
Concept explainers
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
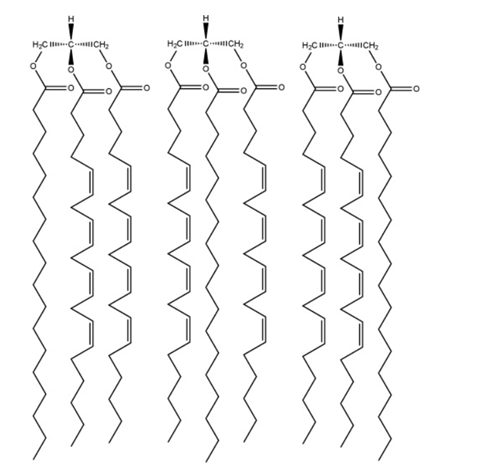
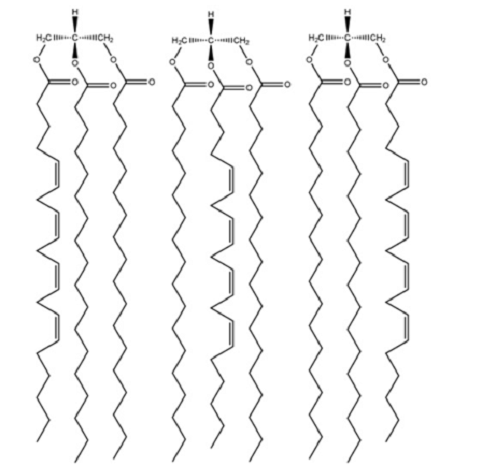
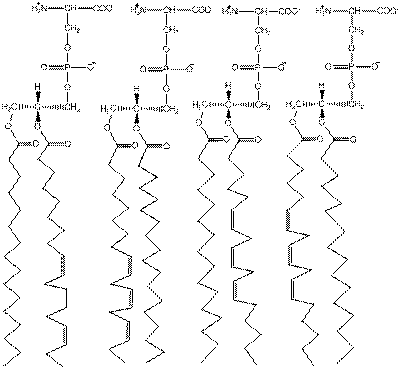
Drawing Structures of Triacylglycerols Draw the structures of (a) all the possible triacylglycerols that can be formed from glycerol with Stearic and arachidonic acid and (b) all the phosphatidylserine isomers that tan be formed from palmitic and linolenic acids.
(a)
To explain:
The formation from glycerol from stearic and arachidonic acid should be described.
Introduction:
Glycerol is the colorless, viscous liquids that belong to the organic group of alcohol. It has three carbon atoms and three hydroxyl groups in its structure that are known as trihydroxyl sugar alcohol.
Explanation of Solution
The chiral molecule is found in the second carbon of glycerol if there is the presence of different substitutes at both ends of carbon. This can lead to the formation of six triacylglycerols from stearic and arachidonic acids as below:
(b)
To explain:
The formation from phosphatidylserine isomers from palmitic and linolenic acids should be described.
Introduction:
A phospholipid with fatty acid is known as phosphatidylserine that covers and protects the cells in the brain and carries messages. It has two fatty acids attached with ester linkage to the C-1 and C-2 of glycerol and serine attached by a phosphodiester linkage to the C-3 of glycerol.
Explanation of Solution
There is a presence of phosphate bonded with a serine residue at C-3 of glycerol in structures of phosphatidylserine. There are two common forms of linolenic acid which means four different phosphatidylserine can be formed from palmitic and linolenic acids as shown below:
Want to see more full solutions like this?
Chapter 8 Solutions
EBK BIOCHEMISTRY
- The pyruvate dehydrogenase complex catalyzes the oxidative decarboxylation of pyruvate to form acetyl CoA. E₁, E2, and E3 are abbreviations for the enzymes of the complex. Classify the enzyme names, prosthetic groups, and reactions as E1, E2, or E3. E₁ E2 Answer Bank E3 transfer of electrons to FAD and then to NAD+ transfer of acetyl group to coenzyme A formation of hydroxyethyl-TPP hydroxyethyl group transferred to lipoamide thiamine pyrophosphate (TPP) FAD lipoamide dihydrolipoyl transacetylase pyruvate dehydrogenase dihydrolipoyl dehydrogenasearrow_forwardPatients with pyruvate dehydrogenase deficiency show high levels of lactic acid in the blood. However, in some cases, treatment with dichloroacetate (DCA), which inhibits the kinase associated with the pyruvate dehydrogenase complex, lowers lactic acid levels. How does DCA act to stimulate pyruvate dehydrogenase activity? DCA activates pyruvate dehydrogenase kinase. DCA increases phosphorylation levels of pyruvate dehydrogenase. DCA inhibits pyruvate dehydrogenase kinase. ODCA activates pyruvate dehydrogenase phosphatase. What does this suggest about pyruvate dehydrogenase activity in patients who respond to DCA? The pyruvate dehydrogenase complex is active only when phosphorylated by the kinase. The pyruvate dehydrogenase complex is active only in the presence of the kinase. The pyruvate dehydrogenase complex is completely inactive. The pyruvate dehydrogenase complex displays some residual activity.arrow_forwardThe reduced coenzymes generated by the citric acid cycle donate electrons in a series of reactions called the electron-transport chain. The energy from the electron-transport chain is used for oxidative phosphorylation. Which compounds donate electrons to the electron- transport chain? H₂O NADH பப NAD+ ATP ADP FADH₂ FAD Which compounds are the final products of the electron-transport chain and oxidative phosphorylation? H₂O NADH NAD+ ΠΑΤΡ Π ADP FADH₂ FAD Which compound is the final electron acceptor in the electron-transport chain? Оно NADH NAD+ ATP ADP FADH₂ FADarrow_forward
- Hexokinase in red blood cells has a Michaelis constant (KM) of approximately 50 μM. Because life is hard enough as it is, let's assume that hexokinase displays Michaelis-Menten kinetics. What concentration of blood glucose yields an initial velocity (V) equal to 90% of the maximal velocity (Vmax)? [glucose] = What does the calculated substrate concentration at 90% Vmax tell you if normal blood glucose levels range between approximately 3.6 and 6.1 mM? Hexokinase operates near Vmax only when glucose levels are low. Hexokinase normally operates far below Vmax. Hexokinase operates near Vmax only when glucose levels are high. Hexokinase normally operates near Vmax mMarrow_forwardClassify each coenzyme or distinguishing characteristic based on whether it corresponds to catalytic or stoichiometric coenzymes. Catalytic coenzymes Answer Bank Stoichiometric coenzymes lipoic acid FAD used once coenzyme A regenerated thiamine pyrophosphate (TPP) NAD+arrow_forwardThe oxidation of malate by NAD+ to form oxaloacetate is a highly endergonic reaction under standard conditions. AG +29 kJ mol¹ (+7 kcal mol-¹) Malate + NAD+ oxaloacetate + NADH + H+ The reaction proceeds readily under physiological conditions. = Why does the reaction proceed readily as written under physiological conditions? The NADH produced during glycolysis drives the reaction in the direction of malate oxidation. The steady-state concentrations of the products are low compared with those of the substrates. The reaction is pushed forward by the energetically favorable oxidation of fumarate to malate. Endergonic reactions such as this occur spontaneously without the input of free energy. Assuming an [NAD+ ]/[NADH] ratio of 8, a temperature of 25°C, and a pH of 7, what is the lowest [malate]/[oxaloacetate] ratio at which oxaloacetate can be formed from malate? [malate] [oxaloacetate]arrow_forward
- Calculate and compare the AG values for the oxidation of succinate by NAD+ and FAD. Use the data given in the table to find the E of the NAD+: NADH and fumarate:succinate couples, and assume that E for the enzyme-bound FAD: FADH2 redox couple is nearly +0.05 V. Oxidant Reductant " E' (V) NAD+ NADH + H+ 2 -0.32 Fumarate Succinate AG°' for the oxidation of succinate by NAD+: AG°' for the oxidation of succinate by FAD: 2 -0.03 Why is FAD rather than NAD+ the electron acceptor in the reaction catalyzed by succinate dehydrogenase? The electron-transport chain can regenerate FAD, but not NAD+. FAD is an oxidant, whereas NAD+ is a reductant. The oxidation of succinate requires two NAD+ molecules but only one FAD molecule. The oxidation of succinate by NAD+ is not thermodynamically feasible. kJ mol-1 kJ mol-1arrow_forwardUse the cellular respiration interactive to help you complete the passage. 2,4-dinitrophenol (DNP) was a popular ingredient in diet pills in the 1930s before it was discovered that moderate doses of the compound cause exceptionally high body temperature and even death. Complete the passage detailing how DNP's mechanism of action explains why it causes both high body temperature and weight loss. 2,4-dinitrophenol (DNP) causes of returning to the mitochondrial matrix through to pass directly across the inner mitochondrial membrane instead proteins. Because of DNP's effect on the mitochondrion, less energy is captured in the form of energy is instead wasted as heat. and more protons electrons ATP NADH sugars cytochrome ATP synthase heatarrow_forwardTo answer this question, you may reference the Metabolic Map. Select the reactions of glycolysis in which ATP is produced. 1,3-Bisphosphoglycerate 3-phosphoglycerate Glyceraldehyde 3-phosphate 1,3-bisphosphoglycerate Fructose 6-phosphate fructose 1,6-bisphosphate Phosphoenolpyruvate pyruvate Glucose glucose 6-phosphate Suppose 17 glucose molecules enter glycolysis. Calculate the total number of inorganic phosphate (P) molecules required as well as the total number of pyruvate molecules produced. P required: pyruvate produced: molecules moleculesarrow_forward
- Suppose a marathon runner depletes carbohydrate stores after a four-hour run. The runner's nutritionist suggests replenishing carbohydrate stores by eating carbohydrates. However, the runner is also concerned about weight loss and wants to know if fats can be directly converted into carbohydrates. How should the nutritionist respond to the runner? Yes, the glyoxylate cycle can be used to convert acetyl CoA into succinate, which can then be converted into carbohydrates. No, the two decarboxylation reactions of the citric acid cycle preclude the net conversion of acetyl CoA into carbohydrates. No, the citric acid cycle converts acetyl CoA into oxaloacetate, but there is no pathway to form glucose from oxaloacetate. Yes, pyruvate carboxylase can convert acetyl CoA into pyruvate, which can be used to form glucose through gluconeogenesis.arrow_forwardThe crossover technique can reveal the precise site of action of a respiratory-chain inhibitor. Britton Chance devised elegant spectroscopic methods for determining the proportions of the oxidized and reduced form of each carrier. This determination is feasible because the forms have distinctive absorption spectra, as illustrated in the graph for cytochrome c. Upon the addition of a new inhibitor to respiring mitochondria, the carriers between NADH and ubiquinol (QH2) become more reduced, and those between cytochrome c and O₂ become more oxidized. Where does your inhibitor act? Complex I Complex II Complex III Complex IV Absorbance coefficient (M-1 cm x 10-5) 10 1.0 0.5 400 Reduced Oxidized 500 Wavelength (nm) 600arrow_forwardWhy are the electrons carried by FADH2 not as energy rich as those carried by NADH? FADH2 carries fewer high-energy electrons than NADH. OFADH2 is less negatively charged than NADH. OFADH2 has a lower phosphoryl-transfer potential than NADH. FADH₂ has a lower reduction potential than NADH. What is the consequence of this difference? Electrons flow from NADH to FADH2 before they are transferred to O₂. Electron flow FADH₂ to O, results in the production of more ATP than does electron flow from NADH. Electron flow from FADH₂ to O, pumps fewer protons than does electron flow from NADH. Electron flow from FADH, to O, consumes more free energy than does electron flow from NADH. A simple equation relates the standard free-energy change, AG", to the change in reduction potential, AE. AG=-FAE Then represents the number of transferred electrons, and F is the Faraday constant with a value of 96.48 kJ mol¹ V-¹. Use the standard reduction potentials provided to determine the standard free energy…arrow_forward
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
