
ORGANIC CHEMISTRY W/BIOLOGICAL TOPICS
6th Edition
ISBN: 9781260325294
Author: SMITH
Publisher: RENT MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.14, Problem 28P
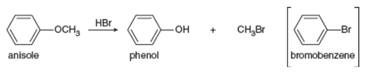
Explain why the treatment of anisole with
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
can you help me solve and highlight these hw
can you please help me draw these structures for HW
can you please help me draw these two structures , it is homework
Chapter 9 Solutions
ORGANIC CHEMISTRY W/BIOLOGICAL TOPICS
Ch. 9.1 - Problem 9.1 Label each ether and alcohol in...Ch. 9.3 - Give the IUPAC name for each compound.Ch. 9.3 - Problem 9.3 Give the structure corresponding to...Ch. 9.3 - Name each of the following ethers.Ch. 9.3 - Name each epoxide.
a. (two ways) b. c. (two...Ch. 9.6 - Problem 9.8 Draw the organic product of each...Ch. 9.6 - Prob. 9PCh. 9.6 - Problem 9.10 Draw the products of each reaction.
...Ch. 9.8 - Problem 9.11 Draw the products formed when each...Ch. 9.8 - Prob. 12P
Ch. 9.11 - Problem 9.18 Draw the products of each reaction,...Ch. 9.11 - Problem 9.19 What is the major product formed...Ch. 9.12 - Prob. 19PCh. 9.12 - Prob. 20PCh. 9.12 - Problem 9.22 Draw the organic products formed in...Ch. 9.12 - Problem 9.23 Draw two steps to convert into each...Ch. 9.13 - Prob. 23PCh. 9.13 - Problem 9.25 Draw the products of each reaction,...Ch. 9.13 - Draw the products formed when (S)-butan-2-ol is...Ch. 9.13 - Draw the product formed when (CH3)2CHOH is treated...Ch. 9.14 - What alkyl halides are formed when each ether is...Ch. 9.14 - Explain why the treatment of anisole with HBr...Ch. 9.15 - Name each thiol.
a. b.
Ch. 9.15 - Draw the product of each reaction. ac b.d.Ch. 9.15 - Give the IUPAC name for each sulfide.
a. b.
Ch. 9.15 - Draw the product of each reaction.
a. b.
Ch. 9.16 - Prob. 33PCh. 9.16 - The cis and trans isomers of 2, 3-dimethyloxirane...Ch. 9.16 - Problem 9.36 Draw the product of each...Ch. 9 - 9.37 Name each compound depicted in the...Ch. 9 - Answer each question using the ball-and-stick...Ch. 9 - Prob. 38PCh. 9 - 9.40 Give IUPAC name for each...Ch. 9 - Prob. 40PCh. 9 - Prob. 41PCh. 9 - 9.46 What alkenes are formed when each alcohol is...Ch. 9 - Prob. 46PCh. 9 - Prob. 51PCh. 9 - 9.57 Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 53PCh. 9 - 9.59 Draw two different routes to each of the...Ch. 9 - Prob. 55PCh. 9 -
9.61 Draw the products formed when each ether is...Ch. 9 - 9.62 Draw a stepwise mechanism for each...Ch. 9 - Draw a stepwise, detailed mechanism for the...Ch. 9 - Prob. 59PCh. 9 - Draw the products of each reaction. a.c. b.d.Ch. 9 - Prob. 64PCh. 9 - Prob. 75P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Label each statement about the polynucleotide ATGGCG as true or false. The polynucleotide has six nucleotides. ...
General, Organic, and Biological Chemistry - 4th edition
2. Define equilibrium population. Outline the conditions that must be met for a population to stay in genetic e...
Biology: Life on Earth (11th Edition)
Why is it unlikely that two neighboring water molecules would be arranged like this?
Campbell Biology (11th Edition)
To test your knowledge, discuss the following topics with a study partner or in writing ideally from memory. Th...
HUMAN ANATOMY
What process causes the Mediterranean intermediate Water MIW to become more dense than water in the adjacent At...
Applications and Investigations in Earth Science (9th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- please help with synthesisarrow_forward10. Stereochemistry. Assign R/S stereochemistry for the chiral center indicated on the following compound. In order to recieve full credit, you MUST SHOW YOUR WORK! H₂N CI OH CI カー 11. () Stereochemistry. Draw all possible stereoisomers of the following compound. Assign R/S configurations for all stereoisomers and indicate the relationship between each as enantiomer, diastereomer, or meso. NH2 H HNH, -18arrow_forwardb) 8. Indicate whether the following carbocation rearrangements are likely to occur Please explain your rational using 10 words or less not likely to occur • The double bond is still in the Same position + Likely to oc occur WHY? -3 H3C Brave Chair Conformers. Draw the chair conformer of the following substituted cyclohexane. Peform a RING FLIP and indicate the most stable conformation and briefly explain why using 20 words or less. CI 2 -cobs ?? MUST INDICATE H -2 -2 Br EQ Cl OR AT Br H& most stable WHY? - 4arrow_forward
- CH 12 Conformational Analysis. Draw all 6 conformers (one above each letter) of the compound below looking down the indicated bond. Write the letter of the conformer with the HIGHEST and LOWEST in energies on the lines provided. NOTE: Conformer A MUST be the specific conformer of the structure as drawn below -4 NOT HOH OH 3 Conformer A: Br OH A Samo Br H 04 Br H H3 CH₂ H anti stagere Br CH clipsed H Brott H IV H MISSING 2 -2 B C D E F X 6 Conformer with HIGHEST ENERGY: 13. (1 structure LOWEST ENERGY: Nomenclature. a) Give the systematic (IUPAC) name structure. b) Draw the corresponding to this name. HINT: Do not forget to indicate stereochemistry when applicable. a) ८८ 2 "Br {t༐B,gt)-bemn€-nehpརི་ཚ༐lnoa Parent name (noname) 4 Bromo Sub = 2-methylethyl-4 Bromo nonane b) (3R,4S)-3-chloro-4-ethyl-2,7-dimethyloctane # -2 -2arrow_forwardin the scope of the SCH4U course! please show all steps as im still learning how to format my answers in the format given, thank you!arrow_forwardhelp me solve this HWarrow_forward
- Molecules of the form AH2 can exist in two potential geometries: linear or bent. Construct molecular orbital diagrams for linear and bent CH2. Identify the relevant point group, include all of the appropriate symmetry labels and pictures, and fill in the electrons. Which geometry would you predict to be more stable, and why? (Please draw out the diagram and explain)arrow_forwardIndicate the variation in conductivity with concentration in solutions of strong electrolytes and weak electrolytes.arrow_forwardThe molar conductivity of a very dilute solution of NaCl has been determined. If it is diluted to one-fourth of the initial concentration, qualitatively explain how the molar conductivity of the new solution will compare with the first.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY