
Chemistry for Engineering Students
4th Edition
ISBN: 9781337398909
Author: Lawrence S. Brown, Tom Holme
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.83PAE
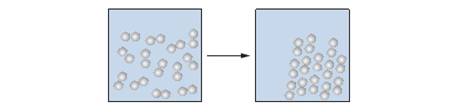
1.83 What type of transition is represented in the following molecular-scale illustration?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the following two half-reactions, determine the pH range in which $NO_2^-\ (aq)$ cannot be found as the predominant chemical species in water.* $NO_3^-(aq)+10H^+(aq)+8e^-\rightarrow NH_4^+(aq)+3H_2O(l),\ pE^{\circ}=14.88$* $NO_2^-(aq)+8H^+(aq)+6e^-\rightarrow NH_4^+(aq)+2H_2O(l),\ pE^{\circ}=15.08$
Indicate characteristics of oxodec acid.
What is the final product when hexanedioic acid reacts with 1º PCl5 and 2º NH3.
Chapter 1 Solutions
Chemistry for Engineering Students
Ch. 1 - Prob. 1COCh. 1 - Prob. 2COCh. 1 - Draw pictures to illustrate simple chemical...Ch. 1 - Explain the difference between inductive and...Ch. 1 - Use appropriate techniques to convert measurements...Ch. 1 - Express the results of calculations using the...Ch. 1 - What are the components involved?Ch. 1 - How do those components interact or connect to...Ch. 1 - What is the ultimate function of the whole system?Ch. 1 - Prob. 1.1PAE
Ch. 1 - In what country is most of the world’s cobalt...Ch. 1 - In what types of technology do the elements...Ch. 1 - Based on the information in Figure 1.1, which...Ch. 1 - Prob. 1.5PAECh. 1 - Prob. 1.6PAECh. 1 - When we make observations in the laboratory, which...Ch. 1 - Which of the following items are matter and which...Ch. 1 - Which macroscopic characteristics differentiate...Ch. 1 - 1.10 Do the terms element and atom mean the same...Ch. 1 - 1.11 Label each of the following as either a...Ch. 1 - 1.12 Why do physical properties play a role in...Ch. 1 - 1.13 Physical properties may change because of a...Ch. 1 - 1.14 Which part of the following descriptions of a...Ch. 1 - 1.15 We used the example of attendance at a...Ch. 1 - 1.16 Complete the following statement: Data that...Ch. 1 - 1.17 Complete the following statement: Data that...Ch. 1 - 1.18 Two golfers are practicing shots around a...Ch. 1 - Prob. 1.19PAECh. 1 - 1.20 Suppose that you are waiting at a corner for...Ch. 1 - 1.21 When a scientist looks at an experiment and...Ch. 1 - 1.22 What is the difference between a hypothesis...Ch. 1 - 1.23 Should the words theory and model be used...Ch. 1 - 1.24 What is a law of nature? Are all scientific...Ch. 1 - 1.25 Describe a miscommunication that can arise...Ch. 1 - 1.26 What is the difference between a qualitative...Ch. 1 - 1.27 Identify which of the following units are...Ch. 1 - 1.28 What is a “derived” unit?Ch. 1 - 1.29 Rank the following prefixes in order of...Ch. 1 - 1.30 The largest computers now include disk...Ch. 1 - Prob. 1.31PAECh. 1 - 1.32 Use the web to determine how the Btu was...Ch. 1 - 1.33 How many micrograms are equal to one gram?Ch. 1 - 1.34 Convert the value 0.120 ppb into ppm.Ch. 1 - 1.35 How was the Fahrenheit temperature scale...Ch. 1 - Superconductors are materials that have no...Ch. 1 - 1.37 Express each of the following temperatures in...Ch. 1 - 1.38 Express (a) 275 oC in K, (b) 25.55 K in oC,...Ch. 1 - 1.39 Express each of the following numbers in...Ch. 1 - 1.40 How many significant figures are there in...Ch. 1 - 1.41 How many significant figures are present in...Ch. 1 - Perform these calculations and express the result...Ch. 1 - 1.43 Calculate the following to the correct number...Ch. 1 - 1.44 In an attempt to determine the velocity of a...Ch. 1 - 1.45 A student finds that the mass of an object is...Ch. 1 - 1.46 Measurements indicate that 23.6% of the...Ch. 1 - 1.47 A student weighs 10 quarters and finds that...Ch. 1 - 1.48 A rock is placed on a balance and its mass is...Ch. 1 - 1.49 A package of eight apples has a mass of 1.00...Ch. 1 - Prob. 1.50PAECh. 1 - 1.51 A person measures 173 cm in height. What is...Ch. 1 - 1.52 The distance between two atoms in a molecule...Ch. 1 - 1.53 Carry out the following unit conversions. (a)...Ch. 1 - 1.54 Carry out each of the following conversions....Ch. 1 - 1.55 Convert 22.3 mL to (a) liters, (b) cubic...Ch. 1 - 1.56 If a vehicle is travelling 92 m/s, what is...Ch. 1 - 1.57 A load of asphalt weights 245 lb. and...Ch. 1 - 1.58 One square mile contains exactly 640 acres....Ch. 1 - 1.59 A sample of crude oil has a density of 0.87...Ch. 1 - 1.60 Mercury has a density of 13.6 g/mL. What is...Ch. 1 - 1.61 The area of the 48 contiguous states is...Ch. 1 - 1.62 The dimensions of aluminium foil in a box for...Ch. 1 - Prob. 1.63PAECh. 1 - 1.64 Wire is often sold in pound spools according...Ch. 1 - 1.65 An industrial engineer is designing a process...Ch. 1 - 1.66 An engineer is working with archaeologists to...Ch. 1 - 1.67 On average, Earth’s crust contains about 8.1...Ch. 1 - Prob. 1.68PAECh. 1 - 1.69 The “Western Stone” in Jerusalem is one of...Ch. 1 - A load of bauxite has a density of 3.15 g/cm3. If...Ch. 1 - 1.71 Is touch screen technology better described...Ch. 1 - Prob. 1.72PAECh. 1 - 1.73 Why are two separate ITO layers required in a...Ch. 1 - 1.74 What are the two properties of ITO that make...Ch. 1 - 1.75 What does it mean that ITO films are made by...Ch. 1 - 1.76 How does Gorilla Glass differ from more...Ch. 1 - 1.77 How can a liquid be distinguished from a fine...Ch. 1 - 1.78 Some farmers use ammonia, NH3, as a...Ch. 1 - 1.79 Use a molecular-level description to explain...Ch. 1 - 1.80 All molecules attract each other to some...Ch. 1 - 1.81 Draw a molecular-scale picture to show how a...Ch. 1 - 1.82 Which of the following molecular-scale...Ch. 1 - 1.83 What type of transition is represented in the...Ch. 1 - 1.84 A student was given two metal cubes that...Ch. 1 - 1.85 Battery acid has a density of 1.285 g/mL and...Ch. 1 - 1.86 Unfermented grape juice used to make wine is...Ch. 1 - 1.87 A solution of ethanol in water has a volume...Ch. 1 - 1.88 Legend has it that Archimedes, a famous...Ch. 1 - 1.89 Imagine that you place a cork measuring...Ch. 1 - 1.90 A calibrated flask was filled to the 25.00-mL...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the final product when D-galactose reacts with hydroxylamine?arrow_forwardIndicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.arrow_forwardIn the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?arrow_forward
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY