
EBK CHEMISTRY
13th Edition
ISBN: 9780134564630
Author: Timberlake
Publisher: YUZU
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11.3, Problem 11.14PP
Give the TUPAC name for each of the following:
a. 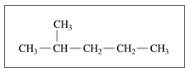
b. 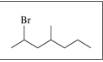
c. 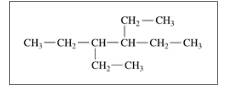
d. 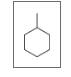
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Which is NOT the typical size of a bacteria?
1000 nm
0.001 mm
0.01 mm
1 um
None
Show work. don't give Ai generated solution
Chapter 11 Solutions
EBK CHEMISTRY
Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following as a formula of an...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Identify each of the following properties as more...Ch. 11.1 - Prob. 11.5PPCh. 11.1 - Prob. 11.6PPCh. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Give the IUPAC name for each of the following...Ch. 11.2 - Draw the condensed structural formula for alkanes...Ch. 11.2 - Draw the condensed structural formula for alkanes...
Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Indicate whether each of the following pairs...Ch. 11.3 - Give the IUPAC name for each of the following a....Ch. 11.3 - Give the TUPAC name for each of the following: a....Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the condensed structural formula for each of...Ch. 11.3 - Draw the line-angle formula for each of the...Ch. 11.3 - Prob. 11.18PPCh. 11.4 - Heptane, used as a solvent for rubber cement, has...Ch. 11.4 - Nonane has a density of 0.79 g/mL and boils at 151...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.4 - Write the balanced chemical equation for the...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Identify the following as alkanes, alkenes,...Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Give the IUPAC name for each of the following: a....Ch. 11.5 - Draw the condensed structural formula, or...Ch. 11.5 - Prob. 11.28PPCh. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Give the IUPAC name for each of the following,...Ch. 11.6 - Draw the condensed structural formula for each of...Ch. 11.6 - Prob. 11.32PPCh. 11.7 - Draw the structural formula for the product in...Ch. 11.7 - Draw the structural formula for the product in...Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Give the IUPAC name for each of the following: a....Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Draw the line-angle formula for each of the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11.8 - Write the balanced chemical equation for the...Ch. 11 - Prob. 11.41UTCCh. 11 - Prob. 11.42UTCCh. 11 - Prob. 11.43UTCCh. 11 - Prob. 11.44UTCCh. 11 - Convert each of the following line-angle formulas...Ch. 11 - Convert each of the following line-angle formulas...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name for each of the following:...Ch. 11 - Give the IUPAC name (including cis or trans, if...Ch. 11 - Give the LUPAC name (including cis or trans, if...Ch. 11 - Prob. 11.51APPCh. 11 - Prob. 11.52APPCh. 11 - Name each of the following aromatic compounds:...Ch. 11 - Prob. 11.54APPCh. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Draw the cis and trans isomers for each of the...Ch. 11 - Prob. 11.59APPCh. 11 - Draw the line-angle formula for each of the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Write a balanced chemical equation for the...Ch. 11 - Give the name for the product from the...Ch. 11 - Give the name for the product from the...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Draw the condensed structural or line-angle...Ch. 11 - Prob. 11.67CPCh. 11 - Prob. 11.68CPCh. 11 - Prob. 11.69CPCh. 11 - Prob. 11.70CPCh. 11 - Prob. 11.71CPCh. 11 - Prob. 11.72CPCh. 11 - Prob. 11.73CPCh. 11 - Margarines are produced from the hydrogenation of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part II. count the expected number of signals in the 1H-NMR spectrum of these compounds HO 0 одев * Cl -cl "D"arrow_forwardPart I. Create a splitting tree diagram to predict the multiplet pattern of proton Hb in the compound below: 3 (Assume that "Jab >>> ³JbC) Ha Hb He он Ha NH2 Ha HCarrow_forwardNonearrow_forward
- SH 0arrow_forward2. Please consider the two all 'cis' isomers of trimethylcyclohexane drawn below. Draw the two chair conformers of each stereoisomer below (1 and 2) and calculate their torsional interaction energies in order to identify the lower energy conformer for each stereoisomer. Based on your calculations, state which of the two stereoisomers 1 and 2 is less stable and which is more stable. [1,3-diaxial CH3 CH3 = 3.7kcal/mol; 1,3-diaxial CH3 H = 0.88kcal/mol; cis-1,2 (axial:equatorial) CH3 CH3 = 0.88kcal/mol; trans-1,2-diequatorial CH3 CH3 = 0.88kcal/mol) all-cis-1,2,3- 1 all-cis-1,2,4- 2arrow_forwardNonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY