
(a)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkene used to synthesize the given compound is
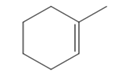
The necessary reagents and special reaction conditions for the synthesis are
Explanation of Solution
The structure of the given compound is
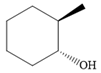
Alcohol with a specific stereochemistry is to be synthesized from an alkene. A reaction involving a carbocation needs to be avoided to prevent unwanted carbocation rearrangements. Also, the OH group must be added to a less substituted carbon, i.e., an anti-Markovnikov addition is needed. Therefore, the reaction needs to be carried out using hydroboration-oxidation.
The appropriate alkene for the synthesis of the given compound would be
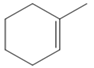
The given compound is synthesized by using the above alkene via hydroboration-oxidation reaction. So the necessary reagents for the reaction are
In the first step, an electrophilic addition of borane across the double bond of the alkene takes place either from above or below the plane of the alkene. So a mixture of enantiomers is obtained after oxidation of the adduct by basic
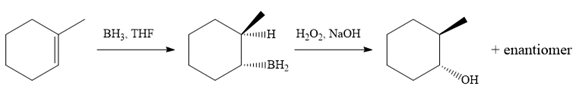
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(b)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkyne used to synthesize the given compound is
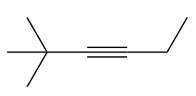
The necessary reagents and special reaction conditions to synthesize the given compound are
Explanation of Solution
The given compound is
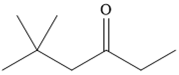
It is a ketone, so the starting compound must be an alkyne. A hydroboration-oxidation reaction can convert an alkyne into a ketone. Since only one molecule of borane is to be added, a bulky reagent like disiamylborane is more appropriate than borane. Also, the dialkylborane part must add to the less hindered carbon of the triple bond. Therefore, the triple bond must be between the carbon bonded to oxygen and the carbon close to the bulky tertiary carbon. Therefore, the alkyne that can be used is

A sterically hindered dialkylborane, like disiamylborane
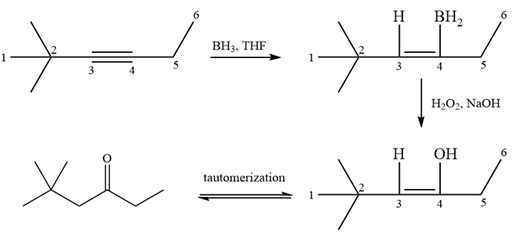
Thus the specific reagents and reaction conditions are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(c)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkyne used to synthesize the given compound is
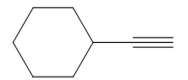
The necessary reagents and special reaction conditions to synthesize the given compound are
Explanation of Solution
The given compound is
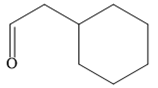
It is an aldehyde, so it can be prepared from a terminal alkyne by hydroboration-oxidation. In the hydroboration reaction, boron is added to the terminal carbon. So the appropriate alkyne for the synthesis of the given compound is
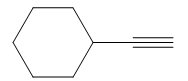
The alkyne is treated with the bulky disiamylborane to prevent the addition of a second molecule and formation of a mixture of products. Subsequent treatment of the adduct by
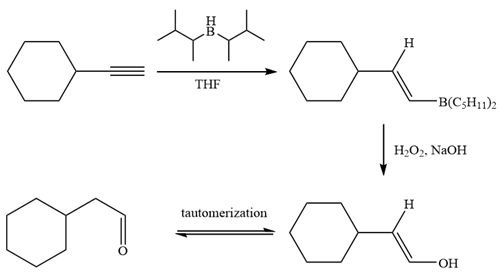
Thus, the necessary reagents and special reaction conditions for the synthesis are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
(d)
Interpretation:
The appropriate alkene or alkyne from which the given compound can be produced is to be determined along with the necessary reagents and special reaction conditions.
Concept introduction:
Oxidation of alkenes and alkynes is carried out by the hydroboration-oxidation reaction. Alkenes are oxidized to alcohol while alkynes are oxidized to the corresponding carbonyl compounds. Terminal alkynes are oxidized to the corresponding aldehyde, and internal alkynes are oxidized to the corresponding ketone by a sterically hindered borane like disiamylborane
The initial addition of borane is a syn, anti-Markovnikov addition via a four-membered cyclic transition state. Boron adds to the less substituted (less hindered) carbon because it is less electronegative than hydrogen. This results in an anti-Markovnikov addition of a water molecule to the double bond.
Answer to Problem 12.53P
The appropriate alkene used to synthesize the given compound is
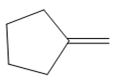
The necessary reagents and special reaction conditions for the synthesis are
Explanation of Solution
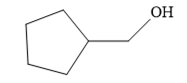
Since the product is an alcohol, an alkene with a methylene substituent on a cyclopentane ring would be appropriate as the starting compound.

Treating this alkene with borane in THF will add
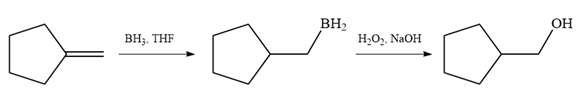
Thus, the necessary reagents for the reaction are
The alkene necessary for the synthesis of the given compound and the reagents and special conditions for the reaction are determined on the basis of the structure of the given compound.
Want to see more full solutions like this?
Chapter 12 Solutions
EBK ORGANIC CHEMISTRY: PRINCIPLES AND M
- curved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the cured electron-pushing arrows for thw following reaction or mechanistic steps. be sure to account for all bond-breaking and bond making stepsarrow_forwardUsing the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.arrow_forwardSynthesis of Dibenzalacetone [References] Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below. Question 1 1 pt Question 2 1 pt Question 3 1 pt H Question 4 1 pt Question 5 1 pt Question 6 1 pt Question 7 1pt Question 8 1 pt Progress: 7/8 items Que Feb 24 at You do not have to consider stereochemistry. . Draw the enolate ion in its carbanion form. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. ⚫ Separate multiple reactants using the + sign from the drop-down menu. ? 4arrow_forward
- Shown below is the mechanism presented for the formation of biasplatin in reference 1 from the Background and Experiment document. The amounts used of each reactant are shown. Either draw or describe a better alternative to this mechanism. (Note that the first step represents two steps combined and the proton loss is not even shown; fixing these is not the desired improvement.) (Hints: The first step is correct, the second step is not; and the amount of the anhydride is in large excess to serve a purpose.)arrow_forwardHi I need help on the question provided in the image.arrow_forwardDraw a reasonable mechanism for the following reaction:arrow_forward
- Draw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M HCl is titrated with 37.75 mL of NaOH. What is the molarity of the NaOH?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning


