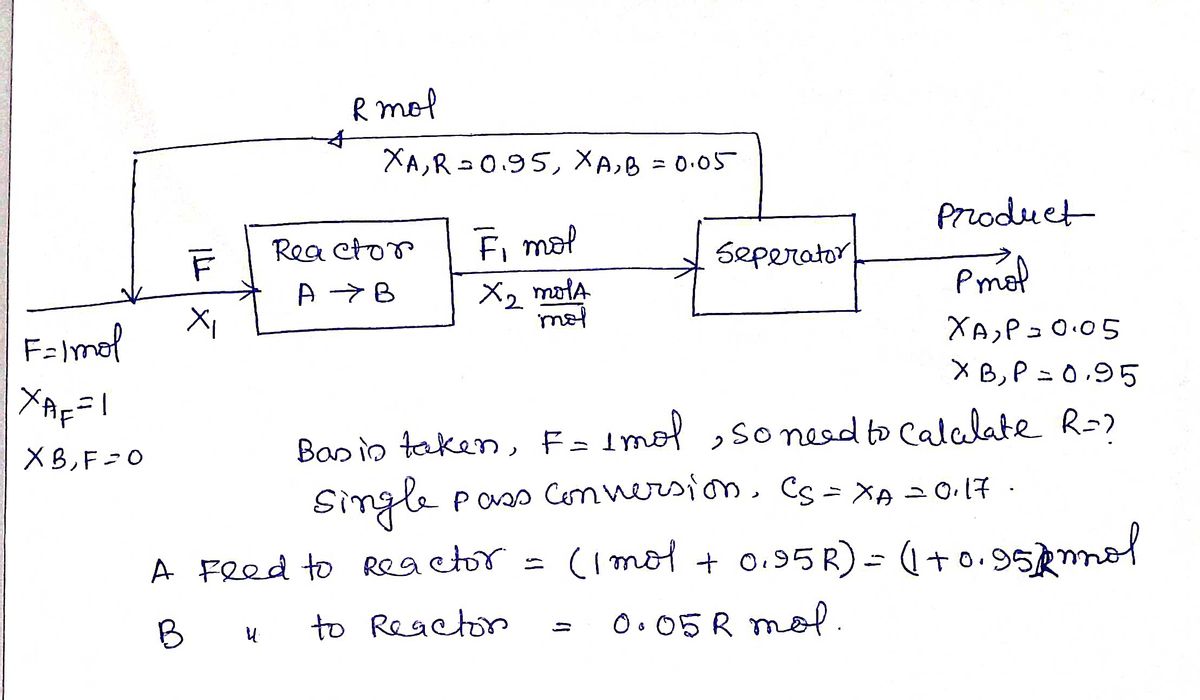
Consider the process shown below where a feed of pure A enters a reactor where the reaction A-B occurs. The separator after the reactor is not working properly. The final circuit product is only 95 mol. 9% B with 5 mol.9% A, and the recycle stream contains 5 mol.% B and 95 mol.% A. Recycle (R) x4.R = 0.95, xB.R = 0.05 Feed (F) Reactor Product (P) XAF = 1 Separator A→ B X4P = 0.05 XB.F = 0 XB.P = 0.95 If the single pass conversion in the reactor is Cs = 17 %, then determine the required recycle rate, expressed as moles recycled (Stream R) per mole of feed (Stream F). Enter your answer rounded off to two decimal places.
Consider the process shown below where a feed of pure A enters a reactor where the reaction A-B occurs. The separator after the reactor is not working properly. The final circuit product is only 95 mol. 9% B with 5 mol.9% A, and the recycle stream contains 5 mol.% B and 95 mol.% A. Recycle (R) x4.R = 0.95, xB.R = 0.05 Feed (F) Reactor Product (P) XAF = 1 Separator A→ B X4P = 0.05 XB.F = 0 XB.P = 0.95 If the single pass conversion in the reactor is Cs = 17 %, then determine the required recycle rate, expressed as moles recycled (Stream R) per mole of feed (Stream F). Enter your answer rounded off to two decimal places.
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question

Transcribed Image Text:Consider the process shown below where a feed of pure A enters a reactor where the reaction A-B occurs. The separator after the reactor is not working properly. The final circuit product is only 95 mol. 9% B with 5 mol.9% A, and
the recycle stream contains 5 mol.% B and 95 mol.% A.
Recycle (R) x4.R = 0.95, xB.R = 0.05
Feed (F)
Reactor
Product (P)
XAF = 1
Separator
A→ B
X4P = 0.05
XB.F = 0
XB.P = 0.95
If the single pass conversion in the reactor is Cs = 17 %, then determine the required recycle rate, expressed as moles recycled (Stream R) per mole of feed (Stream F). Enter your answer rounded off to two decimal places.
Expert Solution
Step 1
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The