occurs for vi = 0 m/sExplain it.arrow_forwardThe velocity distribution function of gas moleculesa) is used to measure their velocity, since the small size of gas molecules means that it cannot be measured in any other wayb) is only used to describe the velocity of particles if their density is very high.c) describes the probability that a gas particle has a velocity in a given interval of velocitiesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY