3. Read the following passage and then solve the problems. In the equation that follows each problem, write in the space provided the mole ratio that can be used to solve the problem. Complete the equation by writing the correct value on the line provided. The reaction of sodium peroxide and water produces sodium hydroxide and oxygen gas. The following balanced chemical equation represents the reaction. 2Na2O2(s) + 2H₂O(1)→ 4NaOH(s) + O₂(g) a) How many moles of sodium hydroxide are produced when 1.00 mol sodium peroxide reacts with water? 80 S tesT weive-109rlax10W 1.00 mol Na2O2 × b) How many moles of oxygen gas are produced when 0.500 mol Na2O2 reacts with water? 0.500 mol Na2O₂ × c) How many moles of sodium peroxide are needed to produce 1.00 mol sodium hydroxide? 1.00 mol NaOH x d) How many moles of water are required to produce 2.15 mol oxygen gas in this reaction? mol H₂O 2.15 mol O₂ x e) How many moles of water are needed for 0.100 mol of sodium peroxide to react completely in this reaction? = mol NaOH mol O₂ mol Na₂O₂ 0.100 mol Na₂O₂ × mol H₂O f) How many moles of oxygen are produced if the reaction produces 0.600 mol sodium hydroxide? mol O₂ 0.600 mol NaOH x 1012157503 inatedor
Thermochemistry
Thermochemistry can be considered as a branch of thermodynamics that deals with the connections between warmth, work, and various types of energy, formed because of different synthetic and actual cycles. Thermochemistry describes the energy changes that occur as a result of reactions or chemical changes in a substance.
Exergonic Reaction
The term exergonic is derived from the Greek word in which ‘ergon’ means work and exergonic means ‘work outside’. Exergonic reactions releases work energy. Exergonic reactions are different from exothermic reactions, the one that releases only heat energy during the course of the reaction. So, exothermic reaction is one type of exergonic reaction. Exergonic reaction releases work energy in different forms like heat, light or sound. For example, a glow stick releases light making that an exergonic reaction and not an exothermic reaction since no heat is released. Even endothermic reactions at very high temperature are exergonic.

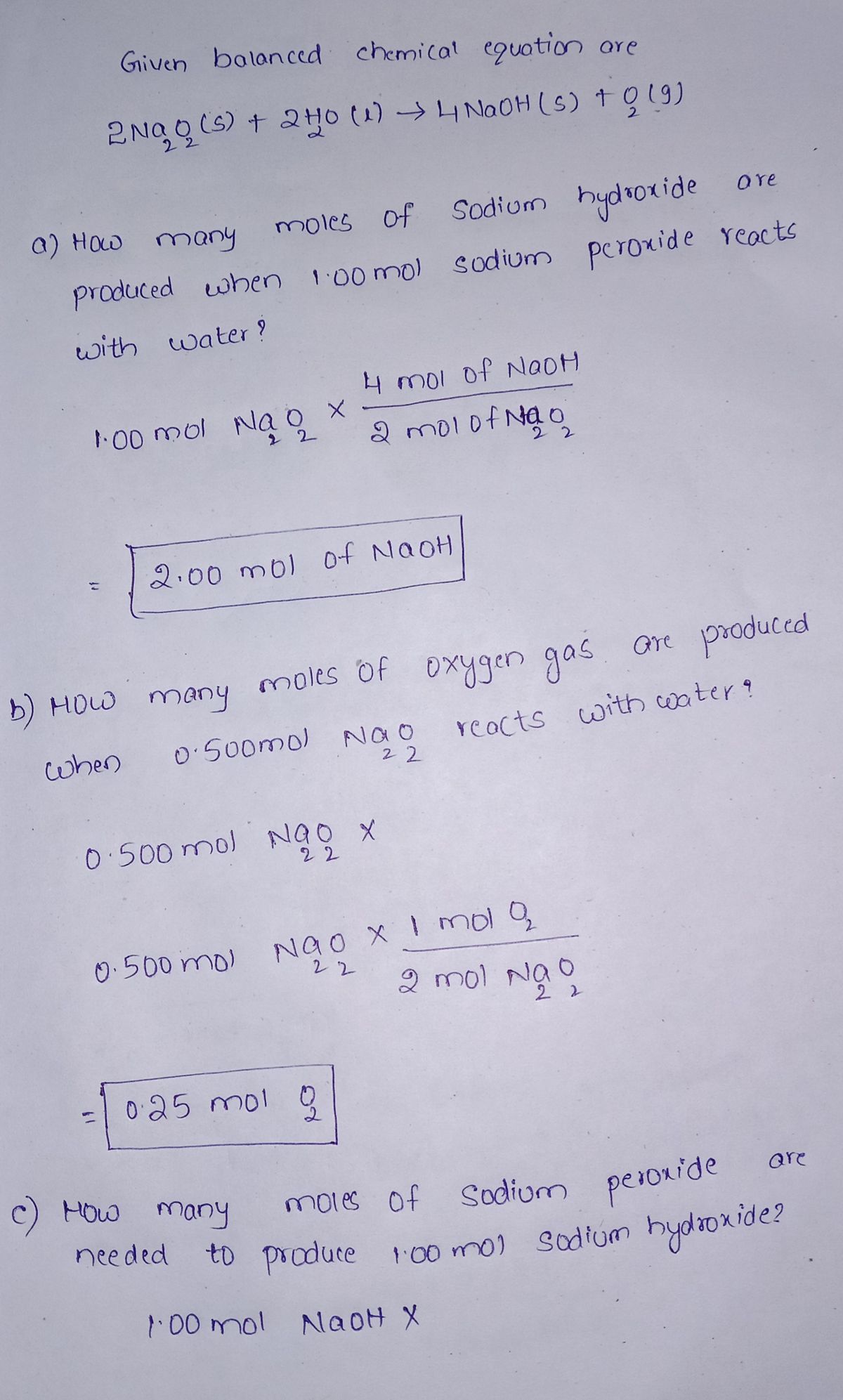
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images









