![WebAssign for Zumdahl's Chemical Principles, 8th Edition [Instant Access], Single-Term](https://s3.amazonaws.com/compass-isbn-assets/textbook_empty_images/large_textbook_empty.svg)
WebAssign for Zumdahl's Chemical Principles, 8th Edition [Instant Access], Single-Term
8th Edition
ISBN: 9780357119112
Author: Zumdahl; Steven S.
Publisher: Cengage Learning US
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 50E
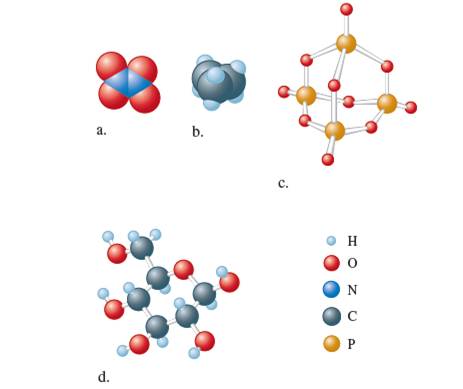
Give the empirical formula of each of these compounds.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer
below.
Η
H's+
6Η Η
Η
Η
Η
Ηδ
Η
Ο
Ο
HH
+Η Η
+Η Η
Η
-8+
CI
H
H:O::::H
H
H
HH
H::O:D:D:H
HH
HH
H:O:D:D:H
..
HH
H:O:D:D:H
H H
Select the correct Lewis dot structure for the following compound:
CH3CH2OH
Rank the following compounds in order of decreasing boiling point.
ннннн
-С-С-Н
.
н-с-
ННННН
H
ΗΤΗ
НННН
TTTĪ
н-с-с-с-с-о-н
НННН
НН
C' Н
н-с-с-с-с-н
НН
||
Ш
НННН
H-C-C-C-C-N-H
ННННН
IV
Chapter 3 Solutions
WebAssign for Zumdahl's Chemical Principles, 8th Edition [Instant Access], Single-Term
Ch. 3 - Prob. 1DQCh. 3 - Consider the equation A+2BAB2 . Imagine that10...Ch. 3 - Prob. 3DQCh. 3 - Prob. 4DQCh. 3 - Prob. 5DQCh. 3 - For the preceding question, which of the following...Ch. 3 - Prob. 7DQCh. 3 - A kerosene lamp has a mass of 1.5 kg. You put 0.5...Ch. 3 - Prob. 9DQCh. 3 - You may have noticed that water sometimes drips...
Ch. 3 - Prob. 11DQCh. 3 - Prob. 12DQCh. 3 - Prob. 13DQCh. 3 - Atoms of three different elements are represented...Ch. 3 - Prob. 15DQCh. 3 - Prob. 16DQCh. 3 - Prob. 17DQCh. 3 - Prob. 18DQCh. 3 - Chlorine exists mainly as two isotopes,...Ch. 3 - According to the law of conservation of mass, mass...Ch. 3 - Prob. 21DQCh. 3 - Prob. 22DQCh. 3 - Prob. 23ECh. 3 - Prob. 24ECh. 3 - Prob. 25ECh. 3 - Prob. 26ECh. 3 - Prob. 27ECh. 3 - An element consists of 1.40% of an isotope with...Ch. 3 - Prob. 29ECh. 3 - Naturally occurring tellurium (Te) has the...Ch. 3 - Gallium arsenide (GaAs) has gained widespread use...Ch. 3 - Prob. 32ECh. 3 - Prob. 33ECh. 3 - Prob. 34ECh. 3 - Prob. 35ECh. 3 - How many atoms of nitrogen are present in 5.00 g...Ch. 3 - Consider the following gas samples: 4.0 g of...Ch. 3 - Prob. 38ECh. 3 - Prob. 39ECh. 3 - Prob. 40ECh. 3 - Prob. 41ECh. 3 - Prob. 42ECh. 3 - In 1987 the first substance to act as a...Ch. 3 - Prob. 44ECh. 3 - Prob. 45ECh. 3 - Vitamin B12 , cyanocobalamin, is essential for...Ch. 3 - Prob. 47ECh. 3 - Prob. 48ECh. 3 - Prob. 49ECh. 3 - Give the empirical formula of each of these...Ch. 3 - Determine the molecular formulas to which the...Ch. 3 - A sample of urea contains 1.121 g N, 0.161 g...Ch. 3 - Prob. 53ECh. 3 - The compound adrenaline contains 56.79% C, 6.56%H,...Ch. 3 - The most common form of nylon (nylon-6) is...Ch. 3 - Prob. 56ECh. 3 - Prob. 57ECh. 3 - Prob. 58ECh. 3 - Prob. 59ECh. 3 - Prob. 60ECh. 3 - Prob. 61ECh. 3 - Prob. 62ECh. 3 - Prob. 63ECh. 3 - Prob. 64ECh. 3 - Prob. 65ECh. 3 - Iron oxide ores, commonly a mixture of FeOand...Ch. 3 - Prob. 67ECh. 3 - Prob. 68ECh. 3 - Prob. 69ECh. 3 - Prob. 70ECh. 3 - Prob. 71ECh. 3 - Prob. 72ECh. 3 - Elixirs such as Alka-Seltzer use the reaction of...Ch. 3 - Prob. 74ECh. 3 - Prob. 75ECh. 3 - Prob. 76ECh. 3 - Prob. 77ECh. 3 - Bacterial digestion is an economical method of...Ch. 3 - Prob. 79ECh. 3 - Prob. 80ECh. 3 - Prob. 81ECh. 3 - Prob. 82ECh. 3 - Hydrogen peroxide is used as a cleaning agent in...Ch. 3 - Silver sulfadiazine burn-treating cream creates a...Ch. 3 - Bornite (Cu3FeS3) is a copper ore used in the...Ch. 3 - DDT, an insecticide harmful to fish, birds, and...Ch. 3 - Prob. 87ECh. 3 - Prob. 88ECh. 3 - Prob. 89ECh. 3 - Prob. 90ECh. 3 - Prob. 91AECh. 3 - Prob. 92AECh. 3 - A sample of a hydrocarbon (a compound consisting...Ch. 3 - Prob. 94AECh. 3 - Prob. 95AECh. 3 - The empirical formula of styrene is CH; the molar...Ch. 3 - A 0.755-g sample of hydrated copper(II) sulfate...Ch. 3 - Prob. 98AECh. 3 - Prob. 99AECh. 3 - Prob. 100AECh. 3 - Prob. 101AECh. 3 - Prob. 102AECh. 3 - Prob. 103AECh. 3 - Prob. 104AECh. 3 - Prob. 105AECh. 3 - Prob. 106AECh. 3 - Prob. 107AECh. 3 - Prob. 108AECh. 3 - Prob. 109AECh. 3 - Prob. 110AECh. 3 - Prob. 111AECh. 3 - Prob. 112AECh. 3 - Prob. 113AECh. 3 - Prob. 114AECh. 3 - Prob. 115AECh. 3 - Prob. 116AECh. 3 - Prob. 117AECh. 3 - Prob. 118AECh. 3 - Prob. 119AECh. 3 - Which of the following statements about chemical...Ch. 3 - Prob. 121AECh. 3 - Prob. 122AECh. 3 - Prob. 123CPCh. 3 - When the supply of oxygen is limited, iron metal...Ch. 3 - Element X forms both a dichloride (XCl2) and a...Ch. 3 - Zinc and magnesium metal each react with...Ch. 3 - An unknown binary compound containing hydrogen...Ch. 3 - A 2.25-g sample of scandium metal is reacted with...Ch. 3 - When M2S3(s) is heated in air, it is converted to...Ch. 3 - Consider a gaseous binary compound with a molar...Ch. 3 - Prob. 131CPCh. 3 - You take 1.00 g of an aspirin tablet (a compound...Ch. 3 - Lanthanum was reacted with hydrogen in a given...Ch. 3 - Prob. 134CPCh. 3 - Consider a mixture of potassium chloride and...Ch. 3 - Prob. 136CPCh. 3 - Prob. 137CPCh. 3 - A gas contains a mixture of NH3(g)andN2H4(g) ,...Ch. 3 - Prob. 139MPCh. 3 - Prob. 140MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forwardWhy isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forward
- What is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forwardComplete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forward
- Q Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forwardb. ὋΗ CH3CH2OH H2SO4arrow_forward
- For the reaction A (g) → 3 B (g), Kp = 0.379 at 298 K. What is the value of ∆G for this reaction at 298 K when the partial pressures of A and B are 5.70 atm and 0.250 atm?arrow_forward14. Calculate the concentrations of Ag+, Ag(S2O3), and Ag(S2O3)23- in a solution prepared by mixing 150.0 mL of 1.00×10-3 M AgNO3 with 200.0 mL of 5.00 M Na2S2O3 Ag+ + S20 Ag(S203)¯ K₁ = 7.4 × 108 Ag(S203)¯ + S20¯ = Ag(S203) K₂ = 3.9 x 104arrow_forwardΗΝ, cyclohexanone pH 4-5 Draw Enamine I I CH3CH2Br THF, reflux H3O+ I Drawing Draw Iminium Ionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY