
CHEMISTRY MOLECULAR NATURE OF MATTER AND
9th Edition
ISBN: 9781266568718
Author: SILBERBERG
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.3, Problem 3.11AFP
Interpretation Introduction
Interpretation:
The balanced equation is to be interpreted for the given atmospheric reaction.
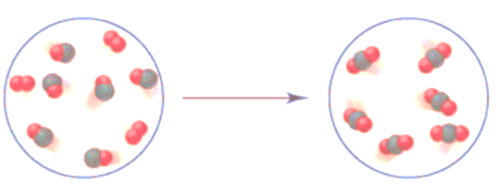
Concept Introduction:
A
The chemical reaction can be symbolized with the help of chemical equation. It involves the chemical formula of the reactant and product molecules that shows the conversion by single headed arrows.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Assign these C-NMR and H-NMR Spectrum
Predict the product of this organic reaction:
IZ
+
HO
i
P+H₂O
Specifically, in the drawing area below draw the skeletal ("line") structure of P.
If there is no reasonable possibility for P, check the No answer box under the drawing area.
No Answer
Click and drag to start drawing a
structure.
☐ :
Predict the products of this organic reaction:
0
O
-----
A
+ KOH ?
CH3-CH2-C-O-CH2-C-CH3
Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them
in any arrangement you like, so long as they aren't touching.)
If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area.
No reaction
Click anywhere to draw the first
atom of your structure.
X
⑤
è
Chapter 3 Solutions
CHEMISTRY MOLECULAR NATURE OF MATTER AND
Ch. 3.1 - Graphite is the crystalline form of carbon used in...Ch. 3.1 - Prob. 3.1BFPCh. 3.1 - At rest, a person inhales 9.72×1021 nitrogen...Ch. 3.1 - Prob. 3.2BFPCh. 3.1 - Prob. 3.3AFPCh. 3.1 - Prob. 3.3BFPCh. 3.1 - Tetraphosphorus decoxide reacts with water to form...Ch. 3.1 - Prob. 3.4BFPCh. 3.1 - Prob. 3.5AFPCh. 3.1 - For many years, compounds known as...
Ch. 3.1 - Use the information in Follow-up Problem 3.6A to...Ch. 3.1 - Prob. 3.6BFPCh. 3.2 - Prob. 3.7AFPCh. 3.2 - Prob. 3.7BFPCh. 3.2 - Prob. 3.8AFPCh. 3.2 - Prob. 3.8BFPCh. 3.2 - Prob. 3.9AFPCh. 3.2 - Prob. 3.9BFPCh. 3.3 - Prob. 3.10AFPCh. 3.3 - Prob. 3.10BFPCh. 3.3 - Prob. 3.11AFPCh. 3.3 - Prob. 3.11BFPCh. 3.4 - Prob. 3.12AFPCh. 3.4 - Prob. 3.12BFPCh. 3.4 - Prob. 3.13AFPCh. 3.4 - Prob. 3.13BFPCh. 3.4 - Prob. 3.14AFPCh. 3.4 - Prob. 3.14BFPCh. 3.4 - Prob. 3.15AFPCh. 3.4 - Prob. 3.15BFPCh. 3.4 - Prob. 3.16AFPCh. 3.4 - Prob. 3.16BFPCh. 3.4 - Prob. 3.17AFPCh. 3.4 - Prob. 3.17BFPCh. 3.4 - Prob. 3.18AFPCh. 3.4 - Prob. 3.18BFPCh. 3 - Prob. 3.1PCh. 3 - Prob. 3.2PCh. 3 - Why might the expression “1 mol of chlorine” be...Ch. 3 - Prob. 3.4PCh. 3 - Prob. 3.5PCh. 3 - Prob. 3.6PCh. 3 - Prob. 3.7PCh. 3 - Prob. 3.8PCh. 3 - Calculate the molar mass of each of the...Ch. 3 - Prob. 3.10PCh. 3 - Prob. 3.11PCh. 3 - Calculate each of the following quantities:
Mass...Ch. 3 - Calculate each of the following quantities:
Amount...Ch. 3 - Prob. 3.14PCh. 3 - Prob. 3.15PCh. 3 - Prob. 3.16PCh. 3 - Prob. 3.17PCh. 3 - Prob. 3.18PCh. 3 - Prob. 3.19PCh. 3 - Calculate each of the following:
Mass % of H in...Ch. 3 - Calculate each of the following:
Mass % of I in...Ch. 3 - Calculate each of the following:
Mass fraction of...Ch. 3 - Calculate each of the following:
Mass fraction of...Ch. 3 - Oxygen is required for the metabolic combustion of...Ch. 3 - Cisplatin (right), or Platinol, is used in the...Ch. 3 - Allyl sulfide (below) gives garlic its...Ch. 3 - Iron reacts slowly with oxygen and water to form a...Ch. 3 - Prob. 3.28PCh. 3 - Prob. 3.29PCh. 3 - The mineral galena is composed of lead(II) sulfide...Ch. 3 - Prob. 3.31PCh. 3 - Prob. 3.32PCh. 3 - List three ways compositional data may be given in...Ch. 3 - Prob. 3.34PCh. 3 - Prob. 3.35PCh. 3 - Prob. 3.36PCh. 3 - Prob. 3.37PCh. 3 - Prob. 3.38PCh. 3 - Prob. 3.39PCh. 3 - What is the molecular formula of each...Ch. 3 - Prob. 3.41PCh. 3 - Prob. 3.42PCh. 3 - Find the empirical formula of each of the...Ch. 3 - An oxide of nitrogen contains 30.45 mass % N. (a)...Ch. 3 - Prob. 3.45PCh. 3 - A sample of 0.600 mol of a metal M reacts...Ch. 3 - Prob. 3.47PCh. 3 - Prob. 3.48PCh. 3 - Prob. 3.49PCh. 3 - Prob. 3.50PCh. 3 - Prob. 3.51PCh. 3 - Prob. 3.52PCh. 3 - Prob. 3.53PCh. 3 - Prob. 3.54PCh. 3 - Prob. 3.55PCh. 3 - Prob. 3.56PCh. 3 - Prob. 3.57PCh. 3 - Prob. 3.58PCh. 3 - Prob. 3.59PCh. 3 - Prob. 3.60PCh. 3 - Prob. 3.61PCh. 3 - Prob. 3.62PCh. 3 - Prob. 3.63PCh. 3 - Prob. 3.64PCh. 3 - Prob. 3.65PCh. 3 - Prob. 3.66PCh. 3 - Prob. 3.67PCh. 3 - Prob. 3.68PCh. 3 - Prob. 3.69PCh. 3 - Prob. 3.70PCh. 3 - Prob. 3.71PCh. 3 - Prob. 3.72PCh. 3 - Prob. 3.73PCh. 3 - Prob. 3.74PCh. 3 - Elemental phosphorus occurs as tetratomic...Ch. 3 - Prob. 3.76PCh. 3 - Solid iodine trichloride is prepared in two steps:...Ch. 3 - Prob. 3.78PCh. 3 - Prob. 3.79PCh. 3 - Prob. 3.80PCh. 3 - Prob. 3.81PCh. 3 - Prob. 3.82PCh. 3 - Prob. 3.83PCh. 3 - Prob. 3.84PCh. 3 - Prob. 3.85PCh. 3 - Prob. 3.86PCh. 3 - Prob. 3.87PCh. 3 - Prob. 3.88PCh. 3 - Prob. 3.89PCh. 3 - Prob. 3.90PCh. 3 - Prob. 3.91PCh. 3 - Prob. 3.92PCh. 3 - Prob. 3.93PCh. 3 - Prob. 3.94PCh. 3 - When 20.5 g of methane and 45.0 g of chlorine gas...Ch. 3 - Prob. 3.96PCh. 3 - Prob. 3.97PCh. 3 - Prob. 3.98PCh. 3 - Prob. 3.99PCh. 3 - Prob. 3.100PCh. 3 - Sodium borohydride (NaBH4) is used industrially in...Ch. 3 - Prob. 3.102PCh. 3 - The first sulfur-nitrogen compound was prepared in...Ch. 3 - Prob. 3.104PCh. 3 - Prob. 3.105PCh. 3 - Prob. 3.106PCh. 3 - Serotonin () transmits nerve impulses between...Ch. 3 - In 1961, scientists agreed that the atomic mass...Ch. 3 - Prob. 3.109PCh. 3 - Isobutylene is a hydrocarbon used in the...Ch. 3 - The multistep smelting of ferric oxide to form...Ch. 3 - Prob. 3.112PCh. 3 - Prob. 3.113PCh. 3 - Prob. 3.114PCh. 3 - Prob. 3.115PCh. 3 - Prob. 3.116PCh. 3 - Prob. 3.117PCh. 3 - Prob. 3.118PCh. 3 - Prob. 3.119PCh. 3 - Prob. 3.120PCh. 3 - For the reaction between solid tetraphosphorus...Ch. 3 - Prob. 3.122PCh. 3 - Prob. 3.123PCh. 3 - Prob. 3.124PCh. 3 - Prob. 3.125PCh. 3 - Prob. 3.126PCh. 3 - Prob. 3.127PCh. 3 - Prob. 3.128PCh. 3 - Ferrocene, synthesized in 1951, was the first...Ch. 3 - Prob. 3.130PCh. 3 - Prob. 3.131PCh. 3 - Citric acid (below) is concentrated in citrus...Ch. 3 - Prob. 3.133PCh. 3 - Nitrogen monoxide reacts with elemental oxygen to...Ch. 3 - Prob. 3.135PCh. 3 - Prob. 3.136PCh. 3 - Manganese is a key component of extremely hard...Ch. 3 - The human body excretes nitrogen in the form of...Ch. 3 - Aspirin (acetylsalicylic acid, C9H8O4) is made by...Ch. 3 - Prob. 3.140PCh. 3 - Prob. 3.141PCh. 3 - Prob. 3.142PCh. 3 - When powdered zinc is heated with sulfur, a...Ch. 3 - Cocaine (C17H21O4N) is a natural substance found...Ch. 3 - Prob. 3.145P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the products of this organic reaction: O CH3 + H2O + HCI A A? CH3-CH2-C-N-CH3 Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. If there's more than one product, draw them in any arrangement you like, so long as they aren't touching. If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No Reaction Click anywhere to draw the first atom of your structure.arrow_forwardWhat is the missing reactant in this organic reaction? R+ HO-C-CH2-CH3 0= CH3 CH3 —CH, C−NH—CH CH3 + H₂O Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. Note for advanced students: you may assume no products other than those shown above are formed. No Answer Click anywhere to draw the first atom of your structure. €arrow_forward个 CHEM&131 9267 - $25 - Intro to Mail - Hutchison, Allison (Student x Aktiv Learnin https://app.aktiv.com Draw the product of the reaction shown below. Ignore inorganic byproducts. + Na2Cr2O7 Acetone, H2SO4 Type here to search Dryng OH W Prarrow_forward
- Predict the products of this organic reaction: OH + NaOH A? Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click and drag to start drawing a structure. ✓ Sarrow_forwardPredict the products of this organic reaction: CH3-C-O-CH2-CH2-C-CH3 + H₂O ? A Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No reaction Click anywhere to draw the first atom of your structure. :☐ darrow_forwardDE d. Draw an arrow pushing mechanism for the following IN O CI N fo 人 P Polle DELL prt sc home end ins F5 F6 F7 F8 F9 F10 F11 F12arrow_forward
- Predict the products of this organic reaction: + H₂O H* ? A Specifically, in the drawing area below draw the skeletal ("line") structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No Reaction Click and drag to start drawing a structure.arrow_forwardPredict the major organic products of the reaction below and draw them on right side of the arrow. If there will be no significant reaction, check the box below the drawing area instead. C Cl CH, OH There will be no significant reaction. + pyridine G Click and drag to start drawing a structure.arrow_forwardWhat is the missing reactant in this organic reaction? H R+ H2O Δ OH 0= CH3-CH-O-CH3 + CH3-C-OH Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answer box under the drawing area. No Answer Click anywhere to draw the first atom of your structure. dyarrow_forward
- You are trying to determine whether the following organic reaction can be done in a single synthesis step. If so, add any missing reagents or conditions in the drawing area below. If it isn't possible to do this reaction in a single synthesis step, check the box below the drawing area instead. Note for advanced students: if you have a choice of reagents to add, you should choose the least reactive and most economical reagents possible. Cl It isn't possible to do this reaction in a single synthesis step. + T OHarrow_forwardPredict the products of this organic reaction: CH3 O CH3-CH-C-O-CH2-CH2-CH3 + H₂OH+ Η ? A Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. No Reaction Click anywhere to draw the first atom of your structure.arrow_forward€ CH3-CH-C-O-CH2-CH2-CH3 + NaOH A? Specifically, in the drawing area below draw the condensed structure of the product, or products, of this reaction. (If there's more than one product, draw them in any arrangement you like, so long as they aren't touching.) If there aren't any products because this reaction won't happen, check the No reaction box under the drawing area. Predict the products of this organic reaction: CH3 O Click anywhere to draw the first atom of your structure. No reaction ✓ Garrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY