Ok what about this one? I will also put the answer I got in there so you can check if I'm correct or not, or if I need to write it another way.
Ok what about this one? I will also put the answer I got in there so you can check if I'm correct or not, or if I need to write it another way.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
100%
Ok what about this one? I will also put the answer I got in there so you can check if I'm correct or not, or if I need to write it another way.

Transcribed Image Text:### Chemical Structure of 2-phenylethan-2-ol
#### Description:
The image above depicts the chemical structure of 2-phenylethan-2-ol, which is a type of alcohol.
#### Structural Details:
- **Benzene Ring**: The left part of the structure is a benzene ring, which consists of six carbon atoms arranged in a hexagonal shape with alternating double bonds.
- **Hydroxyl Group (OH)**: Attached to the second carbon atom in the ethyl chain from the benzene ring is a hydroxyl group, represented by "OH". This defines the compound as an alcohol.
- **Ethyl Chain**: The linear ethyl chain is connected to the benzene ring and contains the hydroxyl group.
#### Nomenclature:
The compound is named "2-phenylethan-2-ol," indicating a two-carbon chain (ethan) with an alcohol group at the second carbon position, and a phenyl group (benzene ring) attached to the first carbon.
This compound is known for its fragrance and is used in various perfumes and flavorings.
![**Spectral Analysis of a Pure Compound**
**Information:**
Each spectrum below was obtained from a pure compound.
- *Mass Spectrum*: Parent peaks (M) are listed for all examples.
- *Infrared (IR) Spectroscopy*: Peaks listed are strong (s) unless otherwise noted for signals above 1500 cm⁻¹.
- *¹H NMR Spectra*: The integral is given in the number of hydrogens (1H) or as a relative ratio. Important coupling constants (J-values) are indicated next to the peaks for some examples. For specific spectra, an inset (grey box) shows a “zoom-in” on a key part of the spectrum.
**Mass Spectrometry (not shown):** [M] = 122 m/z
**Infrared Spectroscopy (not shown):** 3364, 3030, 2973, 1493, 1451, 1408, 1178 cm⁻¹
**¹H Nuclear Magnetic Resonance:**
- A spectrum with a range from 0 to 8 PPM.
- Peaks are labeled as follows:
- At approximately 7.1 PPM, labeled "m" corresponding to 5H.
- At approximately 4.1 PPM, labeled "q" corresponding to 1H.
- At approximately 3.5 PPM, labeled "s" corresponding to 1H.
- At approximately 1.3 PPM, labeled "d" corresponding to 3H.
**¹³C Nuclear Magnetic Resonance:**
- A spectrum with a range from 0 to 160 PPM. It contains several distinct peaks.
This educational page provides insights into how various spectral data can be used to study pure compounds, offering foundational knowledge in structural analysis techniques such as Mass Spectrometry, Infrared Spectroscopy, and Nuclear Magnetic Resonance (NMR).](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fb5f1630e-c465-44f2-a868-b429d99d07e1%2F35fc05f6-0da3-4d10-99e1-a424580a5c56%2Fd409hp_processed.png&w=3840&q=75)
Transcribed Image Text:**Spectral Analysis of a Pure Compound**
**Information:**
Each spectrum below was obtained from a pure compound.
- *Mass Spectrum*: Parent peaks (M) are listed for all examples.
- *Infrared (IR) Spectroscopy*: Peaks listed are strong (s) unless otherwise noted for signals above 1500 cm⁻¹.
- *¹H NMR Spectra*: The integral is given in the number of hydrogens (1H) or as a relative ratio. Important coupling constants (J-values) are indicated next to the peaks for some examples. For specific spectra, an inset (grey box) shows a “zoom-in” on a key part of the spectrum.
**Mass Spectrometry (not shown):** [M] = 122 m/z
**Infrared Spectroscopy (not shown):** 3364, 3030, 2973, 1493, 1451, 1408, 1178 cm⁻¹
**¹H Nuclear Magnetic Resonance:**
- A spectrum with a range from 0 to 8 PPM.
- Peaks are labeled as follows:
- At approximately 7.1 PPM, labeled "m" corresponding to 5H.
- At approximately 4.1 PPM, labeled "q" corresponding to 1H.
- At approximately 3.5 PPM, labeled "s" corresponding to 1H.
- At approximately 1.3 PPM, labeled "d" corresponding to 3H.
**¹³C Nuclear Magnetic Resonance:**
- A spectrum with a range from 0 to 160 PPM. It contains several distinct peaks.
This educational page provides insights into how various spectral data can be used to study pure compounds, offering foundational knowledge in structural analysis techniques such as Mass Spectrometry, Infrared Spectroscopy, and Nuclear Magnetic Resonance (NMR).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
what about this?
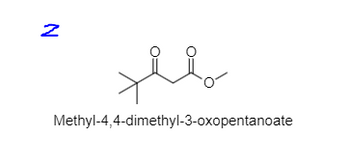
Transcribed Image Text:N
مهلا
Methyl-4,4-dimethyl-3-oxopentanoate
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY