lon Practice Set 1. What is an ions an atom that G positic Or 2. Use a periodic table to complete the following table. lon Symbol # Protons Element Name Fluorine Potassium Aluminum Lithium regative electrical change F Name Jordan Flame class # gain or lases one dan are elections acquiring 5 Ca+2 b. an anion & why? Rb+ 9 53 16 35 17 8 # Electrons 10 54 36 10 18 # of Electrons Lost Or Gained Gained one 3. If Li loses an electron to another atom, why is it written Li+1 (with a +1)? 4. If N gains 3 electrons from other atoms, why is it written N-3(with a -3)? Gained Two Lost One Lost One Gained Two 5. a. What do you think happens to atomic radius (size) of a cation (positive ion) & why?
lon Practice Set 1. What is an ions an atom that G positic Or 2. Use a periodic table to complete the following table. lon Symbol # Protons Element Name Fluorine Potassium Aluminum Lithium regative electrical change F Name Jordan Flame class # gain or lases one dan are elections acquiring 5 Ca+2 b. an anion & why? Rb+ 9 53 16 35 17 8 # Electrons 10 54 36 10 18 # of Electrons Lost Or Gained Gained one 3. If Li loses an electron to another atom, why is it written Li+1 (with a +1)? 4. If N gains 3 electrons from other atoms, why is it written N-3(with a -3)? Gained Two Lost One Lost One Gained Two 5. a. What do you think happens to atomic radius (size) of a cation (positive ion) & why?
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
I need help on both pages, i have answered some.

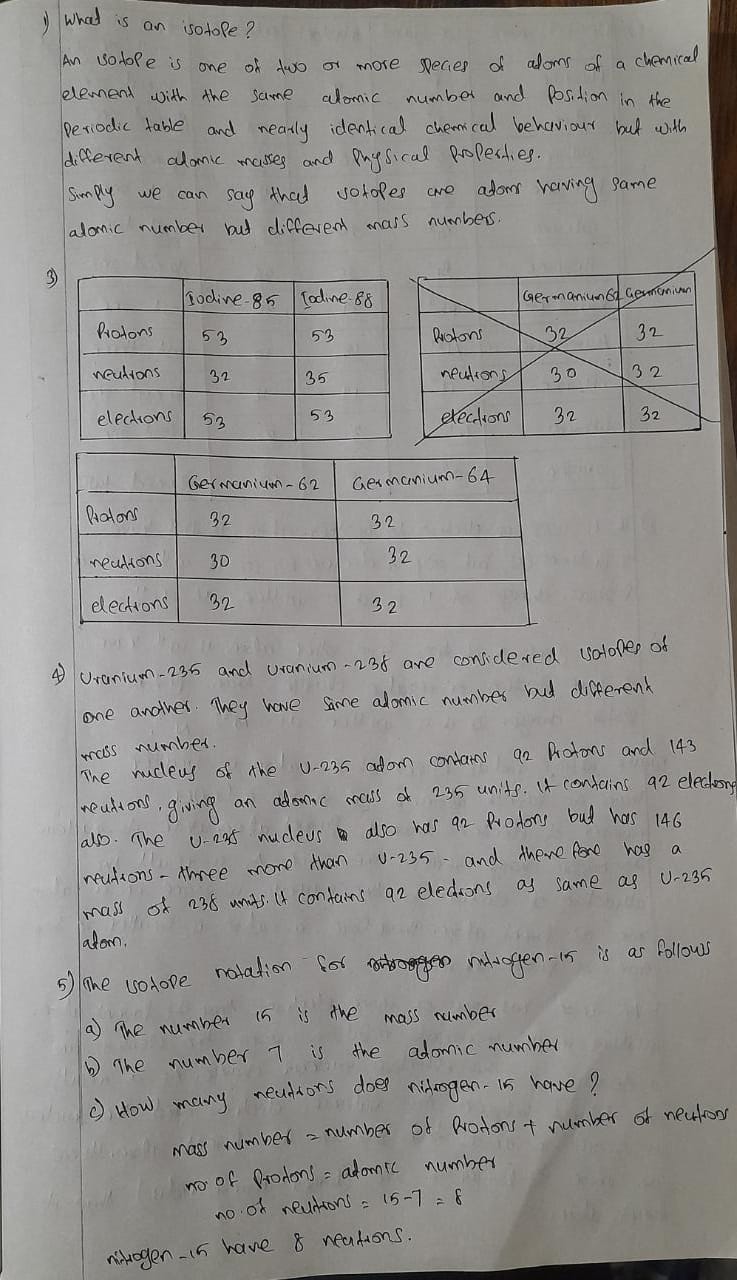
Transcribed Image Text:Isotope Practice Set
1. What is an isotope?
2. Fill in each chart below with information about each isotope. Assume all atoms are neutral.
Carbon- 12 Carbon - 16
Chromium-58 Chromium-63
6
6
6
ional
# protons
# neutrons
# electrons
C
# protons
# neutrons
# electrons
34
24
lodine -
39
24
3. Fill in the isotope names and any missing information, including isotope numbers. Assume
all atoms are neutral.
32
lodine
# protons
#neutrons
# electrons
35
#protons
#neutrons
# electrons
5. The isotope notation for nitrogen-15 is as follows:
a. The number 15 is the
b. The number 7 is the
c. How many neutrons does nitrogen-15 have?
6
10
6
32
30
number.
number.
235 similar, and how are they different?
4. Uranium-235 and Uranium-238 are considered isotopes of one another. How are uranium-
32

Transcribed Image Text:NY
lon Practice Set
an
1. What is on tone an
atom
a form that
positive or regative electrical change
2. Use a periodic table to complete the following table.
Element
Name
Fluorine
Potassium
Aluminum
Lithium
Ion Symbol
F
Ca+2
b. an anion & why?
Rb+
gain or loses one of
# Protons
9
53
16
35
17
#
Hame class
or more electrons acquiring
8
# Electrons
10
54
36
10
18
# of Electrons
Lost Or
Gained
3. If Li loses an electron to another atom, why is it written Li +¹ (with a +1)?
4. If N gains 3 electrons from other atoms, why is it written N-3(with a -3)?
Gained one
Gained Two
Lost One
Lost One
Gained Two
5. a. What do you think happens to atomic radius (size) of a cation (positive ion) & why?
Expert Solution
Step 1
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY