
Interpretation:
The thermal efficiency of the cycle and the fraction of the steam entering the turbine is extracted for the feed water heater should be determined.
Concept Introduction:
The thermal efficiency of the cycle is calculated as:
Here
The entropy is constant for idealistic turbine. Hence an isentropic process is developed between the enter and exit pressures of the idealistic turbine. The idealistic condenser has constant enthalpy.
Answer to Problem 8.8P
And
Explanation of Solution
Given information:
It is given thatcycle is a regenerative cycle in a steam power plant includes one feedwater heater, turbine, condenser and a boiler. Steam enters the turbine at
Figure to describe the process in simplified form is given as below
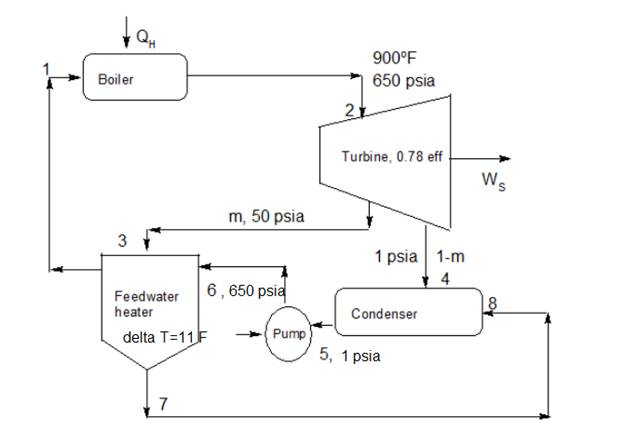
From figure point 1 and point 6 has same operating conditions.
Considering idealistic turbine for which entropy is constant at inlet and outlet
at point 2 and at
Now for idealistic turbine, entropy is constant at inlet and outlet. Hence
Exhaust coming to condenser is at pressure
The steam coming to condenser is wet and saturated. Hence for saturated steam at
At
At
From linear interpolation, if
Similarly
And
And
We also know that
Therefore,
Hence the actual enthalpy of the steam exhaust from the turbine to the condenser is calculated by the thermodynamics efficiency of the work produced from the turbine which is
Hence
and
Exhaust coming to feedwater heater is at pressure
The steam coming to feedwater heater is superheated. Hence for superheated steam at
From linear interpolation, if
Hence the actual enthalpy of the steam exhaust from the turbine to the feedwater heater is calculated by the thermodynamics efficiency of the work produced from the turbine which is
And entropy at
Now,
The idealistic condenser has constant enthalpy and hence the enthalpy of saturated liquid at inlet point 4 of condenser is same as enthalpy at the outlet of condenser at point 5.
And at
Hence work required for circulation of the steam from the condenser to the feedwater heater is calculated fromusing the pump efficiency is
And the work-done by pump is also calculated as
Now it is given that steam for the feedwater heater is at
Hence at point 7 the steam is saturated liquid from feedwater heater going towards the condenser. Hence for
Hence the temperature at the point 1 according to the given condition and in condensing raises the temperature of the feedwater to within
And at
The volume expansivity for boiler is calculated as
Hence the enthalpy and entropy across the boiler is
And
Let mass of steam exhaust from the turbine going towards feedwater heater is
Basis
Applying energy balance across the feedwater heater. Hence fraction of the steam entering the turbine is extracted for the feedwater heater is
And
The thermal efficiency of the cycle is calculated as:
Here
And
Want to see more full solutions like this?
Chapter 8 Solutions
Loose Leaf For Introduction To Chemical Engineering Thermodynamics
- b) Explain the key features of the Langmuir adsorption model - Drawing a diagram with empty and occupied sites. Show how new molecules would adsorb. drawing the diagram, showing free and empty sites, and their number (to use for next section) - Define the capacity and binding affinity parameters in terms of things shown on the diagram Defining the capacity and binding affinity parameters in terms of bound, free sites, and free molecules - Plot what would be a typical breakthrough curve and give an explanation approximately when breakthrough would occur plotting a typical sigmoidal breakthrough curve and saying it would certainly occur by the time capacity is used, but also could be much earlier if the affinity is lowarrow_forwardWater at 20°C flows at a steady average velocity of 5.25 m/s through a smooth pipe of diameter 5.08 cm. The flow is fully developed through the entire section of pipe. The total pipe length is 10.56 m, and there are two 90' elbows. Determine the friction coefficient and the head loss due to friction per meter length of the pipe. Control volume Prepared by Engr. Kirsten Gaarrow_forwardProblem 2. For an irreversible liquid phase reaction A -> B, the reaction rate is of the first order with respect to the reactant concentration C_A. this reaction is performed in a cascade of two identical CSTRs at 100 degrees Celsius. (same reactor size and isothermal). The inlet concentration of A of the first CSTR is 2mol/L. The outlet concentration of A of the 2nd CSTR is 0.5 mol/L. the inlet flow rate of the 1st reactor is 100 L/h. and the feed temperature is 20 degrees Celsius. The average heat capacity of the reactant/product/solvent mixture is a constant: 2J/g*K, the density of the mixture is a constant: 1 kg/L. The heat of reaction is 50 kJ/mol (exothermic). The reaction rate constant at 100 degrees Celsius is 0.5/h. (a) Determine the outlet concentration of A of the first CSTR (b) What is the heat transfer requirement for the first CSTR? (c) if this reaction is performed in a plug-flow reactor, what is the size of plug-flow reactor required for achieving the same conversion…arrow_forward
- The energy release (Q_g) and energy loss (Q_r) curves of an irreversible oxidation reaction are shown below. Q_r curves can be shifted by adjusting the feed temperature. Q,& QE E Qg (a) Are these points of intersection between energy release and energy loss curves stable operating conditions? Point of Intersection A Stable or Unstable B A D T (b) Which point represents the ignition condition? B с D E F Garrow_forwardProblem 1. For an irreversible liquid phase reaction 2A -> B, the reaction rate is of the 2nd order with respect to the reactant concentration CA. The concentration-dependent reaction rate is plotted below. This reaction is performed in a cascade of two identical CSTRS (same reactor size and temperature). The inlet concentration of A of the 1st CSTR is 2 mol/L. The outlet concentration of A of the 2nd CSTR is 1 mol/L. The inlet flow rate of the 1st reactor is 100 L/h. Please use the graphical method to determine the outlet concentration of A of the first CSTR and the size of each CSTR. Please briefly show the procedure for reactor size calculation. (-4-7) 15225050 45 40 35 30 0 0.5 11.761.5 C₂ Q C (mol.L¹) Co 20 2.5arrow_forward15.15 A 0.20-m-thick brick wall (k = 1.3 W/m K) separates the combustion zone of a furnace from its surroundings at 25°C. For an outside wall surface temperature of 100°C, with a convective heat-transfer coefficient of 18 W/m² K, what will be the inside wall surface temperature at steady-state conditions? .arrow_forward
- An MF membrane has pore-size distribution as follows: d(pore)0.33 fraction¼ 1.5 mm, d(pore)0.33 fraction¼ 1.0 mm, and d(pore)0.33 fraction¼ 0.5 mm. Required (a) Determine the distribution of flux density for each pore size. (b) Show by a plot the distribution of pore sizes and the distribution of flux density. solvearrow_forwardPlease show me your steps please thank you.arrow_forwardPlease show your work thanks.arrow_forward
- A pipe 1.2 m diameter and 20 km long transmits water of velocity of 2 m/s. The friction coefficient of pipe is 0.007. Calculate the head loss due to friction?arrow_forwardA gas pipeline, NPS 20 with 0.500 in. wall thickness, transports natural gas (specific gravity = 0.6) at a flow rate of 250 MMSCFD at an inlet temperature of 60°F. Assuming isothermal flow, calculate the velocity of gas at the inlet and outlet of the pipe if the inlet pressure is 1000 psig and the outlet pressure is 850 psig. The base pressure and base temperature are 14.7 psia and 60°F, respectively. Assume compressibility factor Z = 1.00. What is the erosional velocity for this pipeline based on the above data and a compressibility factor Z = 0.90?arrow_forwardH.W.1 : The elementary liquid phase reaction A + BR + S is conducted in a setup consisting of a mixed reactor, into which two reactant solutions are introduced, followed by a plug flow reactor. The component B is used in a large excess so that the reaction is first order with respect to A. Various ways to increase the production rate are suggested, one of which is to reverse the order of these units. How would this change/affect conversion?arrow_forward
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





