
a)

Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
Catalytic hydrogenation is a heterogeneous process which occurs with syn stereochemistry. Both hydrogens adsorbed on the catalytic surface add to the double bond from the same face.
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is ethylbenzene.
Explanation of Solution
The addition of hydrogen takes place to the double bond in the side chain with syn stereochemistry. Both hydrogens add to the double bond from the same face to yield ethylbenzene.
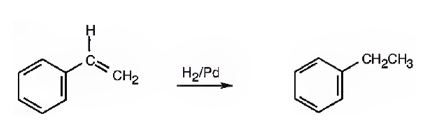
The product of the reaction is ethylbenzene.
b)
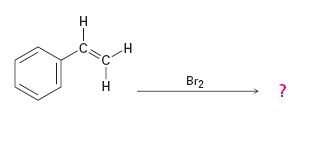
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
Addition of bromine to the double bond in
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
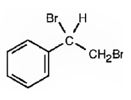
Explanation of Solution
In the first step the addition of bromine to the double bond in 1,2-dimethylcyclohexane results in the formation of a cyclic bromonium ion with the simultaneous elimination of a bromide ion. The large bromonium ion shields one side of the molecule. Hence the attack of the bromide ion occurs from the opposite, unshielded side to yield a trans-product.
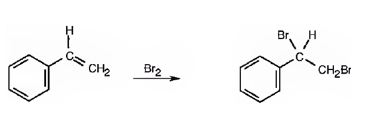
The product of the reaction is
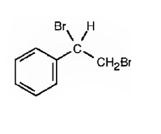
c)
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
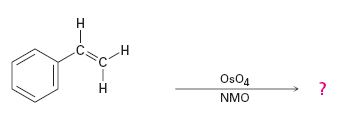
Hydroxylation of double bonds can be carried out directly by treating the alkene with osmium tetroxide, OsO4, in the presence of N-phenylmorpholine N-oxide. The reaction occurs with syn stereochemistry through the formation of a cyclic intermediate, called osmate, formed by the addition of OsO4 to the alkene in a single step. The cyclic osmate is then cleaved to give the cis-1,2-
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
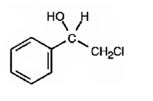
Explanation of Solution
OsO4 adds to the double bond in the side chain from the same side to yield a cyclic osmate in a single step. When treated with NaHSO3 the cyclic osmate gets cleaved to give the diol.
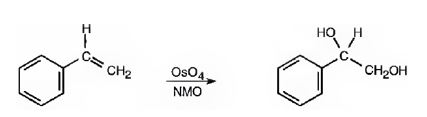
The product of the reaction is
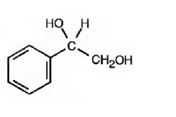
d)
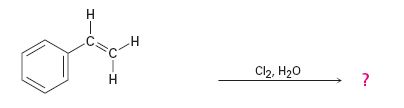
Interpretation:
To predict the product of the reaction shown with regiochemistry, if relevant.
Concept introduction:
The addition of chlorine to alkenes in the presence of water results in the anti addition of the halohydrin, HOCl, to the double bond. In the first step a cyclic chloronium ion is formed by the attack of the π electrons of the double bond on chlorine. In the second step water attacks the chloronium ion from the least shielded side to give an anti addition product. The addition obeys Markovnokov orientation. The negative part (OH) adds to the doubly bonded carbon atom which has more number of substituents.
To predict:
The product of the reaction shown with regiochemistry, if relevant.
Answer to Problem 42AP
The product of the reaction is
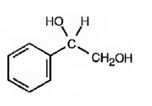
Explanation of Solution
The given alkene has an unsymmetrically substituted double bond in the side chain. In the first step a cyclic chloronium ion is formed by the attack of the double bond on chlorine. In the second step water attacks the the chloronium ion from the opposite less shielded side to give an intermediate which then loses a proton to yield the product.
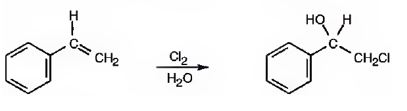
The product of the reaction is
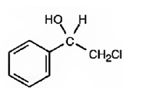
e)
Interpretation:
The product expected in the reaction shown is to be given.
Concept introduction:
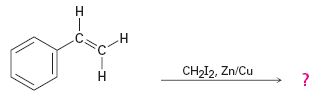
The reaction given is an example of Simmons-Smith reaction. When CH2I2 is treated with Zn/Cu couple, iodomethylzinc iodide, ICH2ZnI, is formed. This ICH2ZnI transfers a CH2 group to the double bond in alkene to form a cyclopropane ring in the product.
To give:
The product expected in the reaction shown.
Answer to Problem 42AP
The product expected in the reaction shown is
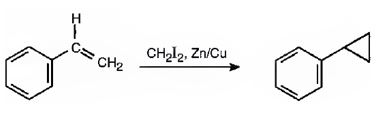
Explanation of Solution
When CH2I2 is treated with Zn/Cu couple, iodomethylzinc iodide, ICH2ZnI, is formed. This ICH2ZnI transfers a CH2 group to the double bond in the side chain to form a cyclopropane ring in the product.
The product expected in the reaction shown is
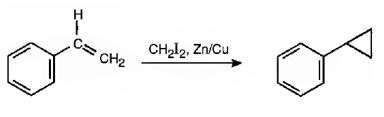
f)
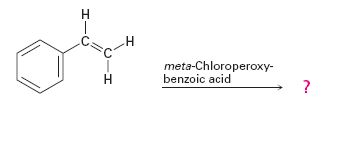
Interpretation:
The product expected in the reaction shown is to be given.
Concept introduction:
Alkenes are oxidized to give
To give:
The product expected in the reaction shown.
Answer to Problem 42AP
The product expected in the reaction shown is
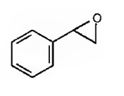
Where RCO3H = meta-chloroperoxybenzoic acid
Explanation of Solution
The oxygen transfer from the peroxy acids to the double bond in the side chain takes place with syn stereochemistry. Both the C-O bonds are formed from the same face of the double bond through a one step process without the formation of an intermediate to yield the product.
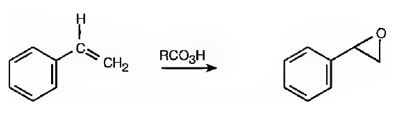
The product expected in the reaction shown is
Want to see more full solutions like this?
Chapter 8 Solutions
EBK ORGANIC CHEMISTRY
- help with the rf values i am so confusedarrow_forwardPredict the organic reactant of X and Y that are involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forwardPredict the major organic product for this reaction.arrow_forward
- Predict the major organic product for this reaction.arrow_forwardPredict the major organic product for this reaction.arrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forward
- Predict the major organic product for this reaction.arrow_forward3) The following molecule, chloral is a common precursor to chloral hydrate, an acetal type molecule that was a first-generation anesthetic. Draw a mechanism that accounts for tis formation and speculate why it does not require the use of an acid catalyst, like most hemiacetal and acetal reaction: (10 pts) H H₂Oarrow_forwardYou are a Quality Manager for a very well-known food ingredient company that produces umami powder, and you are responsible for setting specification limits. The net weight (in grams) of bags of unami powder is monitored by taking samples of six bags on an hourly basis during production. The label on every bag reports a contents of 1KG umami powder. The process mean is μ = 1012 g, and when the process is properly adjusted, it varies with σ = 11 g. QUESTION: Your organisation strives to ensure that >99.97% of bags of umami powder produced conforms to specification. What performance process index value is required to achieve this process yield? Calculate PPK using the following formula: Ppk = (USL – mean)/3 σ Ppk = (mean -LSL)/ 3 σarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


