
Concept explainers
Draw the products formed when A is treated with each reagent: (a)
A
(a)
Interpretation: The product formed when A is treated with
Concept introduction: The addition of
Answer to Problem 28P
The product formed when A is treated with
Explanation of Solution
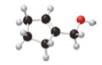
The given molecule is,
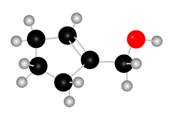
Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of A is,
Figure 2
When A is treated with
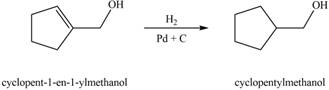
Figure 3
(b)
Interpretation: The product formed when A is treated with
Concept introduction: In presence of peroxide alkene is oxidized to epoxide this is known as epoxidation. The weak pi bond of alkene and weak
Answer to Problem 28P
The product formed when A is treated with
Explanation of Solution
The given molecule is,
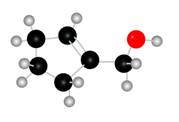
Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the A is,

Figure 2
In presence of
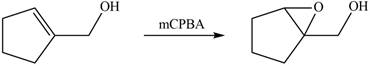
Figure 4
(c)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In presence of strong oxidizing reagents such as
Answer to Problem 28P
The product formed when A is treated with
Explanation of Solution
The given molecule is,
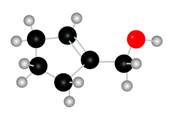
Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the A is,
Figure 2
The chemical reaction of A with
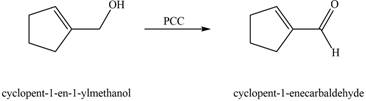
Figure 5
The primary alcohol is oxidized to aldehyde in presence of mild oxidizing reagent
(d)
Interpretation: The product formed when A is treated with
Concept introduction: Alcohols are oxidized to different carbonyl compounds depending upon the reagents and alcohol used. In presence of strong oxidizing reagents such as
Answer to Problem 28P
The product formed when A is treated with
Explanation of Solution
The given molecule is

Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the A is,

Figure 2
When A is treated with
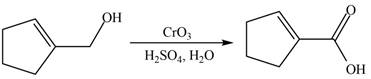
Figure 6
The primary alcohol is oxidized to carboxylic acid in presence of strong oxidizing reagent
(e)
Interpretation: The product of formed when A is treated with Sharpless reagent
Concept introduction: Sharpless epoxidation involves the oxidation of double bond between carbon atoms to epoxide. This oxidation occurs only in allylic alcohol. This is an enantioselective oxidation, which means predominantly one enantiomer is formed. Sharpless reagents are
Answer to Problem 28P
The product of formed when A is treated with Sharpless reagent
Explanation of Solution
The given molecule is
Figure 1
The red coloured balls have two bonds. So, these are the oxygen atoms. Black coloured atoms have four bonds. So, these are the carbon atoms. The grey coloured balls have one bond. So, these are the hydrogen atoms. The molecular structure of the A is,
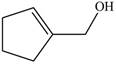
Figure 2
When epoxidation is done using
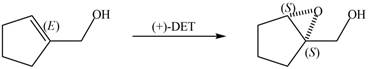
Figure 7
(a) The product formed when A is treated with
(b) The product formed when A is treated with
(c) The product formed when A is treated with
(d) The product formed when A is treated with
(e) The product of formed when A is treated with Sharpless reagent
Want to see more full solutions like this?
Chapter 12 Solutions
CNCT ORG CHEM 6 2020
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Physical Science
Laboratory Manual For Human Anatomy & Physiology
Campbell Biology (11th Edition)
Fundamentals Of Thermodynamics
- ↑ 0 Quiz List - RCC430M_RU05 X Aktiv Learning App × Qdraw resonance structure ×Q draw resonance structure xb My Questions | bartleby ×+ https://app.aktiv.com Draw a resonance structure of pyrrole that has the same number of pi bonds as the original structure. Include all lone pairs in your structure. + N H a 5 19°F Cloudy Q Search Problem 12 of 15 Atoms, Bonds and Rings Charges and Lone Pairs myhp हजु Undo Reset Remove Done Submit Drag To Pan 2:15 PM 1/25/2025arrow_forwardDon't used hand raitingarrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forward
- Indicate the coordination forms of Si in silicates.arrow_forwardBriefly indicate the structure and bonding of silicates.arrow_forward4 Part C Give the IUPAC name and a common name for the following ether: Spell out the full names of the compound in the indicated order separated by a comma.arrow_forward
- Try: Draw possible resonance contributing structures for the following organic species: CH3CH2NO2 [CH2CHCH2] [CH2CHCHO] [CH2CHCH2] [CH2CHNH2]arrow_forwardComplete the following synthesis. (d). H+ ง сarrow_forwardCan the target compound be efficiently synthesized in good yield from the substituted benzene of the starting material? If yes, draw the synthesis. Include all steps and all reactants.arrow_forward
