
ORGANIC CHEMISTRY
4th Edition
ISBN: 9781119745105
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.5, Problem 10ATS
Interpretation Introduction
Interpretation: Synthetic pathway of each of these compounds using
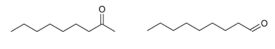
Concept introduction: One of the products is an
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identifying electron-donating and
For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the
benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene.
Molecule
Inductive Effects
NH2
○ donating
NO2
Explanation
Check
withdrawing
no inductive effects
Resonance Effects
Overall Electron-Density
○ donating
O withdrawing
O no resonance effects
O donating
O withdrawing
O donating
withdrawing
O no inductive effects
Ono resonance effects
O electron-rich
electron-deficient
O similar to benzene
O electron-rich
O electron-deficient
O similar to benzene
olo
18
Ar
2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
Rank each of the following substituted benzene molecules in order of which will react fastest (1) to slowest (4) by electrophilic
aromatic substitution.
Explanation
Check
Х
(Choose one)
OH
(Choose one)
OCH3
(Choose one)
OH
(Choose one)
© 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center
Assign R or S to all the chiral centers in each compound drawn below
porat
bg
9
Br
Br
Chapter 11 Solutions
ORGANIC CHEMISTRY
Ch. 11.1 - Prob. 1CCCh. 11.1 - Prob. 2CCCh. 11.2 - Prob. 1LTSCh. 11.2 - Prob. 3PTSCh. 11.3 - Prob. 2LTSCh. 11.3 - Prob. 6ATSCh. 11.4 - Prob. 3LTSCh. 11.4 - Prob. 7PTSCh. 11.4 - Prob. 8ATSCh. 11.5 - Prob. 4LTS
Ch. 11.5 - Propose an efficient synthesis for each of the...Ch. 11.5 - Prob. 10ATSCh. 11 - Prob. 11PPCh. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Prob. 19PPCh. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Prob. 25PPCh. 11 - Prob. 26ASPCh. 11 - Prob. 27ASPCh. 11 - Prob. 28ASPCh. 11 - Prob. 29ASPCh. 11 - Prob. 30ASPCh. 11 - Prob. 31ASPCh. 11 - Prob. 32ASPCh. 11 - Prob. 33IPCh. 11 - Prob. 34IPCh. 11 - Prob. 35IPCh. 11 - Prob. 36IPCh. 11 - Prob. 37IPCh. 11 - Prob. 38IPCh. 11 - Prob. 39IPCh. 11 - Prob. 42CPCh. 11 - The compound Z-3-hexenyl acetate is one of several...Ch. 11 - When a consumer purchases a tomato, smell is one...Ch. 11 - Prob. 45CPCh. 11 - Prob. 46CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (c) (4pts) Mechanism: heat (E1) CH3OH + 1.5pts each _E1 _ (1pt) Br CH3OH (d) (4pts) Mechanism: SN1 (1pt) (e) (3pts) 1111 I H 10 Ill!! H LDA THF (solvent) Mechanism: E2 (1pt) NC (f) Bri!!!!! CH3 NaCN (3pts) acetone Mechanism: SN2 (1pt) (SN1) -OCH3 OCH3 1.5pts each 2pts for either product 1pt if incorrect stereochemistry H Br (g) “,、 (3pts) H CH3OH +21 Mechanism: SN2 (1pt) H CH3 2pts 1pt if incorrect stereochemistry H 2pts 1pt if incorrect stereochemistryarrow_forwardA mixture of butyl acrylate and 4'-chloropropiophenone has been taken for proton NMR analysis. Based on this proton NMR, determine the relative percentage of each compound in the mixturearrow_forwardQ5: Label each chiral carbon in the following molecules as R or S. Make sure the stereocenter to which each of your R/S assignments belong is perfectly clear to the grader. (8pts) R OCH 3 CI H S 2pts for each R/S HO R H !!! I OH CI HN CI R Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning