
(a)
The mass of sugar dissolved in
(a)
Answer to Problem 1QAP
The mass of sugar dissolved in
Explanation of Solution
Given Amount of water is
Temperature is
Formula used:
Where,
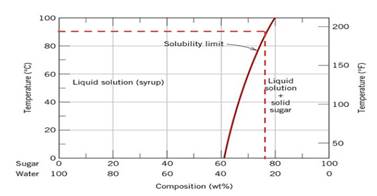
According to diagram
Calculations Put value of
Conclusion So, the mass of sugar dissolved in
(b)
Composition of saturated liquid solution at
(b)
Answer to Problem 1QAP
Composition of the saturated liquid solution is
Explanation of Solution
From the sugar-water phase diagram when the liquid saturated solution from temperature
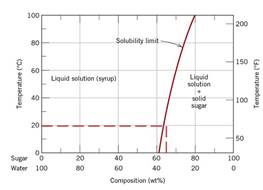
Conclusion Hence, the composition of the saturated liquid solution is
(c)
Mass of solid sugar comes out of the solution upon cooling to
(c)
Answer to Problem 1QAP
Mass of solid sugar comes out of the solution upon cooling to
Explanation of Solution
Given Amount of water is
Cooling Temperature is
Mass of sugar dissolved at
Formula used:
Solubility of sugar in water is given by:
Where,
Mass of sugar solidifies upon cooling to temperature
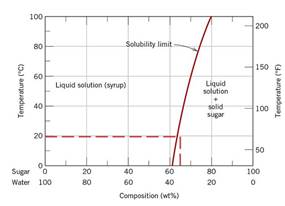
According to diagram
Calculations Put value of
Mass of sugar solidifies upon cooling to temperature
Conclusion So, the mass of sugar that solidified upon cooling to temperature
Want to see more full solutions like this?
Chapter 9 Solutions
MATERIALS SCI + ENGR: INT W/ACCESS
- If you could help me answer these questions in matlab that would be great, I provided an additional picture detailing what the outcome should look like.arrow_forwardUsing the table below, design a third (3th) order Butterworth HPF with a 4 KHz cutoff frequency. What is the additional stage required to HPF to design a third order BPF. Explain your answe Order Stage poles DF 2 stage poles 3 stage DF poles DF 1 1 Optional 2 1.414 3 י 1 1 2 1.848 2 0.765 5 2 1 1.618 1 0.618 6 2 1.932 1.414 2 0.518arrow_forwardQuestion 1 Generate a random sample of standard lognormal data (rlnorm()) for sample size n = 100. Construct histogram estimates of density for this sample using Sturges’ Rule, Scott’s Normal Reference Rule, and the FD Rule. Question 2 Construct a frequency polygon density estimate for the sample in Question 1, using bin width determined by Sturges’ Rule.arrow_forward
- Generate a random sample of standard lognormal data (rlnorm()) for sample size n = 100. Construct histogram estimates of density for this sample using Sturges’ Rule, Scott’s Normal Reference Rule, and the FD Rule.arrow_forwardConsider the feedback controlled blending system shown below, which is designed to keep theoutlet concentration constant despite potential variations in the stream 1 composition. The density of all streamsis 920 kg/m3. At the nominal steady state, the flow rates of streams 1 and 2 are 950 and 425 kg/min,respectively, the liquid level in the tank is 1.3 m, the incoming mass fractions are x1 = 0.27, x2 = 0.54. Noticethe overflow line, indicating that the liquid level remains constant (i.e. any change in total inlet flow ratetranslates immediately to the same change in the outlet flow rate). You may assume the stream 1 flowrate andthe stream 2 composition are both constant. Use minutes as the time unit throughout this problem. Identify any controlled variable(s) (CVs), manipulated variable(s) (MVs),and disturbance variable(s) (DVs) in this problem. For each, explain how you know that’show it is classified.CVs: ___________, MVs: _____________, DVs: ______________ b) Draw a block diagram…arrow_forwardA biofilm consists of living cells immobilized in a gelatinous matrix. A toxic organic solute (species A) diffuses into the biofilm and is degraded to harmless products by the cells within the biofilm. We want to treat 0.2 m3 per hour of wastewater containing 0.14 mole/m of the toxic substance phenol using a system consisting of biofilms on rotating disk as shown below. thickness of the biofilm is 1 mm and we are interested to reduce the phenol concentration in the outlet stream to 0.025 mole/m3. The rate of disappearance of phenol (species A) within the biofilm is described by the following equation rA = - KICA Where k1 = 0.0024 s-1 The diffusivity of phenol in the biofilm at the process temperature of 25°C is 2.0x10 10 m2/s. Phenol is equally soluble in both water and the biofilm. 1. The concentration of A in the biofilm is give by Ca = Bisinh(mz) + Bzcosh(mz), the numerical value of m is. 2. Use m = 3 mm^-1, determine the numerical values with units for B1 and B2 with the unit of…arrow_forward
- A heat transfer experiment is conducted on two identical spheres which are initially at the same temperature. The spheres are cooled by placing them in a channel. The fluid velocity in the channel is non-uniform, having a profile as shown. Which sphere cools off more rapidly? Explain. V 1arrow_forwarduse the integral method if you can, thank you!arrow_forward.I need the correct answer, and if it's wrong, please fix it 7. The midrange voltage gain of an amplifier is 100. The input RC circuit has a lower critical frequency of 1 kHz. The actual voltage gain at f-100 Hz is 100. 10. In a high-pass filter, the roll-off region occurs above the critical frequency.arrow_forward
- A Water at A flows out of the 1-in.-diameter nozzle at 8 ft s and strikes the 0.5 -lb-plate.Determine the height h above the nozzle at which the plate can be supported by the water jetarrow_forwardMy ID# 016948724 last 2 ID# 24 Last 3 ID# 724 Please help to find the correct answer for this problem using my ID# first write le line of action and then help me to find the forces {fx= , fy= mz= and for the last find the moment of inertial about the show x and y axes please show how to solve step by steparrow_forwardMy ID# 016948724 last 2 ID# 24 Last 3 ID# 724 Please help to find the correct answer for this problem using my ID# first write le line of action and then help me to find the forces and the tension {fx= , fy= mz=arrow_forward
 MATLAB: An Introduction with ApplicationsEngineeringISBN:9781119256830Author:Amos GilatPublisher:John Wiley & Sons Inc
MATLAB: An Introduction with ApplicationsEngineeringISBN:9781119256830Author:Amos GilatPublisher:John Wiley & Sons Inc Essentials Of Materials Science And EngineeringEngineeringISBN:9781337385497Author:WRIGHT, Wendelin J.Publisher:Cengage,
Essentials Of Materials Science And EngineeringEngineeringISBN:9781337385497Author:WRIGHT, Wendelin J.Publisher:Cengage, Industrial Motor ControlEngineeringISBN:9781133691808Author:Stephen HermanPublisher:Cengage Learning
Industrial Motor ControlEngineeringISBN:9781133691808Author:Stephen HermanPublisher:Cengage Learning Basics Of Engineering EconomyEngineeringISBN:9780073376356Author:Leland Blank, Anthony TarquinPublisher:MCGRAW-HILL HIGHER EDUCATION
Basics Of Engineering EconomyEngineeringISBN:9780073376356Author:Leland Blank, Anthony TarquinPublisher:MCGRAW-HILL HIGHER EDUCATION Structural Steel Design (6th Edition)EngineeringISBN:9780134589657Author:Jack C. McCormac, Stephen F. CsernakPublisher:PEARSON
Structural Steel Design (6th Edition)EngineeringISBN:9780134589657Author:Jack C. McCormac, Stephen F. CsernakPublisher:PEARSON Fundamentals of Materials Science and Engineering...EngineeringISBN:9781119175483Author:William D. Callister Jr., David G. RethwischPublisher:WILEY
Fundamentals of Materials Science and Engineering...EngineeringISBN:9781119175483Author:William D. Callister Jr., David G. RethwischPublisher:WILEY





