
EP ORGANIC CHEMISTRY,24 MONTH-OWLV2
9th Edition
ISBN: 9781305084391
Author: McMurry
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Question
Chapter 1.10, Problem 14P
Interpretation Introduction
a) The oxygen atom in dimethyl ether, CH3-O-CH3
Interpretation:
The number of nonbonding lone pair of electrons present on oxygen atom in dimethyl ether is to be identified. Further its expected geometry is to be stated.Concept introduction:
The electrons present in the valence shell of an atom that are not involved in bonding with other atoms are called nonbonding or lone pair of electrons. In a molecule, if an atom has only single electrons in all hybridized orbitals then the bonds formed by these orbitals will be equivalent in all respects. The molecule will thus have a regular structure. But if the atom contains single electron as well as unshared pairs of electrons in the hybridized orbitals, the orbitals with unshared pair of electrons will tend to occupy as much space as those orbitals involved in bonding. The bond angles will be slightly different from the expected bond angle and hence the molecule will not have a regular geometry.To determine:
The number of nonbonding lone pair of electrons present on oxygen atom in dimethyl ether and its expected geometry.Interpretation Introduction
b) The nitrogen atom in trimethylamine, CH3-N- [CH3]2
Interpretation:
The number of nonbonding lone pair of electrons present on nitrogen atom in trimethylamine, is to be identified. Further its expected geometry is to be stated.Concept introduction:
The electrons present in the valence shell of an atom that are not involved in bonding with other atoms are called nonbonding or lone pair of electrons. In a molecule, if an atom has only single electrons in all hybridized orbitals then the bonds formed by these orbitals will be equivalent in all respects. The molecule will thus have a regular structure. But if the atom contains single electron as well as unshared pairs of electrons in the hybridized orbitals, the orbitals with unshared pair of electrons will tend to occupy as much space as those orbitals involved in bonding. The bond angles will be slightly different from the expected bond angle and hence the molecule will not have a regular geometry.To determine:
The number of nonbonding lone pair of electrons present on nitrogen atom in trimethylamine and its expected geometry.Interpretation Introduction
c) The phosphorus atom in phosphine, PH3
Interpretation:
The number of nonbonding lone pair of electrons present on phosphorus atom in phosphine is to be identified. Further the expected geometry of phosphorus atom in phosphine is to be stated.Concept introduction:
The electrons present in the valence shell of an atom that are not involved in bonding with other atoms are called nonbonding or lone pair of electrons. In a molecule, if an atom has only single electrons in all hybridized orbitals then the bonds formed by these orbitals will be equivalent in all respects. The molecule will thus have a regular structure. But if the atom contains single electron as well as unshared pairs of electrons in the hybridized orbitals, the orbitals with unshared pair of electrons will tend to occupy as much space as those orbitals involved in bonding. The bond angles will be slightly different from the expected bond angle and hence the geometry of the molecule will be pyramidal.To determine:
The number of nonbonding lone pair of electrons present on phosphorus atom in phosphine and its expected geometry.Answer:
The phosphorus atom in phosphine has one lone pair of electrons. The phosphorus atom is in sp3 hybridized state with one orbital occupied by lone pairs of electrons. Hence the geometry will be pyramid.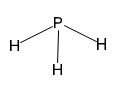
Explanation:
Phosphorus atom has five electrons in its valence shell. It has formed three single bonds with three hydrogen atoms in phosphine. Therefore one lone pair of electrons remains on phosphorus atom. In phosphine the phosphorus atom is in sp3 hybridized state. Three of the sp3 hybrid orbitals containing single electron are utilized for forming three P-H sigma bonds. The fourth sp3 hybrid orbital accommodates the lone pair of electrons and it occupy as much space as a P-H bond does. The H-P-H bond angles deviate slightly from the normal tetrahedral angle. Hence the shape is pyramidal.Conclusion:
The phosphorus atom in phosphine has one lone pair of electrons. The phosphorus atom is in sp3 hybridized state with one orbital occupied by lone pairs of electrons. Hence the structure will be pyramidal.
Interpretation Introduction
d) The sulfur atom in the amino acid methionine
Interpretation:
The number of nonbonding lone pair of electrons present on sulfur atom in the amino acid methionine is to be identified and to state its expected geometry.Concept introduction:
The electrons present in the valence shell of an atom that are not involved in bonding with other atoms are called nonbonding or lone pair of electrons. In a molecule, if an atom has only single electrons in all hybridized orbitals then the bonds formed by these orbitals will be equivalent in all respects. The molecule will thus have a regular structure. But if the atom contains single electron as well as unshared pairs of electrons in the hybridized orbitals, the orbitals with unshared pair of electrons will tend to occupy as much space as those orbitals involved in bonding. The bond angles will be slightly different from the expected bond angle.To determine:
The number of nonbonding lone pair of electrons present on sulfur atom in the amino acid methionine and its expected geometry.Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
30. We will derive the forms of the molecular partition functions for atoms and molecules shortly in
class, but the partition function that describes the translational and rotational motion of a homonuclear
diatomic molecule is given by
Itrans (V,T) =
=
2πmkBT
h²
V
grot (T)
4π²IKBT
h²
Where h is Planck's constant and I is molecular moment of inertia.
The overall partition function is qmolec Qtrans qrot.
Find the energy, enthalpy, entropy, and Helmholtz free energy for the translational and rotational
modes of 1 mole of oxygen molecules and 1 mole of iodine molecules at 50 K and at 300 K and with a
volume of 1 m³. Here is some useful data:
Moment of inertia:
I2 I 7.46 x 10-
45
kg m²
2
O2 I 1.91 x 101
-46
kg m²
K
for each reaction step.
Be sure to account for all bond-breaking and bond-making
steps.
HI
HaC
Drawing Arrows!
H3C
OCH3
H
4 59°F
Mostly sunny
H
CH3
HO
O
CH3
'C'
CH3
Select to Add
Arrows
CH3 1
L
H&C.
OCH3
H H H
H
Select to Add
Arrows
Q Search
Problem 30 of 20
H.
H3C
+
:0:
H
CH3
CH3
20
H2C
Undo
Reset
Done
DELL
Draw the principal organic product of the following reaction.
Chapter 1 Solutions
EP ORGANIC CHEMISTRY,24 MONTH-OWLV2
Ch. 1.3 - Give the ground-state electron configuration for...Ch. 1.3 - How many electrons does each of the following...Ch. 1.4 - Prob. 3PCh. 1.4 - Convert the following representation of ethane,...Ch. 1.4 - What are likely formulas for the following...Ch. 1.4 - Prob. 6PCh. 1.4 - Prob. 7PCh. 1.7 - Draw a line-bond structure for propane, CH3CH2CH3....Ch. 1.7 - Convert the following molecular model of hexane, a...Ch. 1.8 - Draw a line-bond structure for propene, CH3CH=CH2....
Ch. 1.8 - Draw a line-bond structure for 1, 3-butadiene,...Ch. 1.8 - Following is a molecular model of aspirin...Ch. 1.9 - Draw a line-bond structure for propyne, CH3C≡CH....Ch. 1.10 - Prob. 14PCh. 1.12 - Prob. 15PCh. 1.12 - Prob. 16PCh. 1.12 - The following molecular model is a representation...Ch. 1.SE - Convert each of the following molecular models...Ch. 1.SE - The following model is a representation of citric...Ch. 1.SE - The following model is a representation of...Ch. 1.SE - The following model is a representation of...Ch. 1.SE - How many valence electrons does each of the...Ch. 1.SE - Give the ground-state electron configuration for...Ch. 1.SE - Prob. 24APCh. 1.SE - Prob. 25APCh. 1.SE - Draw an electron-dot structure for acetonitrile,...Ch. 1.SE - Draw a line-bond structure for vinyl chloride,...Ch. 1.SE - Fill in any nonbonding valence electrons that are...Ch. 1.SE - Convert the following line-bond structures into...Ch. 1.SE - Convert the following molecular formulas into...Ch. 1.SE - Prob. 31APCh. 1.SE - Oxaloacetic acid, an important intermediate in...Ch. 1.SE - Prob. 33APCh. 1.SE - Potassium methoxide, KOCH3, contains both covalent...Ch. 1.SE - What is the hybridization of each carbon atom in...Ch. 1.SE - Prob. 36APCh. 1.SE - Prob. 37APCh. 1.SE - What bond angles do you expect for each of the...Ch. 1.SE - Propose structures for molecules that meet the...Ch. 1.SE - What kind of hybridization do you expect for each...Ch. 1.SE - Pyridoxal phosphate, a close relative of vitamin...Ch. 1.SE - Prob. 42APCh. 1.SE - Prob. 43APCh. 1.SE - Quetiapine, marketed as Seroquel, is a heavily...Ch. 1.SE - Tell the number of hydrogens bonded to each carbon...Ch. 1.SE - Why do you suppose no one has ever been able to...Ch. 1.SE - Allene, H2C=C=CH2, is somewhat unusual in that it...Ch. 1.SE - Allene (see Problem 1-47) is structurally related...Ch. 1.SE - Complete the electron-dot structure of caffeine,...Ch. 1.SE - Most stable organic species have tetravalent...Ch. 1.SE - A carbanion is a species that contains a...Ch. 1.SE - Divalent carbon species called carbenes are...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - There are two different substances with the...Ch. 1.SE - Prob. 56APCh. 1.SE - Among the most common over-the-counter drugs you...
Knowledge Booster
Similar questions
- Curved arrows are used to illustrate the flow of electrons. Using the provided structures, draw the curved arrows that epict the mechanistic steps for the proton transfer between a hydronium ion and a pi bond. Draw any missing organic structures in the empty boxes. Be sure to account for all lone-pairs and charges as well as bond-breaking and bond-making steps. 2 56°F Mostly cloudy F1 Drawing Arrows > Q Search F2 F3 F4 ▷11 H. H : CI: H + Undo Reset Done DELLarrow_forwardCalculate the chemical shifts in 13C and 1H NMR for 4-chloropropiophenone ? Write structure and label hydrogens and carbons. Draw out the benzene ring structure when doing itarrow_forward1) Calculate the longest and shortest wavelengths in the Lyman and Paschen series. 2) Calculate the ionization energy of He* and L2+ ions in their ground states. 3) Calculate the kinetic energy of the electron emitted upon irradiation of a H-atom in ground state by a 50-nm radiation.arrow_forward
- Calculate the ionization energy of He+ and Li²+ ions in their ground states. Thannnxxxxx sirrr Ahehehehehejh27278283-4;*; shebehebbw $+$;$-;$-28283773838 hahhehdvaarrow_forwardPlleeaasseee solllveeee question 3 andd thankss sirr, don't solve it by AI plleeaasseee don't use AIarrow_forwardCalculate the chemical shifts in 13C and 1H NMR for 4-chloropropiophenone ? Write structure and label hydrogens and carbonsarrow_forward
- 4. Read paragraph 4.15 from your textbook, use your calculated lattice energy values for CuO, CuCO3 and Cu(OH)2 an explain thermal decomposition reaction of malachite: Cu2CO3(OH)2 →2CuO + H2O + CO2 (3 points)arrow_forwardPlease sirrr soollveee these parts pleaseeee and thank youuuuuarrow_forwardIII O Organic Chemistry Using wedges and dashes in skeletal structures Draw a skeletal ("line") structure for each of the molecules below. Be sure your structures show the important difference between the molecules. key O O O O O CHON Cl jiii iiiiiiii You can drag the slider to rotate the molecules. Explanation Check Click and drag to start drawing a structure. Q Search X G ©2025 McGraw Hill LLC. All Rights Reserved. Terms of Use F 3 W C 3/5arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY