
ORGANIC CHEMISTRY-NEXTGEN+BOX (2 SEM.)
4th Edition
ISBN: 9781119761068
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.6, Problem 6.53P
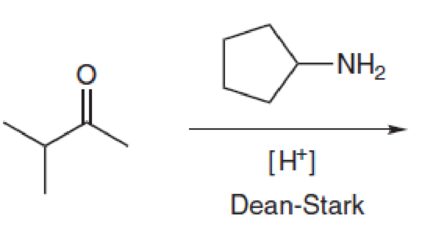
Predict the product(s) for each of the following transformations:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Explain why the S-F bond strength is 367 kJ/mol in SF2 and 329 kJ/mol in SF6.
Would Si(CH3)3F react with AgCl? If so, write out the balanced chemical equation. If not,explain why no reaction would take place.
NH3 reacts with boron halides (BX3 where X = F, Cl, Br, or I) to form H3N-BX3 complexes.Which of these complexes will have the strongest N-B bond? Justify your answer
Chapter 6 Solutions
ORGANIC CHEMISTRY-NEXTGEN+BOX (2 SEM.)
Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Predict the major product for each of the...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...Ch. 6.1 - Identify the reagents you would use to achieve...
Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Predict the major product of each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.3 - Propose a mechanism for each of the following...Ch. 6.4 - Prob. 6.24PCh. 6.4 - Prob. 6.25PCh. 6.4 - Prob. 6.26PCh. 6.4 - Prob. 6.27PCh. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.4 - Predict the major product of each of the following...Ch. 6.5 - Prob. 6.33PCh. 6.5 - Prob. 6.34PCh. 6.5 - Prob. 6.35PCh. 6.5 - Predict the major product that is expected when...Ch. 6.5 - Prob. 6.37PCh. 6.5 - Predict the major product that is expected when...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.5 - Identify the reagents you would use to achieve...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.6 - Predict the product(s) for each of the following...Ch. 6.7 - Prob. 6.62PCh. 6.7 - Prob. 6.63PCh. 6.7 - Prob. 6.64PCh. 6.7 - Prob. 6.65PCh. 6.7 - Prob. 6.67PCh. 6.7 - Prob. 6.68PCh. 6.7 - Prob. 6.69PCh. 6.7 - Predict the major product for each of the...Ch. 6.7 - Predict the major product for each of the...Ch. 6.7 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.8 - Predict the major product for each of the...Ch. 6.9 - Prob. 6.81PCh. 6.9 - Prob. 6.82PCh. 6.9 - Prob. 6.83PCh. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - What reagents would you use to achieve the...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...Ch. 6.9 - Propose an efficient synthesis for the following...
Additional Science Textbook Solutions
Find more solutions based on key concepts
All of the following terms can appropriately describe humans except: a. primary consumer b. autotroph c. hetero...
Human Biology: Concepts and Current Issues (8th Edition)
WHAT IF? Is allopatric speciation more likely to occur on an island close to a mainland or on a more isolated i...
Campbell Biology in Focus (2nd Edition)
Compare each of the mechanisms listed here with the mechanism for each of the two parts of the acid-catalyzed h...
Organic Chemistry (8th Edition)
Explain all answers clearly, with complete sentences and proper essay structure if needed. An asterisk (*) desi...
Cosmic Perspective Fundamentals
In one public health study, replica plating was used to screen 131 fecal samples for gram-negative bacteria tha...
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
Which elements would you expect to lose electrons in chemical changes? a. potassium b. sulfur c. fluorine d. ba...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Synthesize the following:arrow_forwardDid you report your data to the correct number of significant figures? Temperature of cold water (°C) 4.0 Temperature of hot water ("C) 87.0 Volume of cold water (mL) 94.0 Volume of hot water (mL) 78.0 Final temperature after mixing ("C) 41.0 Mass of cold water (g) 94.0 Mass of hot water (g) 78.0 Calorimeter constant (J/°C) 12.44 How to calculate the calorimeter constantarrow_forwardplease draw the arrowsarrow_forward
- Part 1. Draw monomer units of the following products and draw their reaction mechanism (with arrow pushing) Temporary cross-linked polymer Using: 4% polyvinyl alcohol+ methyl red + 4% sodium boratearrow_forwardcan you please answer both these questions and draw the neccesaryarrow_forwardcan you please give the answer for both these pictures. thankyouarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License