2.1 Determine the wavelength of photon emitted when an electron moves from n = 2 orbit to n = 1 orbit in a gold atom. If Z is the atomic number, and for gold Z = 79. Also, by how much energy will the bombarding electrons excite the gold atom to radiate this emission line?
Q: 2. are -6.00 eV, -4.00 eV, and -1.00 eV. The allowed energy levels of a hypothetical hydrogenic…
A:
Q: Qs: Determine the frequency of the wave length which emitted from an atom has an electron transited…
A: Given data: The atom makes the transition from (a) ni= 3 nf=1 (b) ni= 2 nf=1 Formula used: When an…
Q: A hydrogen atom initially in the ground level absorbs a photon, which excites it to the n=4 level.…
A:
Q: Using the Bohr formulas, estimate the average distance from the nucleus for an electron in the…
A: Given value--- electron orbit n = 1 . Atomic number = 92 . We have to find--- What is its kinetic…
Q: a. How much energy is required to ionize a hydrogen atom containing an electron in the n=4 level? b.…
A:
Q: What is the frequency of light (in Hz) of a photon emitted when an electron in a hydrogen atom…
A:
Q: Given that 0.750 mol of a particular atom all release an identical photon, resulting in a total…
A: Given: The moles of a particular atom is 0.750 mol. The total energy of change is -176.0 kJ.
Q: universe.Assuming that none of the other properties of the universe are changed, what would be the…
A:
Q: 1. A hydrogen atom initially in its ground state (n=1) absorbs a photon and ends up in the state for…
A:
Q: What is the the wavelength of a photon emitted when an electron in hydrogen decays from the n= 7…
A:
Q: 4. The sun approximates an ideal blackbody radiator at a temperature of 5825K. (a) By Wien’s Law,…
A:
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: E=−Ry/n2…
A: Given, Rydberg's constant, R=1.0973x107 m-1 n1=6 n2=12
Q: How much energy would it take to remove the electron from unexcited hydrogen completely so that it…
A: The basic line of attack that would be useful to solve this question would beif a body is bound to…
Q: Using the Boh model of an electron orbiting a nucleus, the angular momentum of Earth's orbit around…
A:
Q: The nucleus of a certain atom has a radius of 4.0 × 10^-15 m. An electron orbits the nucleus at a…
A: Given data The radius of the nucleus is rn = 4 x 10-15 m The orbiting radius of the electron is Ro =…
Q: A photon is absorbed by a hydrogen atom in the ground state. If the electron is boosted from to the…
A: Initially electron is in ground state i.e. n = 1 Finally electron jumps to n = 6
Q: A hydrogen atom in an n=2 state absorbs a photon. Part a: What wavelength photons might be emitted…
A:
Q: can find the value of the Rydberg energy using the Data button on the ALEKS toolbar.) Calculate the…
A:
Q: he emission spectrum of Hydrogen has a bright blue-green line at 486 ??. This electron ends in the…
A:
Q: helium, , electron drops from the n=3 state to the n=2 state. (give the answers in Scientific…
A:
Q: ydrogen atom emits a photon when its electron shifts from a higher energy level to a lower one.…
A:
Q: Calculate the ionisation potential for Hydrogen. Given : e = 1.6 x 10-19 C; m = 9.1 x 1031 kg; h =…
A: In chemistry and physics, ionisation energy, also known as ionisation potential, is the amount of…
Q: What are possible emission wavelengths when the electron of a hydrogen atom release energy and make…
A:
Q: The energy E of the electron in a hydrogen atom can be calculated from the Bohr formula: R 23 n² E…
A:
Q: Calculate the wavelength, in nanometers, of the photon emitted when the electron in a hydrogen atom…
A: AnswerWe have to calculate the wavelength λof the photon emitted when…
Q: Describe how the hydrogen atom might absorb a photon of energy less than 13.6 eV. Describe a process…
A: Given:- A photon can be absorbed when it is equal to the difference of energy between…
Q: 4. A proton and an electron recombine to form atomic hydrogen in its 4p state. At what wavelengths…
A:
Q: 1. A hydrogen atom is in the ground state. What frequency photon is required to knock the electron…
A: Using the relation of energy and frequency in this question and then repeat the same for other…
Q: The Lyman series of lines in the emission spectrum of hydrogen corresponds to transitions from…
A:
Q: The figure below shows some energy levels of the lithium atom. I) Considering the conditions for…
A: By absorbing or producing a photon of a particular specific wavelength, electrons can go up and down…
Q: The following diagram shows the complete set of orbitals of a hypothetical atom. The yellow circle…
A: Theory:The energy absorbed or released for transition of electrons between two energy states E1 and…
Q: e E1 = 0.00 eV, E2 = 1.34 eV, and E3 = 3.95 eV. %3D %3D
A: "As per our company policy we can answer only one question at a time. Kindly repost the remaining…
Q: From Bohr's atomic model, write an equation giving the wavelength emitted when an electron passes…
A: To write an equation giving the wavelength emitted when an electron passes from a ni level to an nf…
Q: Consider the Bohr model of the atom. Suppose an electron in a hydrogen atom transitions from the n=3…
A:
Q: (a) What is the wavelength of the photon needed to excite an electron from E₁ to E4? x 10 (b) What…
A:
Q: Calculate the speed of the electron in a hydrogen atom in the state n = 5, in m/s. Express your…
A:
Q: An electron in the n=2 level of an hydrogen atom absorbs a photon of wavelength 410. nm. To which…
A:
Q: How can elements with low atomic numbers have so many spectral lines? 1. Elements with low atomic…
A:
Q: A electron in a hydrogen atom transitions from the n=2 energy level to the ground state (n=1). What…
A: Given data: Initial energy level n = 2 Final energy level n = 1
Q: Solve correctly please. How much energy, in eV, is needed to move an electron in a hydrogen atom…
A:
Q: Find the largest possible wavelength for electrons (m = 9.11 x 10-31kg emitted from sodium (work…
A: Given Mass of electron (m) = 9.11 x 10-31kg Energy (E) = 5.90 x 10-19 J Work…
Q: Show that the frequency of the photon emitted by a hydrogen atom in going from state n + 1 to n is…
A: For an electron in the nth circular orbit of the hydrogen atom, its velocity is given as V=2πrTr is…
2.1 Determine the wavelength of photon emitted when an electron moves from n = 2 orbit to n = 1 orbit in a gold atom. If Z is the atomic number, and for gold Z = 79. Also, by how much energy will the bombarding electrons excite the gold atom to radiate this emission line?
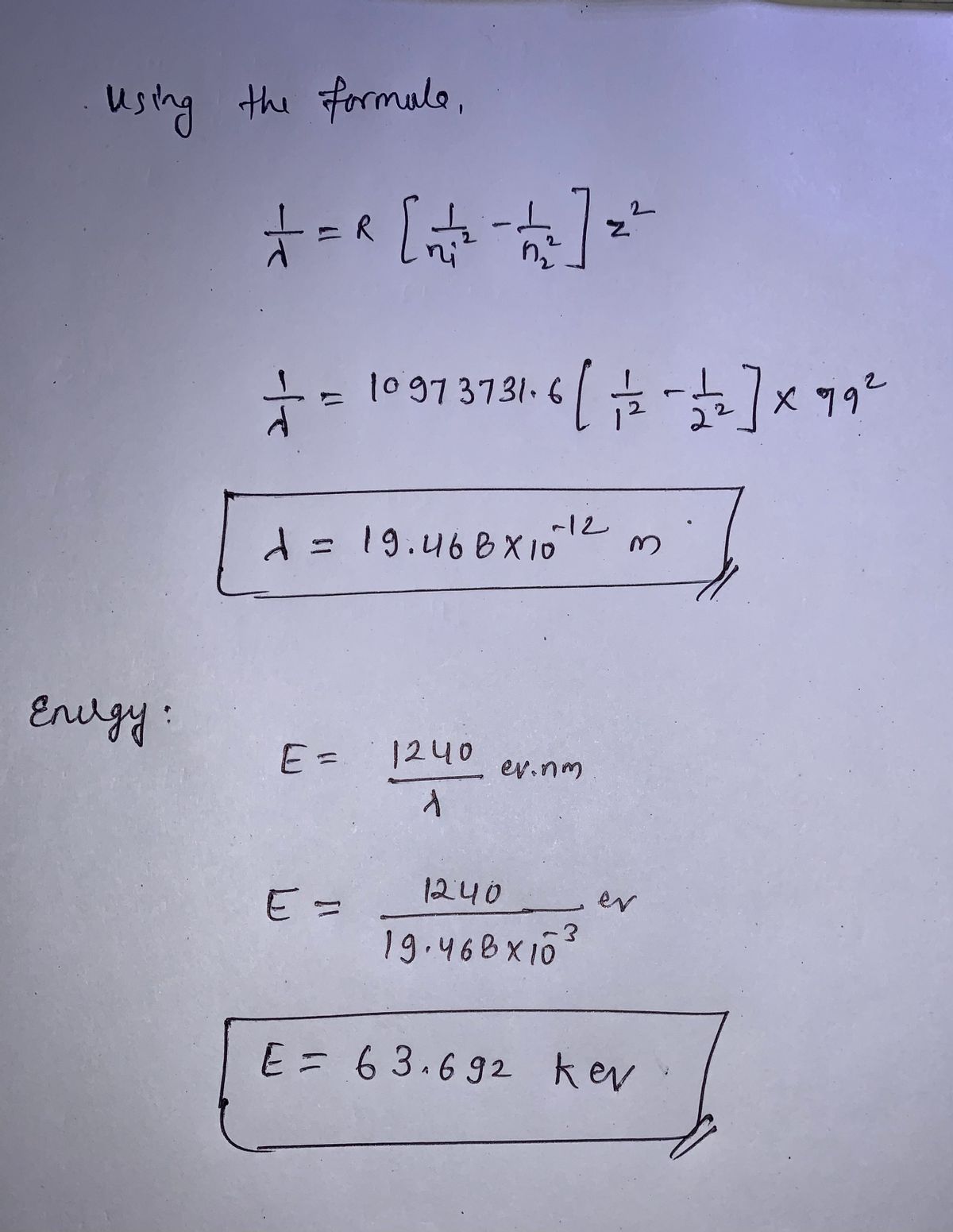
Step by step
Solved in 2 steps with 1 images
