
Concept explainers
Practice Problem G.1
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for this reaction.
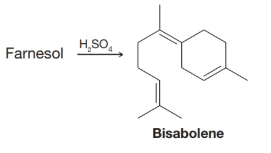
Interpretation:
The mechanism for the formation of Bisabolene from Farnesol in presence of 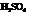 is to be outlined.
is to be outlined.
Concept introduction:
Farnesol is a compound that is formed from isoprene compounds. In general, when geranyl pyrophosphate reacts with isopenenyl pyrophosphate, farnesyl pyrophosphate (intermediate for the synthesis of sesquiterpenes) is formed. This, on oxidation can give farnesol.
Concentrated sulfuric acid acts as a strong dehydrating agent and removes the water molecule from alcohols to form unsaturated hydrocarbon.
Answer to Problem 1PP
Solution:
The mechanism for the synthesis of Bisabolene is explained as follows:

Explanation of Solution
During the first step, Farnesol (unsaturated alcohol) reacts with acid (HA = 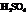 ) to form a hydrated product. Then, the hydrated product formed loses one water molecule to form a carbocation. Consequently, this carbocation rearranges to form a more stable carbocation. At last, the base
) to form a hydrated product. Then, the hydrated product formed loses one water molecule to form a carbocation. Consequently, this carbocation rearranges to form a more stable carbocation. At last, the base 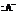 (
( 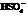 ) abstracts the hydrogen ion from the molecule, forming Bisabolene.
) abstracts the hydrogen ion from the molecule, forming Bisabolene.
The whole mechanism can be depicted as:

Hence, Bisabolene has been formed from Farnesol in the presence of concentrated sulfuric acid.
The Bisabolene has been synthesized from Farnesol in the presence of concentrated sulfuric acid. The mechanism for the synthesis of Bisabolene is outlined as:

Want to see more full solutions like this?
Chapter G Solutions
Organic Chemistry
Additional Science Textbook Solutions
Cosmic Perspective Fundamentals
Principles of Anatomy and Physiology
Human Physiology: An Integrated Approach (8th Edition)
Chemistry: A Molecular Approach (4th Edition)
Genetic Analysis: An Integrated Approach (3rd Edition)
- Draw the Fischer projection from the skeletal structure shown below. HO OH OH OH OH H Q Drawing Atoms, Bonds and Rings Charges I ☐ T HO H H OH HO I CH2OH H OH Drag H OH -CH2OH CHO -COOH Undo Reset Remove Donearrow_forwardplease provide the structure for this problem, thank youarrow_forwardpresented by Morallen Lig Intermine the hand product for the given mution by adding atoms, bonds, nonhonding diarion panda скуль Step 3: Comp the draw the product Step 2: Agama workup Compithe 429 ملولةarrow_forward
- Reaction A 0,0arrow_forwardpresented by Morillon Leaning Predict the organic product for the min кусур HSC Adithane carved arnown to come than that to the condon slchroruis in acid in in aquishri with ноюarrow_forward6.15PM Sun Mar 30 K Draw the major product of this reaction. Include any relevant stereochemistry. Ignore inorganic byproducts. Problem 1 of O H [PhзPCH2CH3]*C|¯ NaH Drawing > Q Atoms, Bonds and Draw or tap a nearrow_forward
- 8:17 PM Sun Mar 30 Draw the major product of this reaction. Ignore inorganic byproducts. HSCH2CH2CH2SH, BF3 Probler Drawing Ato Bonds Clarrow_forwardpresented by Mr L How the coprion. (Il Done in no wraction, dew the starting redential) доarrow_forward8:16 PM Sun Mar 30 K Draw the major product of this reaction. Ignore inorganic byproducts. Proble 1. CH3MgBr 2. H3O+ F Drawingarrow_forward
- о но оarrow_forwardName the major organic product of the following action of 4-chloro-4-methyl-1-pentanol in neutral pollution 10+ Now the product. The product has a molecular formula f b. In a singly hain, the starting, material again converts into a secule with the molecular kormula CIO. but with comply Draw the major organic structure inhalationarrow_forwardMacmillan Learning Alcohols can be oxidized by chromic acid derivatives. One such reagent is pyridinium chlorochromate, (C,H,NH*)(CICTO3), commonly known as PCC. Draw the proposed (neutral) intermediate and the organic product in the oxidation of 1-butanol by PCC when carried out in an anhydrous solvent such as CH₂C₁₂. PCC Intermediate OH CH2Cl2 Draw the intermediate. Select Draw Templates More с H Cr о Product Draw the product. Erase Select Draw Templates More H о Erasearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

