
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.6, Problem 16P
After a proton is removed from the OH group, which compound in each pair would form a cyclic ether more rapidly?
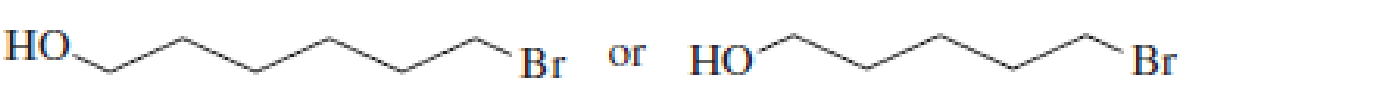

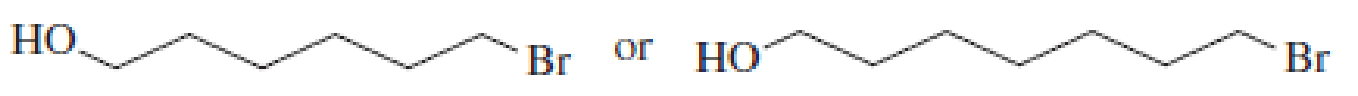
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write chemical equations for:
the reaction of benzoic acid chloride with grignard reagent [CH3MgX]
the reaction of butanoic acid with methyl amine [CH3NH2]
2-(3-Aminopropyl)cyclohexan-1-one is reacted with H₂SO₄. Draw the structures of the products.
Please help me solve number 2
Chapter 8 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 8.1 - Prob. 2PCh. 8.1 - Does increasing the energy barrier for an SN2...Ch. 8.1 - Arrange the following alkyl bromides in order from...Ch. 8.2 - Prob. 7PCh. 8.2 - Which reaction in each of the following pairs...Ch. 8.2 - Prob. 9PCh. 8.2 - Prob. 11PCh. 8.3 - Draw the substitution products that will be formed...Ch. 8.4 - Arrange the following alkyl halides in order from...Ch. 8.5 - Prob. 14P
Ch. 8.5 - Which of the following reactions will go faster if...Ch. 8.6 - After a proton is removed from the OH group, which...Ch. 8.6 - Draw the product of each of the following...Ch. 8.9 - Prob. 20PCh. 8.9 - Prob. 21PCh. 8.11 - Why do the SN1/E1 reactions of tertiary alkyl...Ch. 8.11 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.12 - Prob. 25PCh. 8.12 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - a In which solvent would tert-butyl bromide...Ch. 8.13 - What would be the best way to prepare the...Ch. 8 - Methoxychlor is an insecticide that was intended...Ch. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Explain how the following changes would affect the...Ch. 8 - Prob. 38PCh. 8 - Draw the major product obtained when each of the...Ch. 8 - Which alkyl halide in Problem 39 can undergo an El...Ch. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Starting with bromocyclohexane, how could the...Ch. 8 - Prob. 48PCh. 8 - Fill in the blanks in the following chemical...Ch. 8 - For each of the following alkyl halides, indicate...Ch. 8 - Prob. 51PCh. 8 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 8 - An ether can be prepared by an SN2 reaction of an...Ch. 8 - Give two sets of reactants (each set including an...Ch. 8 - Show how the following compounds could be...Ch. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Draw the structures of the products obtained from...Ch. 8 - cis-4-Bromocyclohexanol and...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - In which solventethanol or diethyl etherwould the...Ch. 8 - The pKa of acetic acid in water is 4.76. What...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Choose the best reagents to complete the following reaction. 오 Na2Cr2O7 H2SO4, H2O Problem 22 of 35 A Na2Cr2O7 H2SO4, H2O H2/Pt B pressure OH 1. NaBH4 C 2. H3O+ D DMP (Dess-Martin Periodinane) CH2Cl2 CrO3 Done Dramabana_Minor Submitarrow_forwardIndicate the products of the reaction of Cycloheptanone with pyrrolidine (cat. H+). Draw the structures of the compounds.arrow_forwardIndicate the products of the reaction of 2-(3-aminopropyl)cyclohexan-1-one with H2SO4. Draw the structures of the compounds.arrow_forward
- Indicate the products of the reaction of 2-cyclopentyl-2-methyl-1,3-dioxolane with H3O+. Draw the structures of the compounds.arrow_forwardQuestion 4 For the molecule shown below, (7 marks): A) Sketch the Newman projection for the view looking along the bond from the perspective of the arrow. B) Then, draw the Newman projection for each 60° rotation along the bond until it returns to the starting point. C) Clearly indicate which Newman projection is the one we see in the structure shown below, and clearly indicate which Newman projection is the highest in energy and which is the lowest in energy. H H Me 'H Me Mearrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts and the amine side product. 'N' 1. NaOH, heat 2. Neutralizing work-up Select to Drawarrow_forward
- Submit Problem 3 of 10 Draw the major product of this reaction. Ignore inorganic byproducts and the amine side product. O 'N' NH 1. NaOH, heat 2. Neutralizing work-up Select to Drawarrow_forwardb) Certain cyclic compounds are known to be conformationally similar to carbohydrates, although they are not themselves carbohydrates. One example is Compound C shown below, which could be imagined as adopting four possible conformations. In reality, however, only one of these is particularly stable. Circle the conformation you expect to be the most stable, and provide an explanation to justify your choice. For your explanation to be both convincing and correct, it must contain not only words, but also "cartoon" orbital drawings contrasting the four structures. Compound C Possible conformations (circle one): Детarrow_forwardLab Data The distance entered is out of the expected range. Check your calculations and conversion factors. Verify your distance. Will the gas cloud be closer to the cotton ball with HCI or NH3? Did you report your data to the correct number of significant figures? - X Experimental Set-up HCI-NH3 NH3-HCI Longer Tube Time elapsed (min) 5 (exact) 5 (exact) Distance between cotton balls (cm) 24.30 24.40 Distance to cloud (cm) 9.70 14.16 Distance traveled by HCI (cm) 9.70 9.80 Distance traveled by NH3 (cm) 14.60 14.50 Diffusion rate of HCI (cm/hr) 116 118 Diffusion rate of NH3 (cm/hr) 175.2 175.2 How to measure distance and calculate ratearrow_forward
- For the titration of a divalent metal ion (M2+) with EDTA, the stoichiometry of the reaction is typically: 1:1 (one mole of EDTA per mole of metal ion) 2:1 (two moles of EDTA per mole of metal ion) 1:2 (one mole of EDTA per two moles of metal ion) None of the abovearrow_forwardPlease help me solve this reaction.arrow_forwardIndicate the products obtained by mixing 2,2-dimethylpropanal with acetaldehyde and sodium ethoxide in ethanol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY