
Human Physiology
15th Edition
ISBN: 9781259864629
Author: Fox, Stuart Ira
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 24RA
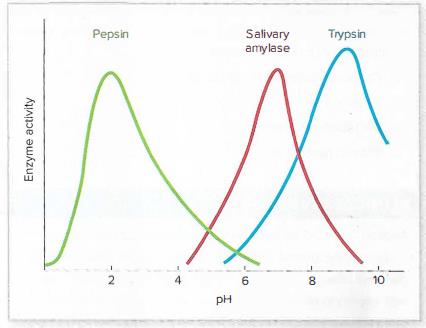
Use the graph here and in figure 4.4 to answer the following questions:
What two pH values produce half-maximal activity of salivary amylase?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An urgent care center experienced the average patient admissions shown in the Table below during the weeks from the first week of December through the second week of April.
Week
Average Daily Admissions
1-Dec
11
2-Dec
14
3-Dec
17
4-Dec
15
1-Jan
12
2-Jan
11
3-Jan
9
4-Jan
9
1-Feb
12
2-Feb
8
3-Feb
13
4-Feb
11
1-Mar
15
2-Mar
17
3-Mar
14
4-Mar
19
5-Mar
13
1-Apr
17
2-Apr
13
Forecast admissions for the periods from the first week of December through the second week of April. Compare the forecast admissions to the actual admissions; What do you conclude?
Analyze the effectiveness of the a drug treatment program based on the needs of 18-65 year olds who are in need of treatment by critically describing 4 things in the program is doing effectively and 4 things the program needs some improvement.
I have the first half finished... just need the bottom half.
Chapter 4 Solutions
Human Physiology
Ch. 4 - Use the lock-and-key model to explain how enzymes...Ch. 4 - Explain how enzymes are named, and the nature of...Ch. 4 - Draw graphs to represent the effects of changes in...Ch. 4 - Prob. 4CPCh. 4 - Prob. 5aCPCh. 4 - Prob. 5bCPCh. 4 - Prob. 5cCPCh. 4 - Prob. 6aCPCh. 4 - Define the terms exergonic reaction and endergonic...Ch. 4 - Prob. 7aCP
Ch. 4 - Prob. 7bCPCh. 4 - Which of these statements about enzymes is...Ch. 4 - Which of these statements about enzyme-catalyzed...Ch. 4 - Which of these statements about lactate...Ch. 4 - In a metabolic pathway, a. the product of one...Ch. 4 - In an inborn error of metabolism,
a. a genetic...Ch. 4 - Which of these represents an endergonic...Ch. 4 - Which of these statements about ATP is true? a....Ch. 4 - When oxygen is combined with 2 hydrogens to make...Ch. 4 - Enzymes increase the rate of chemical reactions...Ch. 4 - According to the law of mass action, which of...Ch. 4 - Explain the relationship between an enzyme's...Ch. 4 - Explain how the rate of enzymatic reactions may be...Ch. 4 - Explain how end-product inhibition represents a...Ch. 4 - Prob. 14RACh. 4 - The coenzymes NAD and FAD can "shuttle" hydrogens...Ch. 4 - Prob. 16RACh. 4 - Why do we need to eat food containing niacin and...Ch. 4 - Metabolic pathways can be likened to intersecting...Ch. 4 - Prob. 19RACh. 4 - Suppose you come across a bottle of enzyme tablets...Ch. 4 - Describe the energy transformations that occur...Ch. 4 - Use the reversible reactions involving the...Ch. 4 - Use the graph here and in figure 4.4 to answer the...Ch. 4 - Use the graph here and in figure 4.4 to answer the...Ch. 4 - Prob. 25RACh. 4 - Prob. 26RACh. 4 - Prob. 27RA
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- 13. Practice Calculations: 3 colonies were suspended in the following dilution series and then a viable plate count and microscope count was performed. Calculate IDF's, TDF's and then calculate the CFU/mL in each tube by both methods. Finally calculate the cells in 1 colony by both methods. Show all of your calculations in the space provided on the following pages. 3 colonies 56 cells 10 μL 10 μL 100 μL 500 με m OS A B D 5.0 mL 990 με 990 με 900 με 500 μL EN 2 100 με 100 μL 118 colonies 12 coloniesarrow_forwardDescribe and give a specific example of how successionary stage is related to species diversity?arrow_forwardExplain down bellow what happens to the cell in pictures not in words: Decreased pH in mitochondria Increased ATP Decreased pH in cytosol Increased hydrolysis Decreasing glycogen and triglycerides Increased MAP kinase activity Poor ion transport → For each one:→ What normally happens?→ What is wrong now?→ How does it mess up the cell?arrow_forward
- 1.) Community Diversity: The brown and orange line represent two different plant communities. a. Which color represents the community with a higher species richness? b. Which color represents the community with a higher species evenness? Relative abundance 0.1 0.04 0.001 2 4 6 8 10 12 14 16 18 20 22 24 Rank abundance c. What is the maximum value of the Simpson's diversity index (remember, Simpson's index is D = p², Simpson's diversity index is 1-D)? d. If the Simpson's diversity index equals 1, what does that mean about the number of species and their relative abundance within community being assessed?arrow_forward1.) Community Diversity: The brown and orange line represent two different plant communities. a. Which color represents the community with a higher species richness? b. Which color represents the community with a higher species evenness? Relative abundance 0.1 0.04 0.001 2 4 6 8 10 12 14 16 18 20 22 24 Rank abundance c. What is the maximum value of the Simpson's diversity index (remember, Simpson's index is D = p², Simpson's diversity index is 1-D)? d. If the Simpson's diversity index equals 1, what does that mean about the number of species and their relative abundance within community being assessed?arrow_forwardwhat measures can a mother to take to improve the produce of her to milk to her newborn baby ?arrow_forward
- 1. Color the line that represents all ancestors of the Eastern white pine tree green (but only the ancestral line NOT shared with other organisms) 2. Oncle the last common ancestor of the Colorado blue spruce tree and Eastern white pine tree. 3. Put a box around the last common ancestor of the sugar maple tree and the dogwood tree. 4. Put a triangle around the last common ancestor of the red pine tree and the american holly bush. 5. Color the line that represents all ancestors of the Ponderosa pine tree red (including all shared ancestors). 6. Color the line that represents all ancestors of the American elm tree blue (including all shared ancestors). 7 Color the line that represents all ancestors of the Sabal palm tree purple (including all shared ancestors) 8. Using a yellow highlighter or colored pencil, circle the clade that includes all pine trees. 9. Using a orange highlighter or colored pencil, circle the clade that includes all gymnosperms 10. Can you tell…arrow_forwardYou have been hired as a public relations specialist to give invertebrates a good name. After all, they are much more than just creepy crawly bugs! Your first task though is to convince yourself that is true. The best way to do that is to start close to home. Find something in your house that is a product obtained directly from an invertebrate or only due to an invertebrate’s actions. Describe the product, its function and utility, as well as any human manufactured alternatives. Be sure to highlight the advantages of obtaining this directly from nature. Keep in mind, a product can be something you use, wear, eat, or enjoy for its visual appeal.arrow_forwardUse the following tree diagram to answer Questions #8-10. 8) Which of the following two animals are the most closely related based on the tree to the left? a) Pig and camel b) Hippo and pig c) Deer and cow 9) CIRCLE on the tree diagram where the common ancestor between a hippo and a cow is. 10) Put a SQUARE on the tree diagram where the common ancestor between a pig and a peccary is.arrow_forward
- Explain: Healthy Cell Function Overview→ Briefly describe how a healthy cell usually works: metabolism (ATP production), pH balance, glycogen storage, ion transport, enzymes, etc. Gene Mutation and Genetics Part→ Focus on the autosomal recessive mutation and explain: How gene mutation affects the cell. How autosomal inheritance works. Compare the normal and mutated gene sequences simply. → Talk about possible consequences of a faulty hydrolytic enzyme.arrow_forwardCan you fill out those termsarrow_forwardExplain down bellow what happens to the cell: Decreased pH in mitochondria Increased ATP Decreased pH in cytosol Increased hydrolysis Decreasing glycogen and triglycerides Increased MAP kinase activity Poor ion transport → For each one:→ What normally happens?→ What is wrong now?→ How does it mess up the cell?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning


Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning


Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning

Nutrition and Diet - GCSE Biology (9-1); Author: Mr Exham Biology;https://www.youtube.com/watch?v=SFE1DfAlipo;License: Standard Youtube License