
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
14th Edition
ISBN: 9780136873891
Author: Brown
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 11, Problem 6E
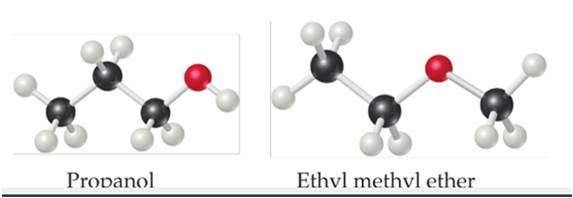
The molecules have the same molecular formula (C3H8O) but different chemical structures.
a. Which molecule(s), if any, can engage in hydrogen?
b. Which molecule do you expect to have a larger dipole moment?
c. One of there molecules has a normal boiling point of 97.2ºC, while the other one has a normal boiling point of 10.8ºC. Assign each molecule to its normal boiling point [Section 11.2□ and 11.5□]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Electrochemistry. Briefly describe the Donnan potential.
Indicate what the Luther equation is used for?
Indicate one aspect that benefits and another that makes it difficult to use the hydroquinone electrode to measure pH.
Chapter 11 Solutions
CHEMISTRY THE CENTRAL SCIENCE >EBOOK<
Ch. 11.2 - Which of the following substances is most likely...Ch. 11.2 - Prob. 11.1.2PECh. 11.3 - Prob. 11.2.1PECh. 11.3 - Prob. 11.2.2PECh. 11.4 - What information about water is needed to...Ch. 11.4 - Prob. 11.3.2PECh. 11.5 -
In the mountains, water in an open container will...Ch. 11.5 - Prob. 11.4.2PECh. 11.6 - Prob. 11.5.1PECh. 11.6 - Prob. 11.5.2PE
Ch. 11.7 - Liquid crystalline phases are produced by which of...Ch. 11.7 - Prob. 11.6.2PECh. 11 - Prob. 1DECh. 11 - Prob. 1ECh. 11 - Prob. 2ECh. 11 - Prob. 3ECh. 11 - If 42.0 kj of heat is added to a 32.0-g sample of...Ch. 11 - Prob. 5ECh. 11 - The molecules have the same molecular formula...Ch. 11 - Prob. 7ECh. 11 - Prob. 8ECh. 11 - Prob. 9ECh. 11 - Prob. 10ECh. 11 - Prob. 11ECh. 11 - Prob. 12ECh. 11 - Prob. 13ECh. 11 - Prob. 14ECh. 11 - Prob. 15ECh. 11 - Prob. 16ECh. 11 - Describe the intermolecular forces that must be...Ch. 11 - Prob. 18ECh. 11 - List the following molecules in order of...Ch. 11 - True or false: a. For molecules with similar...Ch. 11 - Prob. 21ECh. 11 - Prob. 22ECh. 11 - Prob. 23ECh. 11 - Prob. 24ECh. 11 - Prob. 25ECh. 11 - Prob. 26ECh. 11 - Prob. 27ECh. 11 - Prob. 28ECh. 11 - Prob. 29ECh. 11 - Prob. 30ECh. 11 - A number of salts containing the tetrahedral...Ch. 11 - Prob. 32ECh. 11 - a. What is the relationship between surface...Ch. 11 - Prob. 34ECh. 11 - Prob. 35ECh. 11 - Prob. 36ECh. 11 - The boiling points, surface tension, and...Ch. 11 - Prob. 38ECh. 11 - Prob. 39ECh. 11 - Prob. 40ECh. 11 - Prob. 41ECh. 11 - Prob. 42ECh. 11 - 11.43 For many years drinking water has been...Ch. 11 - Prob. 44ECh. 11 - Prob. 45ECh. 11 - The fluorocarbon compound C2 Cl 3£'y has a normal...Ch. 11 - 11.47 Indicate whether each statement is true or...Ch. 11 - Prob. 48ECh. 11 - 11,49 Which of the following affects the vapor...Ch. 11 - Prob. 50ECh. 11 - Prob. 51ECh. 11 - Prob. 52ECh. 11 - Prob. 53ECh. 11 - Prob. 54ECh. 11 - Prob. 55ECh. 11 - Prob. 56ECh. 11 - Prob. 57ECh. 11 - Prob. 58ECh. 11 - Prob. 59ECh. 11 - Prob. 60ECh. 11 - Prob. 61ECh. 11 - Prob. 62ECh. 11 - Prob. 63ECh. 11 - Prob. 64ECh. 11 - In terms of the arrangement and freedom of motion...Ch. 11 - Prob. 66ECh. 11 - Prob. 67ECh. 11 - Prob. 68ECh. 11 - Prob. 69ECh. 11 - Prob. 70ECh. 11 - Prob. 71ECh. 11 - Prob. 72ECh. 11 - Prob. 73AECh. 11 - Prob. 74AECh. 11 - Prob. 75AECh. 11 - Prob. 76AECh. 11 - Prob. 77AECh. 11 - The table below shows the normal boiling points of...Ch. 11 - Prob. 79AECh. 11 - Prob. 80AECh. 11 - Prob. 81AECh. 11 - Prob. 82AECh. 11 - Prob. 83AECh. 11 - Prob. 84AECh. 11 - Prob. 85AECh. 11 - Prob. 86AECh. 11 - Prob. 87AECh. 11 - Prob. 88AECh. 11 - Prob. 89AECh. 11 - Prob. 90IECh. 11 - Prob. 91IECh. 11 - Prob. 92IECh. 11 - 11.93 The vapor pressure of ethanol (C2H5OH) at 19...Ch. 11 - Prob. 94IECh. 11 - Prob. 95IECh. 11 - Prob. 96IECh. 11 - Prob. 97IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- At an electrified interface according to the Gouy-Chapman model, what types of interactions do NOT occur between the ions and the solvent according to this theory?arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. Hint: In this case you must choose the best answer to demonstrate the stereochemistry of H2 addition. 1.03 2. (CH3)2S BIZ CH₂OH 2. DMS KMnO4, NaOH ΖΗ Pd or Pt (catalyst) HBr 20 1 HBr ROOR (peroxide) HO H-SO HC 12 11 10 BH, THE 2. H2O2, NaOH Brz cold HI 19 18 17 16 MCPBA 15 14 13 A Br H₂O BH3⚫THF Brz EtOH Pd or Ni (catalyst) D₂ (deuterium) 1. Os04 2. H2O2 CH3CO3H (peroxyacid) 1. MCPBA 2. H₂O* H B + H H H "H C H H Darrow_forwardExplain how Beer’s Law can be used to determine the concentration in a selected food sample. Provide examples.arrow_forward
- Explain the importance of having a sampling plan with respect to food analysis. Explain the importance of having a sampling plan with respect to food analysis. Provide examples.arrow_forwardPlease predict the products for each of the following reactions. Clearly show the regiochemistry (Markovnikov vs anti-Markovnikov) and stereochemistry (syn- vs anti- or both). If a mixture of enantiomers is formed, please draw all the enantiomers. cold KMnO4, NaOH 2. DMS 1. 03 CH3OH Br2 1. 03 2. (CH3)2S H₂ Pd or Pt (catalyst) HBr 18 19 20 1 HBr ROOR (peroxide) H₂O H₂SO4 HCI HI 17 16 6 15 MCPBA 1. BH3 THF 2. H₂O2, NaOH 1. OsO4 2. H₂O₂ 110 CH3CO₂H (peroxyacid) 1. MCPBA 2. H₂O* Br2 H₂O BH3 THF B12 EtOH Pd or Ni (catalyst) D₂ (deuterium) Bra A B C D H OH H OH OH H OH α α α OH H OH OH фон d H "Harrow_forwardBriefly indicate the models that describe the structure of the interface: Helmholtz-Perrin, Gouy-Chapman, Stern and Grahame models.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Liquids: Crash Course Chemistry #26; Author: Crash Course;https://www.youtube.com/watch?v=BqQJPCdmIp8;License: Standard YouTube License, CC-BY
Chemistry of Group 16 elements; Author: Ch-11 Chemical Engg, Chemistry and others;https://www.youtube.com/watch?v=5B1F0aDgL6s;License: Standard YouTube License, CC-BY