QUESTION 3 What is the extinction coefficient of Fe(SCN)2+ in this experiment? Hint: The value you need is located on page 168 of your lab manual. Give the answer in L/mol-cm as shown in the manual. QUESTION 4 How do you convert between %T and Absorbance? OA. A = %T x 100 B. A = 1/%T x 100 OC. A = log (100/%T) OD.A = log %T
QUESTION 3 What is the extinction coefficient of Fe(SCN)2+ in this experiment? Hint: The value you need is located on page 168 of your lab manual. Give the answer in L/mol-cm as shown in the manual. QUESTION 4 How do you convert between %T and Absorbance? OA. A = %T x 100 B. A = 1/%T x 100 OC. A = log (100/%T) OD.A = log %T
Oh no! Our experts couldn't answer your question.
Don't worry! We won't leave you hanging. Plus, we're giving you back one question for the inconvenience.
Submit your question and receive a step-by-step explanation from our experts in as fast as 30 minutes.
You have no more questions left.
Message from our expert:
Our experts need more information to provide you with a solution. Need to see the page 168 of your lab manual Please resubmit your question, making sure it's detailed and complete. We've credited a question to your account.
Your Question:
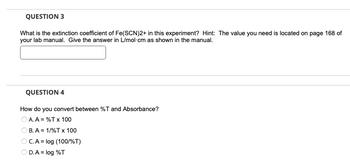
Transcribed Image Text:QUESTION 3
What is the extinction coefficient of Fe(SCN)2+ in this experiment? Hint: The value you need is located on page 168 of
your lab manual. Give the answer in L/mol-cm as shown in the manual.
QUESTION 4
How do you convert between %T and Absorbance?
OA. A = %T x 100
B. A = 1/%T x 100
OC. A = log (100/%T)
OD.A = log %T
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

