
To determine:The shape of
Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution:
Trigonal pyramid.
Explanation of Solution
The electron dot structure of

It has three bonds and one pair of lone electrons. So, its shape is trigonal pyramid.
(c)
To determine:The shape of
Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution:
Linear.
Explanation of Solution
The electron dot structure of
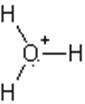
It has two bonds and three pairs of lone electrons. So, its shape is linear.
(d)
To determine:The shape of
. Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution:
Octahedral.
Explanation of Solution
The electron dot structure of
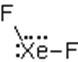
It has six bonds and no lone pair of electrons. So, its shape is octahedral.
(e)
To determine:The shape of
. Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution:
Square pyramidal.
Explanation of Solution
The electron dot structure of
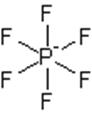
It has five bonds and one pair of lone electrons. So, its shape is square pyramidal.
(f)
To determine:The shape of
Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution:
Tetrahedral.
Explanation of Solution
The electron dot structure of

It has four bonds and no lone pair of electrons. So, its shape is tetrahedral.
(g)
To determine:The shape of
. Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution: Tetrahedral.
Explanation of Solution
The electron dot structure of
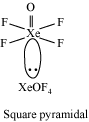
It has four bonds and no lone pair of electrons. So, its shape is tetrahedral.
(h)
To determine:The shape of
Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution: Tetrahedral.
Explanation of Solution
The electron dot structure of

It has four bonds and no lone pair of electrons. So, its shape is tetrahedral.
(i)
To determine:The shape of
. .concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution: Square planar.
Explanation of Solution
The electron dot structure of
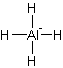
It has four bonds and two pairs of lone electrons. So, its shape is square planar.
(j)
To determine:The shape of
Concept Introduction:
- To predict the shapes, we have to considerer the special distribution of the atoms and the pairs of free electrons.
- The pairs of free electrons are those do not form part of bonds between atoms.
- The electron dot structure shows the valence electrons distribution around the atoms.
| Number of bonds | Number of pairs of free electrons | Shapes |
| 2 | 0 | Linear |
| 2 | 3 | Linear |
| 2 | 1 | Bent |
| 4 | 0 | Tetrahedral |
| 4 | 2 | Square planar |
| 3 | 0 | Trigonal planar |
| 5 | 1 | Square pyramidal |
| 3 | 1 | Trigonal pyramidal |
| 6 | 0 | Octahedral |
Answer to Problem 8.1P
Solution: Trigonal planar.
Explanation of Solution
The electron dot structure of

It has three bonds and no lone pair of electrons. So, its shape is trigonal planar.
Want to see more full solutions like this?
Chapter 8 Solutions
EBK CHEMISTRY
- Using the Nernst equation to calculate nonstandard cell voltage A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 3+ 3Cu²+ (aq) +2Al(s) → 3 Cu(s)+2A1³* (aq) 2+ Suppose the cell is prepared with 5.29 M Cu in one half-cell and 2.49 M A1³+ in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. x10 μ ☑ 00. 18 Ar Иarrow_forwardPlease help me solve this homework problemarrow_forwardPlease help me answer this homework questionarrow_forward
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3+ H2(g)+2OH¯ (aq) + 2Fe³+ (aq) → 2H₂O (1)+2Fe²+ (aq) 0 kJ x10 Х ? olo 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 184.2 mL of a 0.7800M solution of dimethylamine ((CH3) NH with a 0.3000M solution of HClO4. The pK₁ of dimethylamine is 3.27. Calculate the pH of the base solution after the chemist has added 424.1 mL of the HClO solution to it. 2 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO 4 solution added. Round your answer to 2 decimal places. pH = ☐ ☑ ? 000 18 Ar 1 Barrow_forwardUsing the Nernst equation to calculate nonstandard cell voltage A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: MnO2 (s)+4H* (aq)+2Cr²+ (aq) → Mn²+ (aq)+2H₂O (1)+2Cr³+ (aq) + 2+ 2+ 3+ Suppose the cell is prepared with 7.44 M H* and 0.485 M Cr²+ in one half-cell and 7.92 M Mn² and 3.73 M Cr³+ in the other. Calculate the cell voltage under these conditions. Round your answer to 3 significant digits. ☐ x10 μ Х 5 ? 000 日。arrow_forward
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. NO (g) +H₂O (1) + Cu²+ (aq) → HNO₂ (aq) +H* (aq)+Cu* (aq) kJ - ☐ x10 x10 olo 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid b An analytical chemist is titrating 116.9 mL of a 0.7700M solution of aniline (C6H5NH2) with a 0.5300M solution of HNO3. The pK of aniline is 9.37. Calculate the pH of the base solution after the chemist has added 184.2 mL of the HNO 3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☐ ☑ 5arrow_forwardQUESTION: Find the standard deviation for the 4 different groups 5.298 3.977 223.4 148.7 5.38 4.24 353.7 278.2 5.033 4.044 334.6 268.7 4.706 3.621 305.6 234.4 4.816 3.728 340.0 262.7 4.828 4.496 304.3 283.2 4.993 3.865 244.7 143.6 STDEV = STDEV = STDEV = STDEV =arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





