
EP ESSENTIAL ORG.CHEM.-MOD.MASTERING
3rd Edition
ISBN: 9780133858501
Author: Bruice
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 52P
- a. Which of the indicated bonds in each compound is shorter?
- b. Indicate the hybridization of the C, O, N, and halogen atoms in each of the compounds.
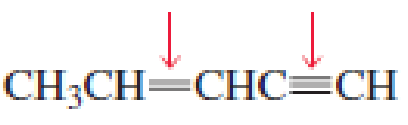
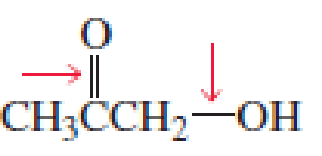
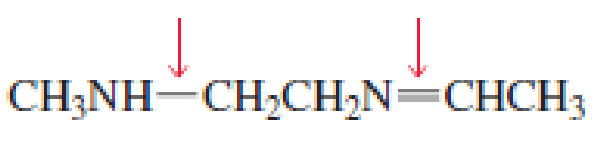
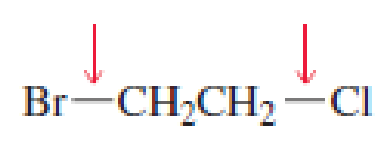
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
can someone answer the questions and draw out the complete mechanism
Please help, draw and me the proper mechanisms.
Please help on part 2 not part 3. Please write in the proper reagents
Chapter 1 Solutions
EP ESSENTIAL ORG.CHEM.-MOD.MASTERING
Ch. 1.1 - Oxygen has three isotopes, 16O, 17O, and 18O. The...Ch. 1.2 - Prob. 2PCh. 1.2 - How many valence electrons do chlorine, bromine,...Ch. 1.2 - Look at the relative positions of each pair of...Ch. 1.3 - a. Find potassium (K) in the periodic table and...Ch. 1.3 - Which bond is more polar?Ch. 1.3 - Which of the following has a. the most polar bond?...Ch. 1.3 - Use the symbols + and to show the direction of...Ch. 1.3 - After examining the potential maps for LiH, HF,...Ch. 1.4 - An atom with a formal charge does not necessarily...
Ch. 1.4 - Prob. 12PCh. 1.4 - a. Draw two Lewis structures for C2H6O. b. Draw...Ch. 1.4 - Draw the lone-pair electrons that are not shown in...Ch. 1.4 - Prob. 16PCh. 1.4 - Which of the atoms in the molecular models in...Ch. 1.4 - Prob. 18PCh. 1.7 - What orbitals are used to form the 10 sigma bonds...Ch. 1.9 - Put a number in each of the blanks: a. ___ s...Ch. 1.11 - Predict the approximate bond angles in a. the...Ch. 1.11 - According to the potential map for the ammonium...Ch. 1.12 - Prob. 25PCh. 1.13 - a. Predict the relative lengths and strengths of...Ch. 1.13 - Prob. 28PCh. 1.14 - Which of the bonds in a carbonoxygen double bond...Ch. 1.14 - Caffeine is a natural insecticide, found in the...Ch. 1.14 - a. What is the hybridization of each of the carbon...Ch. 1.14 - Prob. 33PCh. 1.14 - Describe the orbitals used in bonding and the bond...Ch. 1.15 - Account for the difference in the shape and color...Ch. 1.15 - Which of the following molecules would you expect...Ch. 1 - Draw a Lewis structure for each of the following...Ch. 1 - Prob. 38PCh. 1 - What is the hybridization of all the atoms (other...Ch. 1 - Prob. 40PCh. 1 - Draw the condensed structure of a compound that...Ch. 1 - Prob. 42PCh. 1 - Prob. 43PCh. 1 - Draw a Lewis structure for each of the following...Ch. 1 - Prob. 45PCh. 1 - List the bonds in order from most polar to least...Ch. 1 - What is the hybridization of the indicated atom in...Ch. 1 - Write the Kekul structure for each of the...Ch. 1 - Assign the missing formal charges.Ch. 1 - Predict the approximate bond angles for the...Ch. 1 - Prob. 51PCh. 1 - a. Which of the indicated bonds in each compound...Ch. 1 - In which orbitals are the lone pairs in nicotine?...Ch. 1 - Draw the missing lone-pair electrons and assign...Ch. 1 - Rank the following compounds from highest dipole...Ch. 1 - Prob. 56PCh. 1 - a. Which of the species have bond angles of 109.5?...Ch. 1 - Prob. 58PCh. 1 - Sodium methoxide (CH3ONa) has both ionic and...Ch. 1 - a. Why is a H 8 H bond (0.74 ) shorter than a C 8...Ch. 1 - Which compound has a larger dipole moment, CHCl3...Ch. 1 - Which compound has a longer C 8 Cl bond?Ch. 1 - Prob. 63PCh. 1 - The following compound has two isomers. One isomer...
Additional Science Textbook Solutions
Find more solutions based on key concepts
An electric motor has an effective resistance of 32.0 and an inductive reactance of 45.0 when working under l...
Fundamentals of Physics Extended
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
Describe the evolution of mammals, tracing their synapsid lineage from early amniote ancestors to true mammals....
Loose Leaf For Integrated Principles Of Zoology
Describe the role and impact of microbes on the earth.
Microbiology Fundamentals: A Clinical Approach
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY