
- (a) Revisit Example 7-1. What is the error in assuming the concentration of species B is constant and what limits can you put on the calculated value of k? (I.e., k = 0.24 ±?)
- (b) Revisit Example 7-3. Explain why the regression was carried out twice to find k′ and k.
- (c) Revisit Example 7-4. Regress the data to lit the rate law
What is the difference in the correlation and sums of squares compared with those given in Example 7-4? Why was it necessary to regress the data twice, once to obtain Table E7-4.3 and once to obtain Table E7-4.4?
(a)
Interpretation:
The error in assuming the concentration of species B is constant and the limits that can be put over the calculated value of
Concept introduction:
The integral method is the quickest method to use to determine the rate law if the order turns out to zero, first, or second order. In the integral method, we guess the reaction order, α, in the combined batch reactor mole balance and rate law equation.
Integrate the differential equation to obtain the concentration as a function of time. If the order we assume is correct, the appropriate plot of the concentration-time data should be linear. The integral method is used most often when the reaction order is known and it is desired to evaluate the specific reaction rate constant at different temperatures to determine the activation energy.
Answer to Problem 7.1P
The error in assuming the concentration of species B is constant and the limits that can be put over the calculated value of
Explanation of Solution
The given liquid phase reaction which takes place in a batch reactor is as follows.
The initial concentration of Trityl (A) in the feed is
The initial concentration of methanol (B) in the feed is
The temperature of the batch reactor is
The data for time and various concentration of A is given in the table below.
The rate law for the above mentioned reaction is given below.
Where,
The value of
Where,
Substitute
The value of
Substitute
So, the differential rate equation for the equation is given below.
The integration of the above equation with appropriate limits is given below and Substitute
If
Thus, the value of conversion,
| 0 | |||||||
Thus, the graph that can be plotted between
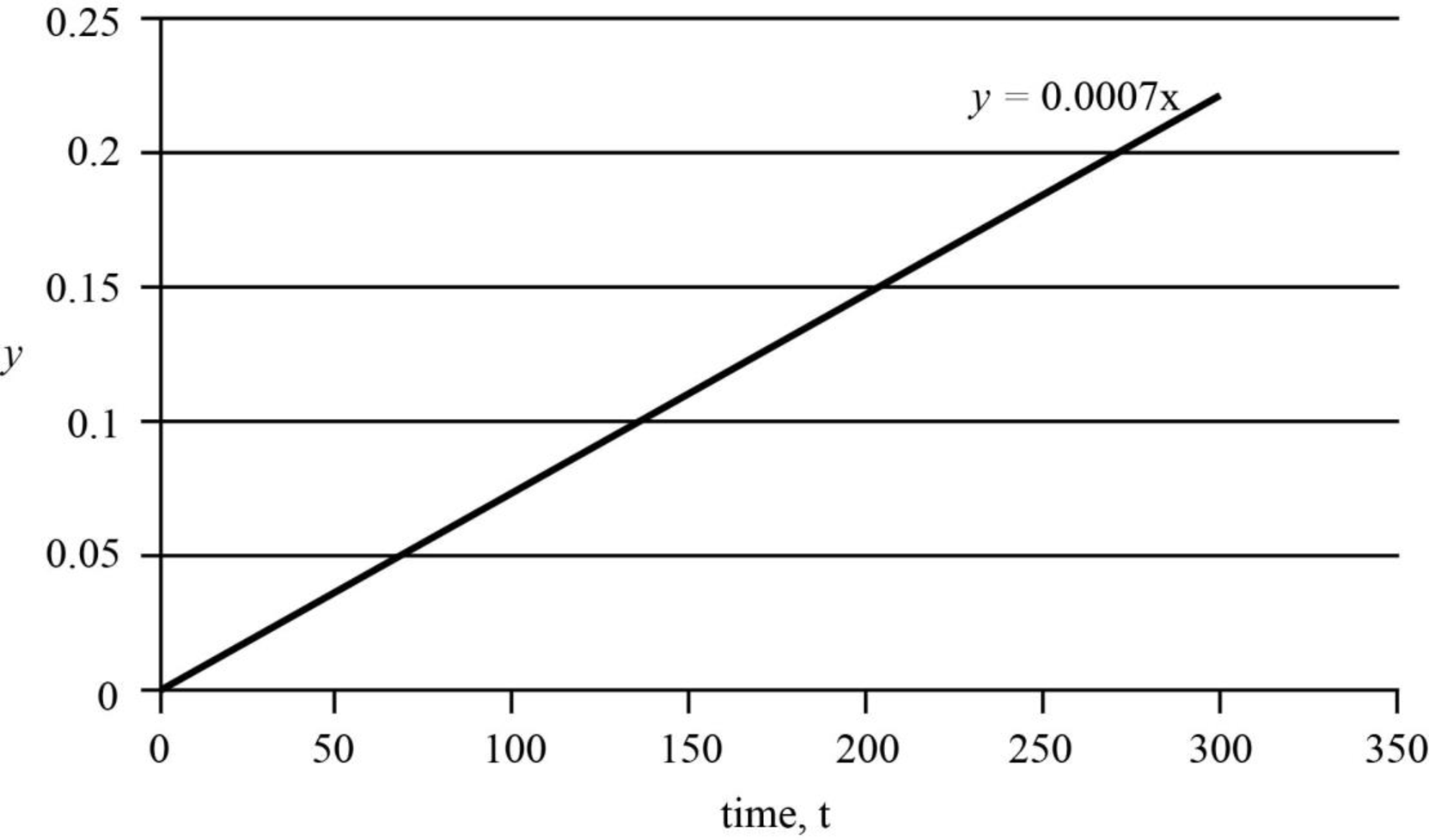
Figure 1
The intercept,
The actual value of
(b)
Interpretation:
The reason as to why regression is carried out twice to find
Concept Introduction:
In nonlinear regression analysis, we search for those parameter values that minimize the sum of the squares of the differences between the measured values and the calculated values for all the data points.
The initial estimates of the parameter values (e.g., reaction order, specific rate constant) in order to calculate the concentration for each data point,
Explanation of Solution
After the first regression, the equation order is predicted
Thus, the value of rate constant can only be calculated at
(c)
Interpretation:
The difference between correlation and sums of square compared with values in the given example is to be stated. The reason as to why regression is carried out twice is to be stated.
Concept Introduction:
In nonlinear regression analysis, we search for those parameter values that minimize the sum of the squares of the differences between the measured values and the calculated values for all the data points.
The initial estimates of the parameter values (e.g., reaction order, specific rate constant) in order to calculate the concentration for each data point,
Explanation of Solution
The given rate law is as follows.
During the first regression, the equation order is predicted as an integer and the corresponding rate constant is also integer. So, the regression is proceeded at the order of
Want to see more full solutions like this?
Chapter 7 Solutions
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Additional Engineering Textbook Solutions
Thermodynamics: An Engineering Approach
Starting Out With Visual Basic (8th Edition)
Starting Out with C++ from Control Structures to Objects (9th Edition)
Starting Out with Java: From Control Structures through Data Structures (4th Edition) (What's New in Computer Science)
Computer Science: An Overview (13th Edition) (What's New in Computer Science)
Starting Out with Python (4th Edition)
- A biodiesel mixture consisting of 60 mol% methyl oleate (MO), 25 mol% methyl linoleate (ML), and 15 mol% methyl palmitate (MP) is held at 373.15 K and 200 MPa. Given PC-SAFT parameters: segment number \( m_i = [5.7, 6.3, 4.8] \), segment diameter \( \sigma_i = [3.95, 3.98, 3.91] \) Å, dispersion energy \( \epsilon_i/k = [260, 270, 250] \) K, and binary interaction parameters \( k_{ij} = 0.01 \), determine the isentropic speed of sound (m/s) using the PC-SAFT Helmholtz energy formulation and the thermodynamic identity\[c^2 = \left( \frac{\partial P}{\partial \rho} \right)_T + \frac{T \left( \frac{\partial P}{\partial T} \right)_\rho^2 }{ \rho^2 c_v },\]assuming the density is precomputed at 200 MPa and \( c_v \) is obtained from ideal mixing of pure-component values.arrow_forwardA steady Williamson nanofluid containing Cu nanoparticles flows over a permeable wedge with wall suction \( V_w = 0.015 \, \text{m/s} \), under a transverse magnetic field \( B_0 = 0.6 \, \text{T} \). The flow obeys the Buongiorno model, with \( D_B = 9 \times 10^{-10} \), \( D_T = 3.5 \times 10^{-8} \), and activation energy \( E_a = 65 \times 10^3 \). Hall and ion-slip effects are included with \( m_e = 0.4 \), \( \beta = 0.15 \). Thermal conductivity varies as \( k(T) = 0.6 (1 + 0.002 (T - 305)) \). Apply velocity and thermal jump conditions with \( \alpha_u = 0.9 \), \( \alpha_T = 0.8 \), \( \lambda = 2 \times 10^{-7} \). Using Keller’s method and similarity variables for wedge parameter \( m = 0.4 \), determine the entropy generation number \( N_s \) at \( x = 0.03 \), where \[N_s = \frac{k(T)}{T_\infty^2} \left( \frac{\partial T}{\partial y} \right)^2 + \frac{\mu}{T_\infty} \left( \frac{\partial u}{\partial y} \right)^2.\]arrow_forwardE. coli was continuously cultured in a continuous stirred tank fermenter with a working volume of 1 L by chemostat. A medium containing 4.0 g/L of glucose as a carbon source was fed to the fermenter at a constant flow rate of 0.5 L/hr, and the glucose concentration in the output stream was 0.20 g/L. The cell yield with respect to glucose was 0.42 g dry cells per gram glucose.arrow_forward
- Please do not use any AI tools to solve this question. I need a fully manual, step-by-step solution with clear explanations, as if it were done by a human tutor. No AI-generated responses, please.arrow_forwardIn the published paper, "Exergy-based Greenhouse gas metric of buildings", use the value of the Exergy Loss of Emission of carbon dioxide to evaluate the index value for 1000 occupants for 50 years building life span in kilogram per person per yeararrow_forwardA CO₂-saturated brine (ionic strength = 2.5 mol/kg, pH = 3, a_H⁺ = 0.01) flows at 2 cm/s through a horizontal tubular reactor (L = 1 m, D = 0.05 m) packed with 5 kg of olivine (Zhuravlev-BET surface area = 30 m²/g). The system operates at 90 °C and 40 bar, and external mass transfer resistance is negligible. The rate-limiting step is electron transfer at the mineral surface, governed by Marcus theory, with λ = 0.75 eV, ΔG° = –0.30 eV, and k₀ = 10⁶ s⁻¹. The Mg²⁺ activity coefficient is γ = 0.76 (from PHREEQC with Pitzer model). For Mg₂SiO₄ + 4 H⁺ → 2 Mg²⁺ + SiO₂(aq) + 2 H₂O, determine the total moles of Mg²⁺ released after 10 minutes at steady state.arrow_forward
- In baseball, batters frequently attempt to hit a ball as far as possible. However, baseballs are inelastic with an officially required "coefficient of restitution" CR ≈ 0.55 on ash wood. The coefficient of restitution of a dropped ball iswhere H is the initial drop height, h is the max. height on the rebound, and h ≈ 2πH tan δ for a homogeneous material. Assuming that a baseball is homogeneous and has a storage modulus approximately the same as that of cork (E' = 18.6 MPa), what must the value of the loss modulus E'' be so that the ball is regulation?arrow_forwardThe creep strain rate of a polymer (in “Hz”) is given by where T is the temperature and Q = 100. kJ/mol is the activation energy. How long t will it take for a rod of this polymer to extend from 10. mm to 15 mm at 100. °C?arrow_forwardCreep compliance J(t) An amorphous polymer has Tg = 100 °C. A creep modulus of 1/J = 1 GPa was measured after t₁ = 1 h at T₁ = 90 °C. Suppose that log 10 a (T) = 17.5(T-Tg) 52+(T-Tg) 1 for this material. What is the shift factor a at T = T₁ relative to the reference Tg? What is the time t₂ required to reach a modulus of 1 GPa at T2 = 80 °C? TR = T = 100 °C |J(t) = 1 GPa-1 + log a(T₁) T₁ = 75 °C T₂ = 50 °C log tr log t₁ log t log t₂ = ?arrow_forward
- A 0.45 mol/kg aqueous solution of 3-(methylamino)propylamine and 1-methylpiperazine (1:1 molar) flows at 0.5 g/s through a 2.5 m horizontal stainless steel coil (inner diameter 1.2 mm), entering at 358.15 K and 22 MPa and exiting at 20 MPa. A constant wall heat flux of 28 W is applied. Local density and isobaric heat capacity are obtained through from the Benedict–Webb–Rubin equation of state, with a 3% increase in heat capacity to account for wall sorption. Dynamic viscosity is obtained using the Green-Kubo relation with a given integral value of 2.1 × 10⁻¹⁰ Pa².s, and thermal conductivity is assumed constant. The local Nusselt number is corrected for thermal development using Nu(x) = 3.66 + (0.065 · Gz(x)^0.7) / (1 + 0.04 · Gz(x)^0.7), where Gz(x) = D · Re(x) · Pr(x) / x. Through a spatially resolved numerical integration of the 1D steady-state energy equation and determine the outlet temperature (K).arrow_forwardA pilot process is being planned to produce antibiotic P. Antibiotic P is a compound secreted by microorganism A during the stationary phase. To produce P, substrate S is required. The growth of microorganism A follows the Monod equation, with a maximum specific growth rate $\mu_m = 1 h^{-1}$ and a half-saturation constant $K_s = 700\ mg/L$.The pilot process uses a chemostat with a working volume of $1000\ L$. In this chemostat, the outflow is processed to separate microorganisms, which are then concentrated tenfold and recycled. A sterile medium containing $15\ g/L$ of substrate is supplied at a flow rate of $100\ L/h$, while the recycled flow (concentrated) contains $5\ g/L$ and is also fed into the chemostat.Microorganism A yields $0.5\ g$ of biomass per $1\ g$ of substrate consumed $(Y^M_{X/S} = 0.5\ g\ A/g\ S)$, and its death rate $(k_d)$ is negligible. Additionally, $1\ g$ of microorganism A produces $0.05\ g$ of antibiotic P per hour $(q_P = 0.05 g\ P/h\cdot g\ A)$, and $1\ g$…arrow_forwardIn the production of ethyl acetate via reactive distillation, the column operates at 5 bar with an equimolar feed (ethanol + acetic acid) at 80°C. The reaction follows: \[CH_3COOH + C_2H_5OH \rightleftharpoons CH_3COOC_2H_5 + H_2O \quad (K_{eq} = 4.2 \text{ at } 80°C)\] Given: - NRTL parameters for all binary pairs - Tray efficiency = 65% - Vapor-liquid equilibrium exhibits positive azeotrope formation Calculate the exact minimum reflux ratio required to achieve 98% ethyl acetate purity in the distillate, assuming: 1) The reaction reaches equilibrium on each tray 2) The heavy key component is waterarrow_forward
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





