
CHEMISITRY W/OWL PKG LOOSELEAF
9th Edition
ISBN: 9781285903859
Author: ZUMDAHL
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7, Problem 91E
Given the valence electron orbital level diagram and the description, identify the element or ion.
a. A ground state atom
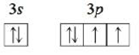
b. An atom in an excited state (assume two electrons occupy the 1s orbital)
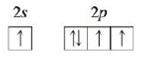
c. A ground state ion with a charge of – 1

Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method:
Regular Tomato Sauce
Salt Reduced Tomato Sauce
223.4
148.7
353.7
278.2
334.6
268.7
305.6
234.4
340.0
262.7
304.3
283.2
244.7
143.6
QUESTION: For both groups of data calculate the answers attached in the image.
Give reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium.
HAND WRITTEN PLEASE
edict the major products of the following organic reaction:
u
A
+
?
CN
Some important notes:
• Draw the major product, or products, of the reaction in the drawing area below.
• If there aren't any products, because no reaction will take place, check the box below the drawing area instead.
Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers.
Explanation
Check
Click and drag to start drawing a structure.
Х
© 2025 McGraw Hill LLC. All Rights Reserved. Te
LMUNDARY
Chapter 7 Solutions
CHEMISITRY W/OWL PKG LOOSELEAF
Ch. 7 - Four types of electromagnetic radiation (EMR) are...Ch. 7 - Characterize the Bohr model of the atom. In the...Ch. 7 - What experimental evidence supports the quantum...Ch. 7 - List the most important ideas of the quantum...Ch. 7 - What are quantum numbers? What information do we...Ch. 7 - How do 2p orbitals differ from each other? How do...Ch. 7 - Four blocks of elements in a periodic table refer...Ch. 7 - What is the difference between core electrons and...Ch. 7 - Prob. 9RQCh. 7 - The radius trend and the ionization energy trend...
Ch. 7 - Prob. 1ALQCh. 7 - Defend and criticize Bohrs model. Why was it...Ch. 7 - The first four ionization energies for the...Ch. 7 - Compare the first ionization energy of helium to...Ch. 7 - Which has the larger second ionization energy,...Ch. 7 - Explain why a graph of ionization energy versus...Ch. 7 - Without referring to your text, predict the trend...Ch. 7 - Account for the fact that the line that separates...Ch. 7 - Explain electron from a quantum mechanical...Ch. 7 - Choose the best response for the following. The...Ch. 7 - Consider the following statement "The ionization...Ch. 7 - Prob. 12ALQCh. 7 - How does probability fit into the description of...Ch. 7 - What is meant by an orbital?Ch. 7 - Explain the difference between the probability...Ch. 7 - Is the following statement true or false? The...Ch. 7 - Which is higher in energy, the 2s or 2p orbital,...Ch. 7 - Prove mathematically that it is more energetically...Ch. 7 - What type of relationship (direct or inverse) e...Ch. 7 - What do we mean by the frequency of...Ch. 7 - Explain the photoelectric effectCh. 7 - Describe briefly why the study of electromagnetic...Ch. 7 - How does the wavelength of a fast-pitched baseball...Ch. 7 - The following is an energy-level diagram for...Ch. 7 - The Bohr model works for only one electron...Ch. 7 - We can represent both probability and radial...Ch. 7 - Consider the representations of the p and d atomic...Ch. 7 - The periodic table consists of four blocks of...Ch. 7 - Many times the claim is made that subshells...Ch. 7 - Prob. 30QCh. 7 - Elements with very large ionization energies also...Ch. 7 - The changes in electron affinity as one goes down...Ch. 7 - Why is it much harder to explain the line spectra...Ch. 7 - Scientists use emission spectra to confirm the...Ch. 7 - Does the minimization of electron-electron...Ch. 7 - In the hydtogen atom, what is the physical...Ch. 7 - The work function is the energy required to remove...Ch. 7 - Many more anhydrous lithium salts are hygroscopic...Ch. 7 - The laser in an audio CD player uses light with u...Ch. 7 - An FM radio station broadcasts at 99.5 MHz....Ch. 7 - Microwave radiation has a wavelength on the order...Ch. 7 - A photon of ultraviolet (UV) light possesses...Ch. 7 - Octyl methoxycinoamate and oxybenzone are common...Ch. 7 - Human color vision is " produced" by the nervous...Ch. 7 - Consider the following waves representing...Ch. 7 - One type of electromagnetic radiation has a...Ch. 7 - Carbon absorbs energy at a wavelength of 150. nm....Ch. 7 - X rays have wavelengths on the order of 1 1010 m....Ch. 7 - The work function of an element is the energy...Ch. 7 - It takes 208.4 kJ of energy to remove 1 mole of...Ch. 7 - It takes 7.21 1019 J of energy to remove an...Ch. 7 - Ionization energy is the energy required to remove...Ch. 7 - Calculate the de Broglie wavelength for each of...Ch. 7 - Neutron diffraction is used in determining the...Ch. 7 - A particle has a velocity that is 90.% of the...Ch. 7 - Calculate the velocities of electrons with de...Ch. 7 - Calculate the wavelength of light emiued when each...Ch. 7 - Calculate the wavelength of light emitted when...Ch. 7 - Using vertical lines, indicate the transitions...Ch. 7 - Using vertical lines, indicate the transitions...Ch. 7 - Calculate the longest and shortest wavelengths of...Ch. 7 - Assume that a hydrogen atoms electron has been...Ch. 7 - Does a photon of visible light ( 400 to 700 nm)...Ch. 7 - An electron is excited from the n = 1 ground state...Ch. 7 - Calculate the maximum wavelength of light capable...Ch. 7 - Consider an electron for a hydrogen atom in an...Ch. 7 - An excited hydrogen atom with an electron in the n...Ch. 7 - An excited hydrogen atom emits light with a...Ch. 7 - Using the Heisenberg uncertainty principle,...Ch. 7 - The Heisenberg uncertainty principle can be...Ch. 7 - What are the possible values for the quantum...Ch. 7 - Identify each of the following orbitals and...Ch. 7 - Which of the following sets of quantum numbers are...Ch. 7 - Which of the following sets of quantum numbers are...Ch. 7 - What is the physical significance of the value of...Ch. 7 - In defining the sizes of orbitals, why must we use...Ch. 7 - Total radial probability distributions for the...Ch. 7 - Tbe relative orbital levels for the hydrogen atom...Ch. 7 - How many orbitals in an atom can have the...Ch. 7 - How many electrons in an atom can have the...Ch. 7 - Give the maximum number of electrons in an atom...Ch. 7 - Give the maximum number of electrons in an atom...Ch. 7 - Draw atomic orbital diagrams representing the...Ch. 7 - For elements l36, there are two exceptions to the...Ch. 7 - The elements Si, Ga, As, Ge, Al, Cd, S, and Se are...Ch. 7 - The elements Cu, O, La, Y, Ba, Tl, and Bi are all...Ch. 7 - Write the expected electron configurations for...Ch. 7 - Write the expected electron configurations for...Ch. 7 - Write the expected ground-state electron...Ch. 7 - Using only the periodic table inside the front...Ch. 7 - Given the valence electron orbital level diagram...Ch. 7 - Identify the following elements. a. An excited...Ch. 7 - In the ground state of mercury, Hg, a. how many...Ch. 7 - In the ground state of element 115, Uup, a. how...Ch. 7 - Give a possible set of values of the four quantum...Ch. 7 - Give a possible set of values of the four quantum...Ch. 7 - Valence electrons are those electrons in the...Ch. 7 - How many valence electrons do each of the...Ch. 7 - A certain oxygen atom has the electron...Ch. 7 - Which of the following electron configurations...Ch. 7 - Which of elements 1-36 have two unpaired electrons...Ch. 7 - Which of elements 136 have one unpaired electron...Ch. 7 - One bit of evidence that the quantum mechanical...Ch. 7 - Identify how many unpaired electrons are present...Ch. 7 - Prob. 105ECh. 7 - Arrange the following groups of atoms in order of...Ch. 7 - Prob. 107ECh. 7 - Arrange the atoms in Exercise 108 in order of...Ch. 7 - In each of the following sets, which atom or ion...Ch. 7 - In each of the following sets, which atom or ion...Ch. 7 - Element 106 has been named seaborgium, Sg, in...Ch. 7 - The first ionization energies of As and Se are...Ch. 7 - Rank the elements Be, B, C, N, and O in order of...Ch. 7 - Consider the following ionization energies for...Ch. 7 - The following graph plots the first, second, and...Ch. 7 - For each of the following pairs of elements (C and...Ch. 7 - For each of the following pairs of elements (Mg...Ch. 7 - The electron affinities of the elements from...Ch. 7 - In the second row of the periodic table, Be, N,...Ch. 7 - Prob. 121ECh. 7 - Order the atoms in each of the following sets from...Ch. 7 - The electron affinity for sulfur is more negative...Ch. 7 - Which has the more negative electron affinity, the...Ch. 7 - Write equations corresponding to the following: a....Ch. 7 - Using data from the text, determine the following...Ch. 7 - Prob. 127ECh. 7 - Cesium was discovered in natural mineral waters in...Ch. 7 - 'The bright yellow light emitted by a sodium vapor...Ch. 7 - Does the information on alkali metals in Table 2-8...Ch. 7 - Predict the atomic number of the next alkali metal...Ch. 7 - Complete and balance the equations for the...Ch. 7 - Prob. 134ECh. 7 - "Lithium" is often prescribed as a...Ch. 7 - A carbon-oxygen double bond in a certain organic...Ch. 7 - Photogray lenses incorporate small amounts of...Ch. 7 - Mars is roughly 60 million km from the earth. How...Ch. 7 - Consider the following approximate visible light...Ch. 7 - One of the visible lines in the hydrogen emission...Ch. 7 - Using Fig. 2-30, list the elements (ignore the...Ch. 7 - Are the following statements true for the hydrogen...Ch. 7 - Although no currently known elements contain...Ch. 7 - Which of the following orbital designations are...Ch. 7 - The four most abundant elements by mass in the...Ch. 7 - Consider the eight most abundant elements in the...Ch. 7 - An ion having a 4+ charge and a mass of 49.9 u has...Ch. 7 - The successive ionization energies for an unknown...Ch. 7 - In the ground state of cadmium, Cd, a. how many...Ch. 7 - Prob. 152CWPCh. 7 - It takes 476 kJ to remove 1 mole of electrons from...Ch. 7 - Calculate, to four significant figures, the...Ch. 7 - Assume that a hydrogen atoms electron bas been...Ch. 7 - Determine the maximum number of electrons that can...Ch. 7 - Consider the ground state of arsenic, As. How many...Ch. 7 - Which of the following statements is(are) true? a....Ch. 7 - Identify the following three elements. a. The...Ch. 7 - For each of the following pairs of elements,...Ch. 7 - Which of the following statements is(are) true? a....Ch. 7 - Three elements have the electron configurations...Ch. 7 - The figure below represents part of the emission...Ch. 7 - One of the emission spectral lines for Be3+ has a...Ch. 7 - The figure below represents part of the emission...Ch. 7 - When lhe excited electron in a hydrogen atom falls...Ch. 7 - Prob. 167CPCh. 7 - For hydrogen atoms, the wave function for the...Ch. 7 - The wave function for the 2pz, orbital in the...Ch. 7 - Answer the following questions, assuming that ms,...Ch. 7 - Assume that we are in another universe with...Ch. 7 - Without looking at data in the text, sketch a...Ch. 7 - The following numbers are the ratios of second...Ch. 7 - We expect the atomic radius to increase going down...Ch. 7 - The ionization energy for a 1s electron in a...Ch. 7 - An atom of a particular element is traveling at...Ch. 7 - As the weapons officer aboard the Srarship...Ch. 7 - Answer the following questions based on the given...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Sketch the intermediates for A,B,C & D.arrow_forwardCan the molecule on the right-hand side of this organic reaction be made in good yield from no more than two reactants, in one step, by moderately heating the reactants? O ? A . If your answer is yes, then draw the reactant or reactants in the drawing area below. You can draw the reactants in any arrangement you like. . If your answer is no, check the box under the drawing area instead. Explanation Check Click and drag to start drawing a structure. ㅇ 80 F5 F6 A 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cente FIGarrow_forwardIn methyl orange preparation, if the reaction started with 0.5 mole of sulfanilic acid to form the diazonium salt of this compound and then it converted to methyl orange [0.2 mole]. If the efficiency of the second step was 50%, Calculate: A. Equation(s) of Methyl Orange synthesis: Diazotization and coupling reactions. B. How much diazonium salt was formed in this reaction? C. The efficiency percentage of the diazotization reaction D. Efficiency percentage of the whole reaction.arrow_forward
- Hand written equations pleasearrow_forwardHand written equations pleasearrow_forward> each pair of substrates below, choose the one that will react faster in a substitution reaction, assuming that: 1. the rate of substitution doesn't depend on nucleophile concentration and 2. the products are a roughly 50/50 mixture of enantiomers. Substrate A Substrate B Faster Rate X Ś CI (Choose one) (Choose one) CI Br Explanation Check Br (Choose one) © 2025 McGraw Hill LLC. All Rights Farrow_forward
- NMR spectrum of ethyl acetate has signals whose chemical shifts are indicated below. Which hydrogen or set of hydrogens corresponds to the signal at 4.1 ppm? Select the single best answer. The H O HỌC—C—0—CH, CH, 2 A ethyl acetate H NMR: 1.3 ppm, 2.0 ppm, 4.1 ppm Check OA B OC ch B C Save For Later Submit Ass © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center |arrow_forwardHow many signals do you expect in the H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red Note for advanced students: In this question, any multiplet is counted as one signal. 1 Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. O ✓ No additional Hs to color in top molecule ง No additional Hs to color in bottom…arrow_forwardin the kinetics experiment, what were the values calculated? Select all that apply.a) equilibrium constantb) pHc) order of reactiond) rate contstantarrow_forward
- true or false, given that a 20.00 mL sample of NaOH took 24.15 mL of 0.141 M HCI to reach the endpoint in a titration, the concentration of the NaOH is 1.17 M.arrow_forwardin the bromothymol blue experiment, pKa was measured. A closely related compound has a Ka of 2.10 x 10-5. What is the pKa?a) 7.1b) 4.7c) 2.0arrow_forwardcalculate the equilibrium concentration of H2 given that K= 0.017 at a constant temperature for this reaction. The inital concentration of HBr is 0.050 M.2HBr(g) ↔ H2(g) + Br2(g)a) 4.48 x 10-2 M b) 5.17 x 10-3 Mc) 1.03 x 10-2 Md) 1.70 x 10-2 Marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY