
Concept explainers
(a)
To determine: The value of Vmax and Km of enzyme prostaglandin endoperoxide synthase.
Introduction:
Prostaglandin is class of lipid which is present at a site of injury and tissue damage in body. It is involved in healing process and induces inflammation, and initiation of pain.
(a)
Explanation of Solution
Pictorial representation:
Table 1 shows the rate of formation of prostaglandin from arachidonic acid and Fig.1 shows the Lineweaver Burk plot, a double reciprocal plotting for 1/V0 Vs. 1/[S].
Table 1
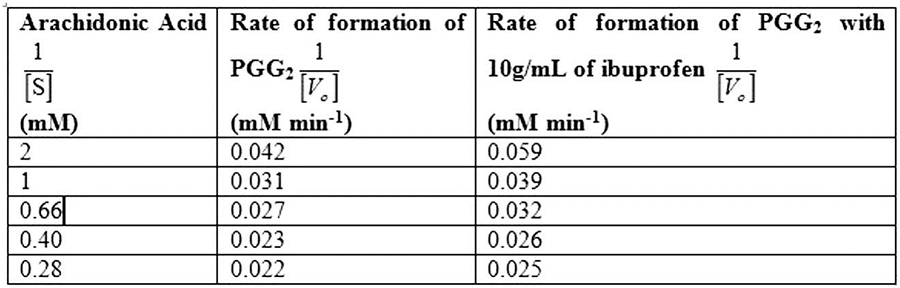
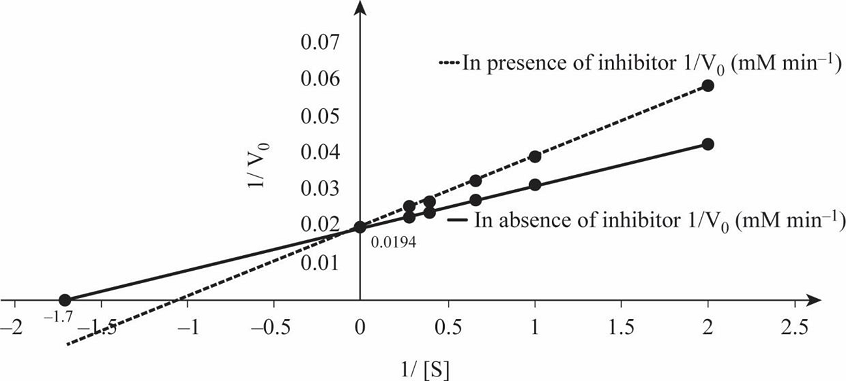
Fig.1: Lineweaver Burk plot.
Lineweaver-Burk equation is the reciprocal of Michaelis-Menten equation, and given as:
Michaelis-Menten equation
Lineweaver-Burk equation
So, by reciprocating the values given in first and second columns of the given table, we get 1/[S] and 1/V0 values in absence of the inhibitor, as mentioned in Table 1. By plotting these values we obtain the Lineweaver-Burk graph depicted in Fig.1. From the graph, calculating the V max in the absence of inhibitor:
Calculating the Km in the absence of inhibitor:
The Vmax of enzyme in the absence of inhibitor is 51.5mM/min, while Km of enzyme in the absence of inhibitor is 0.59mM.
(b)
To determine: The type of inhibition that ibuprofen exerts on prostaglandin endoperoxide synthase.
Introduction:
Enzyme inhibitors are defined as chemical molecules that bind at active site of enzymes and prevent the binding of substrate with enzyme. There are two types of inhibitors such as reversible and irreversible inhibitors.
(b)
Explanation of Solution
Lineweaver-Burk equation is the reciprocal of Michaelis-Menten equation is given as:
Michaelis-Menten equation
Lineweaver-Burk equation
So, by reciprocating the given values in column first and third, we get the rate of formation of prostaglandin from arachidonic acid in presence of inhibitor ibuprofen, as mentioned in Table 1. By plotting these values we obtain the Lineweaver-Burk graph for 1/[S] and 1/V0 in presence of inhibitor, as depicted in Fig.1.
Calculating the Vmax in presence of inhibitor:
Vmax in the presence of inhibitor:
Km of enzyme in the presence of inhibitor:
The Vmax of enzyme both in the presence and absence of inhibitor is 51.54 mM/min, while Km of enzyme in the presence and absence of inhibitor is 0.83mM and 0.59mM.
Prostaglandin is involved in initiation of pain, and it is synthesized by prostaglandin endoperoxide synthase. Ibuprofen inhibits the activity of this enzyme by binding at the active site and preventing binding of substrate with enzyme. The double reciprocal graph in Fig.1 shows when competitive inhibitor ibuprofen is present, the Vmax remains unchanged while Km increases. Thus, -1/Km value is closer to the origin in the graph depicted in Fig.1. In competitive inhibition, Vmax remains unchanged while Km increases. Therefore, ibuprofen is a competitive inhibitor of prostaglandin.
The inhibition of prostaglandin by ibuprofen is example of competitive inhibition.
Want to see more full solutions like this?
Chapter 6 Solutions
EBK LEHNINGER PRINCIPLES OF BIOCHEMISTR
- The following data were recorded for the enzyme catalyzed conversion of S -> P. Question: Estimate the Vmax and Km. What would be the rate at 2.5 and 5.0 x 10-5 M [S] ?arrow_forwardPlease helparrow_forwardThe following data were recorded for the enzyme catalyzed conversion of S -> P Question: what would the rate be at 5.0 x 10-5 M [S] and the enzyme concentration was doubled? Also, the rate given in the table is from product accumulation after 10 minuets of reaction time. Verify these rates represent a true initial rate (less than 5% turnover). Please helparrow_forward
- The following data was obtained on isocitrate lyase from an algal species. Identify the reaction catalyzed by this enzyme, deduce the KM and Vmax , and determine the nature of the inhibition by oxaloacetate. Please helparrow_forwardIn the table below, there are sketches of four crystals made of positively-charged cations and negatively-charged anions. Rank these crystals in decreasing order of stability (or equivalently increasing order of energy). That is, select "1" below the most stable (lowest energy) crystal. Select "2" below the next most stable (next lowest energy) crystal, and so forth. A B 鹽 (Choose one) +2 C +2 +2 (Choose one) D 鹽雞 (Choose one) (Choose one)arrow_forward1. Draw the structures for the fats A. 16:2: w-3 and B. 18:3:49,12,15 2. Name each of the molecules below (image attached)arrow_forward
- draw the structures for the fats A. 16:2:w-3 B 18:3:9,12,15arrow_forward1. Below is a template strand of DNA. Show the mRNA and protein that would result. label the ends of the molecules ( refer to attached image)arrow_forwardAttach the followina labels to the diagram below: helicase, single stranded binding proteins, lagging strand, leading strand, DNA polymerase, primase, 5' ends (3), 3' ends (3) (image attached)arrow_forward
- 1. How much energy in terms of ATP can be obtained from tristearin (stearate is 18:0) Show steps pleasearrow_forwardMultiple choice urgent!!arrow_forward1. Write the transamination reaction for alanine. Indicate what happens next to each of the molecules in the reaction, and under what conditions it happens. 2.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





