
Principles of Instrumental Analysis, 6th Edition
6th Edition
ISBN: 9788131525579
Author: Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher: Cenage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 3, Problem 3.21QAP
Interpretation Introduction
Interpretation:
The location of the contact in a slide wire along its length for the circuit shown below should be identified.
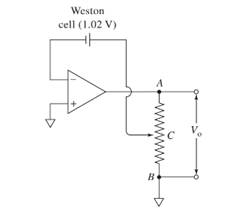
Concept introduction:
A voltage follower circuit is here with a Weston cell in the feedback loop. Since the op-amp has very high input resistance, the voltage from the Weston cell will appear across the output resistance part BC. The voltage across BC can be obtained using potential divider concept. Using this we can find the point at which the contact C is to be placed.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For each scenario below, select the color of the solution using the indicator thymol blue during the titration.
When you first add indicator to your Na2CO3solution, the solution is basic (pH ~10), and the color is ["", "", "", "", ""] .
At the equivalence point for the titration, the moles of added HCl are equal to the moles of Na2CO3. One drop (or less!) past this is called the endpoint. The added HCl begins to titrate the thymol blue indicator itself. At the endpoint, the indicator color is ["", "", "", "", ""] .
When you weren't paying attention and added too much HCl (~12 mL extra), the color is ["", "", "", "", ""] .
When you really weren't paying attention and reached the second equivalence point of Na2CO3, the color is
The following reaction is run in which the initial conditions include only methane (CH4) at a concentration of0.115 M. Once equilibrium was established, the concentration of acetylene (C2H2) was measured to be 0.035M. What is the value of the equilibrium constant, K?2 CH4 (g) ⇋ C2H2 (g) + 3 H2 (g)
Calculate the equilibrium concentration of carbon dioxide for the following reaction:2 COF2 (g) ⇋ CF4 (g) + CO2 (g) Kc = 2.00 at 10.00 °C. at equilibrium [COF2] = 0.255M; [CF4] = 0.118M
Chapter 3 Solutions
Principles of Instrumental Analysis, 6th Edition
Ch. 3 - Prob. 3.1QAPCh. 3 - Prob. 3.2QAPCh. 3 - Prob. 3.3QAPCh. 3 - Prob. 3.4QAPCh. 3 - Prob. 3.5QAPCh. 3 - Prob. 3.6QAPCh. 3 - Prob. 3.7QAPCh. 3 - Prob. 3.8QAPCh. 3 - Prob. 3.9QAPCh. 3 - Prob. 3.10QAP
Ch. 3 - Prob. 3.11QAPCh. 3 - Prob. 3.12QAPCh. 3 - Prob. 3.13QAPCh. 3 - Prob. 3.14QAPCh. 3 - Prob. 3.15QAPCh. 3 - Prob. 3.16QAPCh. 3 - Prob. 3.17QAPCh. 3 - Prob. 3.18QAPCh. 3 - Prob. 3.19QAPCh. 3 - Prob. 3.20QAPCh. 3 - Prob. 3.21QAPCh. 3 - Prob. 3.22QAPCh. 3 - Prob. 3.23QAPCh. 3 - Prob. 3.24QAPCh. 3 - Prob. 3.25QAP
Knowledge Booster
Similar questions
- In a benzene derivative that has -CH2CH3, indicate how it can be substituted by -COOH.arrow_forwardIn a sulfonated derivative of benzene, indicate how -SO3H can be eliminated.arrow_forwardWhat is the equilibrium expression (law of mass action) for the following reaction:CO2 (g) + H2O (l) ⇋ H+ (aq) + HCO3- (aq)arrow_forward
- Indicate the compound resulting from adding NaOH cyclopentane-CH2-CHO.arrow_forwardUse the provided information to calculate Kc for the following reaction at 550 °C: H2(g) + CO2(g) ⇌ CO(g) + H2O(g) Kc = ?CoO(s) + CO(g) ⇌ Co(s) + CO2(g) Kc1 = 490CoO(s) + H2(g) ⇌ Co(s) + H2O(g) Kc2 = 67arrow_forwardCalculate Kc for the reaction: I2 (g) ⇋ 2 I (g) Kp = 6.26 x 10-22 at 298Karrow_forward
- For each scenario below, select the color of the solution using the indicator thymol blue during the titration. When you first add indicator to your Na2CO3solution, the solution is basic (pH ~10), and the color is ["", "", "", "", ""] . At the equivalence point for the titration, the moles of added HCl are equal to the moles of Na2CO3. One drop (or less!) past this is called the endpoint. The added HCl begins to titrate the thymol blue indicator itself. At the endpoint, the indicator color is ["", "", "", "", ""] . When you weren't paying attention and added too much HCl (~12 mL extra), the color is ["", "", "", "", ""] . When you really weren't paying attention and reached the second equivalence point of Na2CO3, the color isarrow_forwardTo convert cyclopentane-CH2-CHO to cyclopentane-CH2-CH3, compound A is added, followed by (CH3)3CO-K+, DMS at 100oC. Indicate which compound A is.arrow_forwardIndicate how to obtain the compound 2-Hydroxy-2-phenylacetonitrile from phenylmethanol.arrow_forward
- Indicate the reagent needed to go from cyclopentane-CH2-CHO to cyclopentane-CH2-CH=CH-C6H5.arrow_forwardesc Write the systematic name of each organic molecule: structure CH3 CH3-C=CH2 CH3-CH2-C-CH2-CH3 CH-CH3 CH3 ☐ ☐ ☐ CI-CH-CH=CH2 Explanation Check F1 F2 name 80 F3 F4 F5 F6 A 7 ! 2 # 3 4 % 5 6 & 7 Q W E R Y FT 2025 Mcarrow_forwardTwo reactants X and Z are required to convert the compound CH3-CH2-CH2Br to the compound CH3-CH2-CH=P(C6H5)3. State reactants X and Z.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning