
Concept explainers
(a)
Interpretation:
The order of decreasing bond energy of
Concept introduction:
A molecular orbital diagram is a tool that is used to describe the
(a)
Answer to Problem 11.37P
The order of decreasing bond energy of
Explanation of Solution
The molecular orbital diagram of
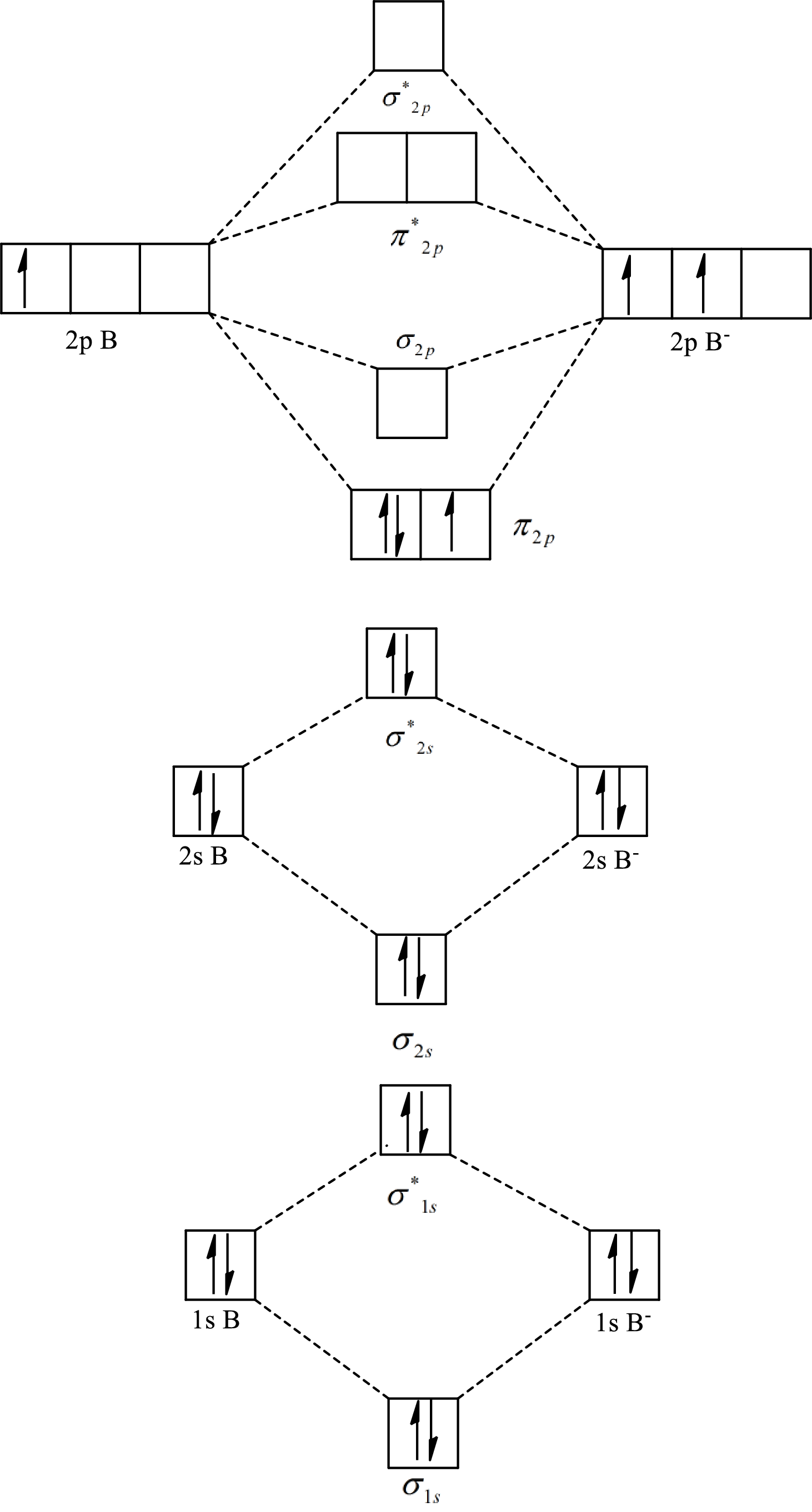
The formula to calculate the bond order is as follows:
Substitute 7 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
The molecular orbital diagram of
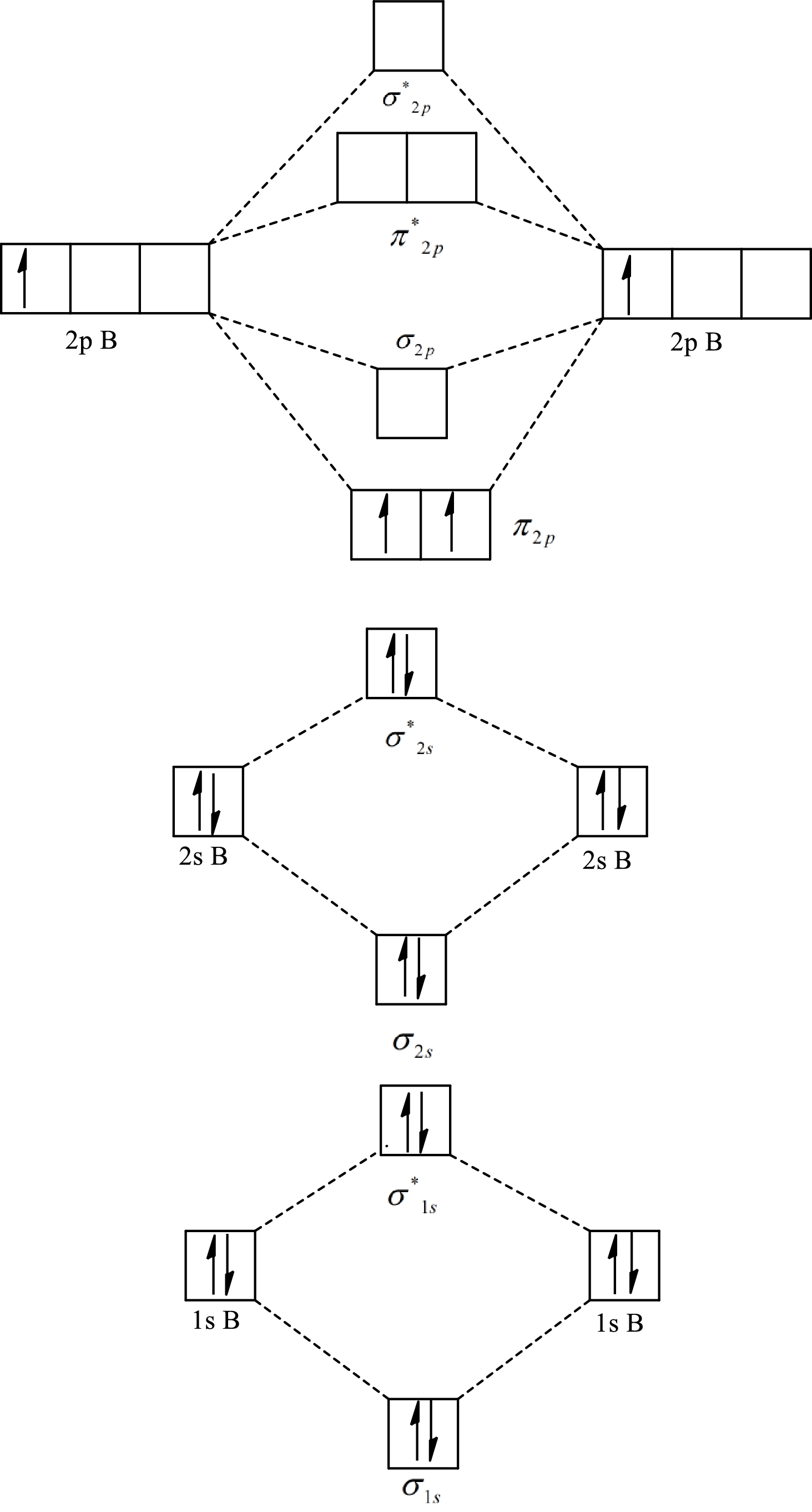
Substitute 6 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
The molecular orbital diagram of
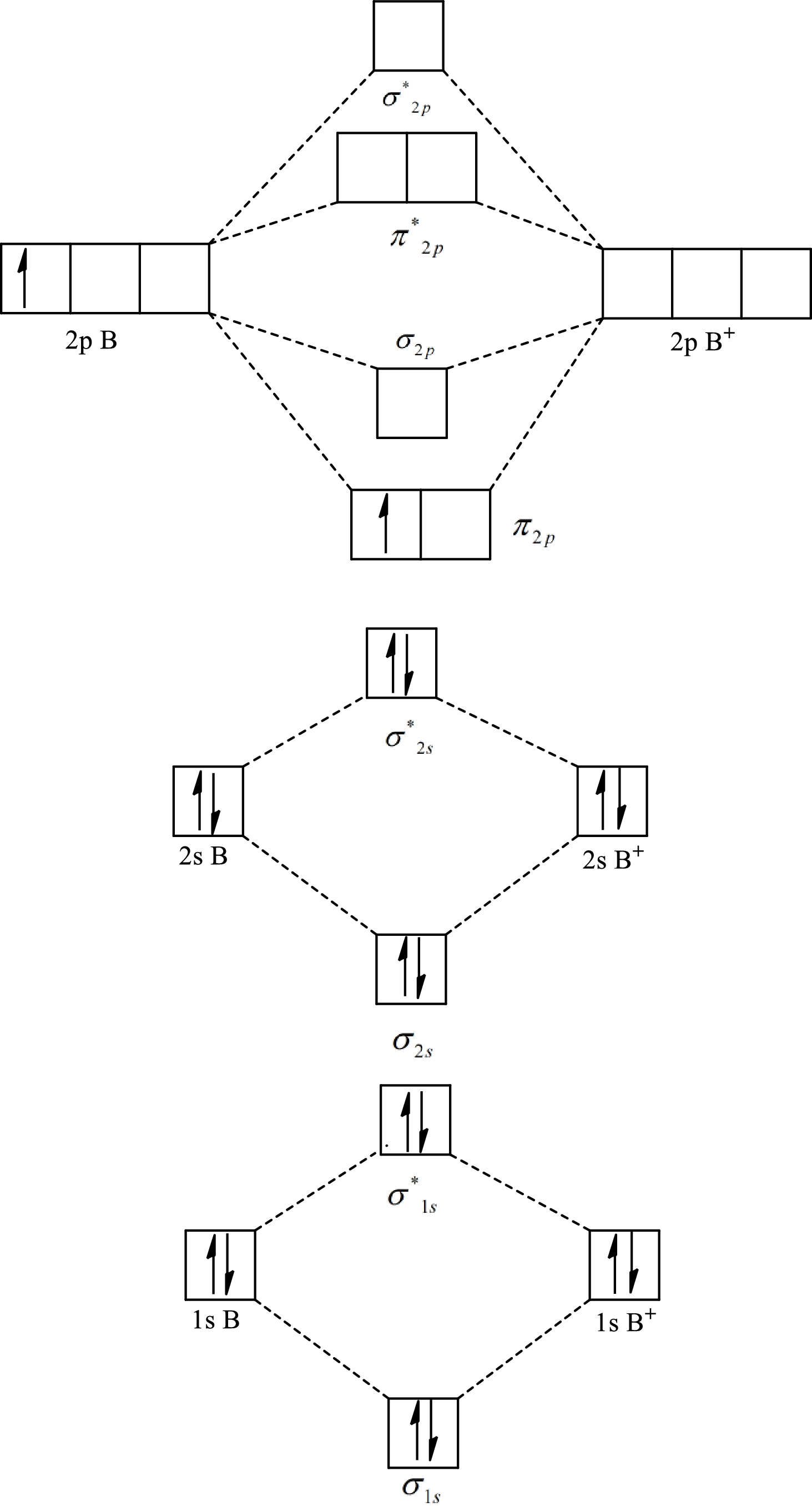
Substitute 5 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
Bond energy is directly proportional to the bond order. The highest bond order of
So the order of decreasing bond energy is
The order of decreasing bond energy of
(b)
Interpretation:
The order of decreasing bond length of
Concept introduction:
A molecular orbital diagram is a tool that is used to describe the chemical bonding formed between different molecules. It is used to predict the bond strength and the electronic transitions that a molecule can undergo.
(b)
Answer to Problem 11.37P
The order of the decreasing bond length is
Explanation of Solution
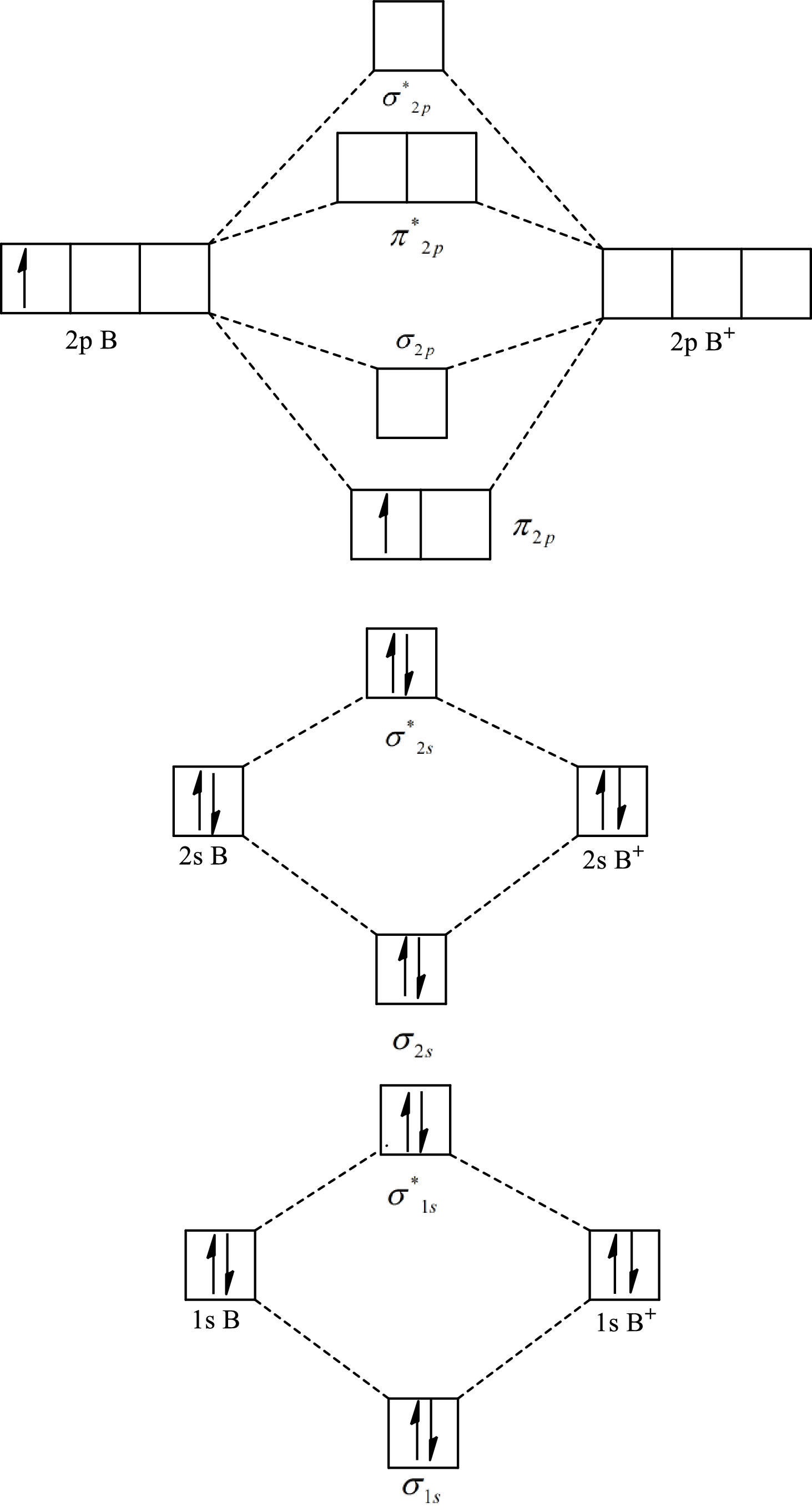
The molecular orbital diagram of
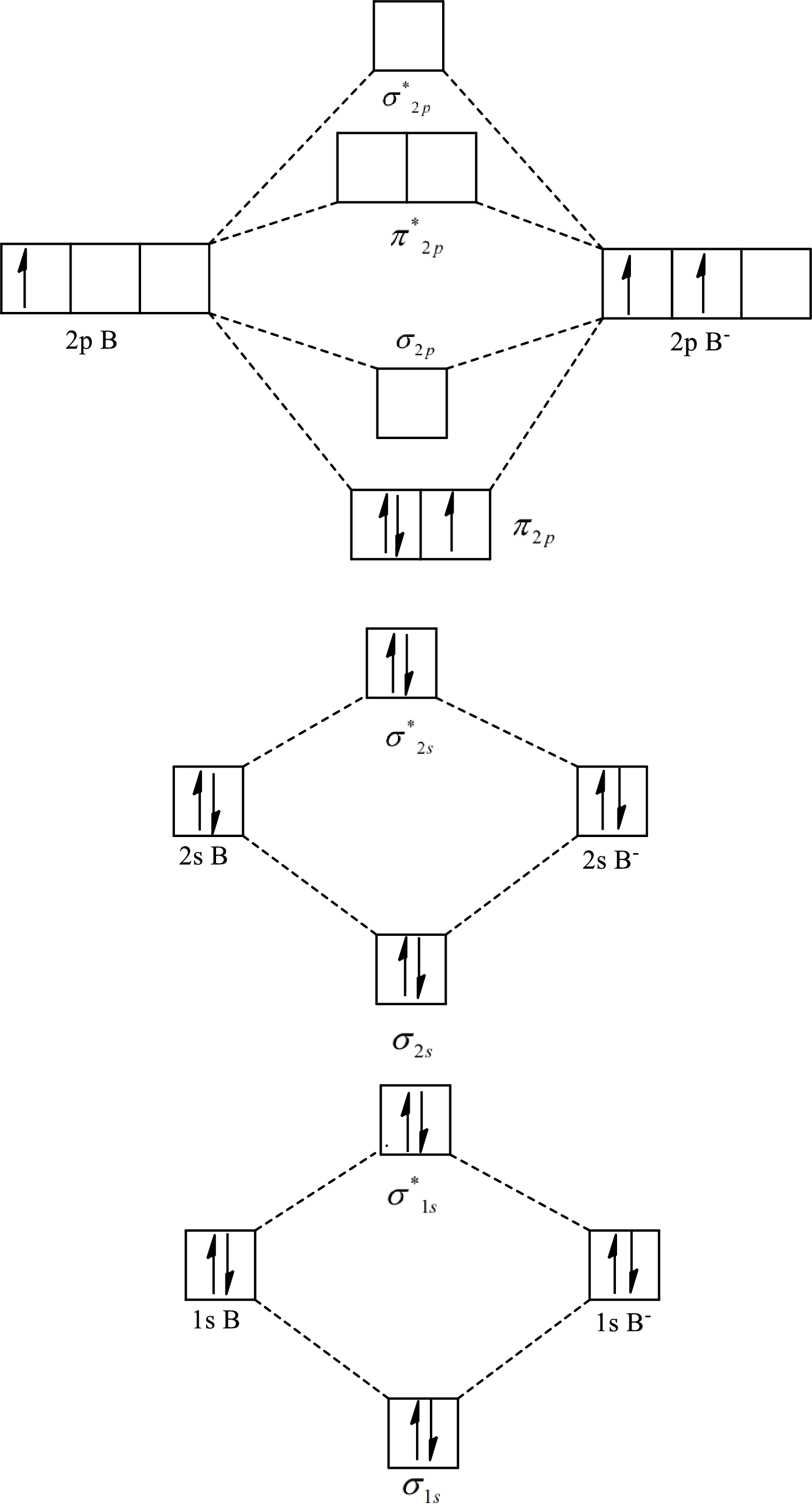
The formula to calculate the bond order is as follows:
Substitute 7 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
The molecular orbital diagram of
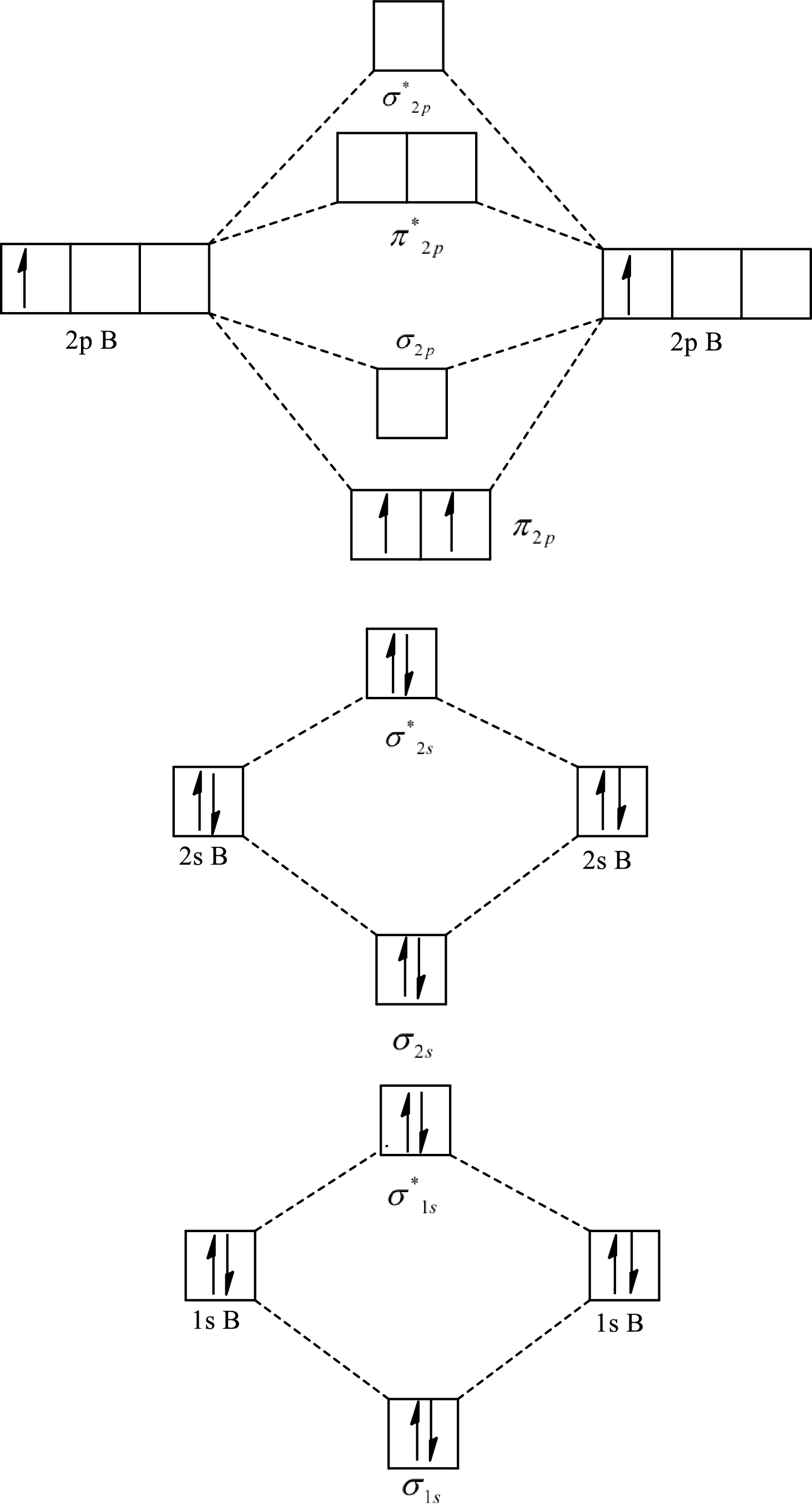
Substitute 6 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
The molecular orbital diagram of
Substitute 5 for the number of electrons in bonding orbitals and 4 for the number of electrons in antibonding orbitals in equation(1).
Bond length decreases with the increase in bond energy. The highest bond order of
So the order of the decreasing bond length is
The order of the decreasing bond length is
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry: The Molecular Nature of Matter and Change - Standalone book
- Complete the mechanismarrow_forwardV Biological Macromolecules Drawing the Haworth projection of an aldose from its Fischer projection Draw a Haworth projection of a common cyclic form of this monosaccharide: H C=O HO H HO H H OH CH₂OH Explanation Check Click and drag to start drawing a structure. Xarrow_forwardComplete the mechanismarrow_forward
- Complete the mechanismarrow_forward8 00 6 = 10 10 Decide whether each of the molecules in the table below is stable, in the exact form in which it is drawn, at pH = 11. If you decide at least one molecule is not stable, then redraw one of the unstable molecules in its stable form below the table. (If more than unstable, you can pick any of them to redraw.) Check OH stable HO stable Ounstable unstable O OH stable unstable OH 80 F6 F5 stable Ounstable X Save For Later Sub 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy C ཀྭ་ A F7 매 F8 F9 4 F10arrow_forwardJust try completing it and it should be straightforward according to the professor and TAs.arrow_forward
- The grading is not on correctness, so if you can just get to the correct answers without perfectionism that would be great. They care about the steps and reasoning and that you did something. I asked for an extension, but was denied the extension.arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers. Something that looks reasonable or correct would be sufficient. If you can get many of them correct that would be great!arrow_forwardShow your work and do something that is reasonable. It does not have to be 100% correct. Just show something that looks good or pretty good as acceptable answers. Something that looks reasonable or correct would be sufficient. If you can get many of them correct that would be great!arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





