
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
7th Edition
ISBN: 9780134240152
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10, Problem 61P
cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they react with HO–.
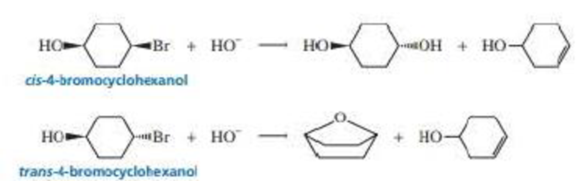
a. Why do they form the same elimination product?
h. Explain, by showing the mechanisms, why different substitution products are obtained.
c. How many stereoisomers does each or the elimination and substitution reactions form?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculating the pH at equivalence of a titration
3/5
Izabella
A chemist titrates 120.0 mL of a 0.7191M dimethylamine ((CH3)2NH) solution with 0.5501 M HBr solution at 25 °C. Calculate the pH at equivalence. The
pk of dimethylamine is 3.27.
Round your answer to 2 decimal places.
Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HBr solution added.
pH = ☐
✓
18
Ar
Bo
Alcohols can be synthesized using an acid-catalyzed hydration of an alkene. An alkene is combined with aqueous acid (e.. sulfuric acid in water). The
reaction mechanism typically involves a carbocation intermediate.
>
3rd attempt
3343
10
8
Draw arrows to show the reaction between the alkene and hydronium ion.
that
2nd attempt
Feedback
1st attempt
تعمال
Ju See Periodic Table See Hint
F
D
Ju See Periodic Table See Hint
Draw the simplified curved arrow mechanism for the reaction of acetone and CHgLi to give the major product.
4th attempt
Π
Draw the simplified curved arrow mechanism
T
3rd attempt
Feedback
Ju
See Periodic Table See Hint
H
-H
H
-I
H
F
See Periodic Table See Hint
Chapter 10 Solutions
Organic Chemistry; Modified MasteringChemistry with Pearson eText -- ValuePack Access Card; Study Guide and Student Solutions Manual for Organic Chemistry, Books a la Carte Edition (7th Edition)
Ch. 10.2 - Prob. 1PCh. 10.2 - Prob. 2PCh. 10.2 - Prob. 3PCh. 10.2 - Prob. 4PCh. 10.3 - Four alkenes are formed from the E1 reaction of...Ch. 10.3 - If 2-fluoropentane could undergo an E1 reaction,...Ch. 10.3 - Prob. 7PCh. 10.3 - Propose a mechanism for the following reaction:Ch. 10.4 - Prob. 9PCh. 10.4 - What products will be obtained from the El...
Ch. 10.4 - Prob. 11PCh. 10.5 - Prob. 12PCh. 10.6 - Prob. 14PCh. 10.7 - Why do cis-1-bromo-2-ethylcyclohexane and...Ch. 10.7 - Which isomer reacts more rapidly in an E2...Ch. 10.7 - Prob. 18PCh. 10.8 - Prob. 19PCh. 10.8 - Prob. 20PCh. 10.9 - Prob. 21PCh. 10.9 - Explain why only a substitution product and no...Ch. 10.9 - Prob. 23PCh. 10.9 - Prob. 24PCh. 10.9 - Prob. 25PCh. 10.9 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 10.10 - A small amount of another organic product is...Ch. 10.10 - What is the best way to prepare the following...Ch. 10.10 - Prob. 29PCh. 10.10 - Prob. 30PCh. 10.10 - Why is a cumulated diene not formed in the...Ch. 10.10 - What product is obtained when the following...Ch. 10.11 - Prob. 33PCh. 10.11 - Prob. 34PCh. 10 - Draw the major product obtained when each of the...Ch. 10 - Prob. 36PCh. 10 - a. Indicate how each of the following factors...Ch. 10 - Prob. 38PCh. 10 - A chemist wanted to synthesize the...Ch. 10 - Prob. 40PCh. 10 - Prob. 41PCh. 10 - Prob. 42PCh. 10 - Starting with an alkyl halide, how could the...Ch. 10 - Indicate which species in each pair gives a higher...Ch. 10 - Prob. 45PCh. 10 - For each of the following alkyl halides, indicate...Ch. 10 - Prob. 47PCh. 10 - When 2-bromo-2,3-dimethylbutane reacts with a...Ch. 10 - Prob. 49PCh. 10 - When the following compound undergoes solvolysis...Ch. 10 - cis-1-Bromo-4-tert-butylcyclohexane and...Ch. 10 - Draw the substitution and elimination products.Ch. 10 - Prob. 53PCh. 10 - Prob. 54PCh. 10 - Which of the following hexachlorocyclohexanes is...Ch. 10 - Explain why the rate of the reaction of...Ch. 10 - Prob. 57PCh. 10 - Two elimination products are obtained from the...Ch. 10 - Draw the structures or the product of the obtained...Ch. 10 - How could you prepare the following compounds from...Ch. 10 - cis-4-Bromocyclohexanol and...Ch. 10 - Prob. 62PCh. 10 - Prob. 63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Select the correct reagent to accomplish the first step of this reaction. Then draw a mechanism on the Grignard reagent using curved arrow notation to show how it is converted to the final product. 4th attempt Part 1 (0.5 point) Select the correct reagent to accomplish the first step of this reaction. Choose one: OA Mg in ethanol (EtOH) OB. 2 Li in THF O C. Li in THF D. Mg in THF O E Mg in H2O Part 2 (0.5 point) Br Part 1 Bri Mg CH B CH, 1 Draw intermediate here, but no arrows. © TE See Periodic Table See Hint See Hint ין Harrow_forwardSelect the product for the following reaction. HO HO PCC OH ○ OH O HO ○ HO HO HOarrow_forward5:45 Х Select the final product for the following reaction sequence. O O 1. Mg. ether 2.D.Oarrow_forward
- Based on the chart Two similarities between the molecule with alpha glycosidic linkages. Two similarities between the molecules with beta glycosidtic linkages. Two differences between the alpha and beta glycosidic linkages.arrow_forwardplease help fill in the tablearrow_forwardAnswer F pleasearrow_forward
- 4. Refer to the data below to answer the following questions: The octapeptide saralasin is a specific antagonist of angiotensin II. A derivative of saralasin is used therapeutically as an antihypertensive. Amino acid analysis of saralasin show the presence of the following amino acids: Ala, Arg, His, Pro, Sar, Tyr, Val, Val A.Sar is the abbreviation for sarcosine, N-methyl aminoethanoic acid. Draw the structure of sarcosine. B. N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Tyr-Val-His Sar-Arg-Val His-Pro-Ala Val-Tyr-Val Arg-Val-Tyr What is the structure of saralasin?arrow_forwardWhat is the structure of the DNA backbone?arrow_forwardPLEASE PLEASE PLEASE use hand drawn structures when possarrow_forward
- . M 1- MATCH each of the following terms to a structure from the list below. There is only one correct structure for each term and structures may be used more than once. Place the letter of the structure in the blank to the left of the corresponding term. A. Sanger dideoxy method C. Watson-Crick B. GAUCGUAAA D. translation E. HOH2C OH OH G. transcription I. AUGGCUGAG 0 K. OPOH2C 0- OH N- H NH2 F. -OPOH2C 0- OH OH H. Maxam-Gilbert method J. replication N L. HOH2C a. b. C. d. e. f. g. B M. AGATCGCTC a pyrimidine nucleoside RNA base sequence with guanine at the 3' end. DNA base sequence with cytosine at the 3' end. a purine nucleoside DNA sequencing method for the human genome 2'-deoxyadenosine 5'-phosphate process by which mRNA directs protein synthesis OH NH2arrow_forwardPlease use hand drawn structures when neededarrow_forwardB. Classify the following amino acid. Atoms other than carbon and hydrogen are labeled. a. acidic b. basic C. neutral C. Consider the following image. Which level of protein structure is shown here? a. primary b. secondary c. tertiary d. quaternary D. Consider the following image. H RH H HR H R HR HR RH Which level of protein structure is shown in the box? a. primary b. secondary R c. tertiary d. quaternary コー Rarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY